Abstract
Mogamulizumab is a monoclonal antibody that binds to C–C chemokine receptor 4 (CCR4), initiating antibody-dependent cellular cytotoxicity. CCR4 is highly expressed in the cutaneous T-cell lymphoma subtypes mycosis fungoides and Sézary syndrome (SS), and mogamulizumab has been shown to be effective in patients with these conditions who were refractory to at least one prior systemic treatment. One of the more common adverse events encountered with mogamulizumab is rash, which may mimic disease progression and lead to premature discontinuation. Moreover, there has been some evidence to suggest that mogamulizumab-associated rash (MAR) is associated with improved outcomes in some patients, particularly those with SS. This report presents the case of a 72-year-old woman with SS, which manifested with macular and papular lesions and abnormal blood cytometry, who was treated with mogamulizumab after failure of bexarotene and photopheresis combination therapy. She achieved a complete response (CR), but experienced lymphopenia associated with histologically proven eosinophilic folliculitis (EF) of the scalp and alopecia. The EF responded well to initial topical corticosteroids, defined by regression of erythema and pustular involvement and reduction in pruritus-like symptoms, but without hair regrowth. Mogamulizumab was withdrawn after 32 cycles, but CR was maintained. To date, EF persists in the form of diffuse erythema without pustules or pruritus. A link between cluster of differentiation 4 lymphopenia and EF has previously been established; therefore, EF should be considered in patients who develop rash and lymphopenia while receiving treatment with mogamulizumab. MAR has been associated with clinical response to mogamulizumab, and this case report adds to the evidence that EF may also be associated with sustained clinical response following treatment cessation. However, regular monitoring is required to prevent a relapse of SS. Prospective studies are needed to confirm whether such an association between EF and CR following mogamulizumab exists.
Keywords
Background
Cutaneous T-cell lymphomas (CTCLs) are a heterogeneous collection of non-Hodgkin lymphomas that present in skin and involve malignant populations of T lymphocytes. 1 The most common CTCL subtypes are mycosis fungoides (MF) and Sézary syndrome (SS). 1 C–C chemokine receptor 4 (CCR4) is consistently expressed on the surface of tumor cells in MF and SS, and is involved in cell trafficking of lymphocytes to the skin.2–5 Mogamulizumab is a monoclonal antibody that binds to CCR4 and is currently licensed in both the USA and Europe.6,7 Slight variations exist in the indications, such that mogamulizumab is indicated for the treatment of adult patients with MF or SS who have received at least one prior systemic therapy in Europe, 6 whereas the US indication specifies the treatment of adult patients with relapsed or refractory MF or SS after at least one prior systemic therapy. 7 Mogamulizumab (1 mg/kg intravenous weekly for the first 28-day cycle, then on days 1 and 15 of subsequent cycles; n = 186) was shown to be effective by virtue of superior progression-free survival (PFS) versus the comparator vorinostat (400 mg daily; n = 186), in patients with histologically confirmed relapsed or refractory MF or SS and who had failed at least one previous systemic therapy in the phase III MAVORIC study [median PFS, 7.7 months (95% confidence interval (CI), 5.7–10.3) versus 3.1 months (95% CI, 2.9–4.1); p < 0.0001].8,9 Moreover, in a real-world study of 21 patients with MF or SS treated with mogamulizumab, PFS was estimated at 22 months. 10
A commonly observed safety signal in the phase III MAVORIC trial was ‘drug eruption’, which was observed in 24% of patients in the mogamulizumab arm (n = 184) versus 1% of patients in the vorinostat arm (n = 186) of the trial. 8 This has since come to be known as mogamulizumab-associated rash (MAR); some patients experiencing MAR, particularly those with SS, had improved clinical responses compared with those without MAR.11-13 One explanation for this phenomenon is the immunomodulatory effect of mogamulizumab contributing to the restoration of efficient immunity through reduction of the number of CCR4+ malignant cells and benign T cells [including exhausted lymphocytes and activated regulatory T cells (Tregs)] and subsequent emergence of cluster of differentiation (CD)8+, naïve and stem cell memory CD4+ T cells.11-13
Because mogamulizumab works by depleting CCR4+ T lymphocytes,6,7,12,13 lymphopenia [an absolute lymphocyte count (ALC) <1000/mm3] is considered to be a pharmacologic effect of the drug.9,14 Indeed, results from the phase I/II study of mogamulizumab in previously treated MF and SS noted that 75% of patients treated with mogamulizumab (n = 42; 1 patient with peripheral T cell lymphoma was also included in the safety analysis) had a reduction in lymphocyte count following treatment with mogamulizumab; 41% of these patients experienced grade III lymphopenia and 26% of patients experienced grade IV lymphopenia. 14 Lymphopenia can, in some cases, cause general immunosuppression, 15 and an immunocompromised state is a risk factor for an immunosuppression-associated form of eosinophilic folliculitis (EF). 16 Furthermore, low CD4 cell count has been associated with the development of EF in patients with human immunodeficiency virus.17,18 This report presents the case of a patient who experienced clinically confirmed EF following mogamulizumab treatment for SS, associated with lymphopenia and immune response. The reporting of this study conforms to the CARE guidelines. 19
Patient information
A 72-year-old female patient was referred with a history of MF (diagnosed in 2004 at 54 years of age), which presented with skin patches and pruritus, and evolved into secondary SS in 2018 (Supplemental Figure S1).
She received chlormethine, topical steroids, and ultraviolet light therapy for MF in 2004, which was stopped in 2017 following symptom relapse. In April 2018, abnormal flow cytometry (FC; 70% abnormal T lymphocytes) and skin patches on the patient’s arm and abdominal and lumbar regions led to initiation of bexarotene (increased from 225 to 300 mg/m2/day after 15 days) and photopheresis. The patient was subsequently diagnosed with secondary SS in July 2018. As a result of neutropenia, bexarotene and photopheresis combination therapy was stopped in September 2018 and then reinstated at the initial reduced dosage of 225 mg/m2/day in November 2018. In February 2019, FC revealed that 85% abnormal T cells were present (approximately 6.31 G/L) out of 255,000 lymphocytes analyzed per tube. These cells were CD30− and had a CD4:CD8 ratio of 88 (an established marker of blood involvement and clinical outcome in MF/SS;10,20 FC also revealed abnormal cells that were CD4+/CD8−/CD7−/CD26−/CD3+ low/CD5+/CD2+ low), consistent with the diagnosis of SS. A computed tomography (CT) scan at this time revealed no abnormal findings.
After the discontinuation of bexarotene and photopheresis in March 2019 due to neutropenia that did not resolve with dose reduction, the patient suffered a relapse of SS. Examinations showed macular and papular lesions on her elbow, left leg, and scalp; blood counts of 9467 lymphocytes/mm3 and 1242 neutrophils/mm3; and 85% abnormal T cells, with a CD4:CD8 ratio of 88 on FC. The multidisciplinary team discussed the case and agreed to initiate mogamulizumab 1 mg/kg (weekly on days 1, 8, 15, and 22 of the first 28-day cycle, followed by infusions every 2 weeks on days 1 and 15) from April 2019. Follow-up FC and precision immune phenotyping assessments were performed at least every 3 months and imaging was undertaken annually from July 2019 to March 2022.
Clinical findings
Mogamulizumab was associated with treatment benefits from initiation in April 2019, complete response (CR) in the skin was achieved, defined by the absence of patches, by July 2019. Furthermore, a reduction of abnormal T cells by FC to a level below the limits of detection (among 60,000 analyzed) and a CD4:CD8 ratio of 2.72 indicated CR in the blood compartment in July 2019. At the twelfth infusion of mogamulizumab (August 2019), grade III lymphopenia (ALC 122 cells/mm3) was observed during routine blood testing and was followed by the onset of an itchy pustular rash of the scalp in October 2019. FC of the peripheral blood showed no abnormal T cells, a CD4/CD8 ratio of 2.9, low CD2+ and CD3+ expression, and CD3−, CD7−, and CD26− cells were not present. Immunohistochemical analysis of the scalp lesions performed in October 2019 revealed diffuse lymphohistiocytic infiltrate and CD3+ and CD20+ expression; CD8+ cells were predominant versus CD4+ cells, with no CD30+ cells observed. Alcian blue and periodic acid-Schiff staining (the antibodies used are detailed in Supplemental Table S1) did not reveal the presence of mucin or pathogens within the follicular sheath. The rash was subsequently associated with the development of alopecia after a few weeks (Figure 1).
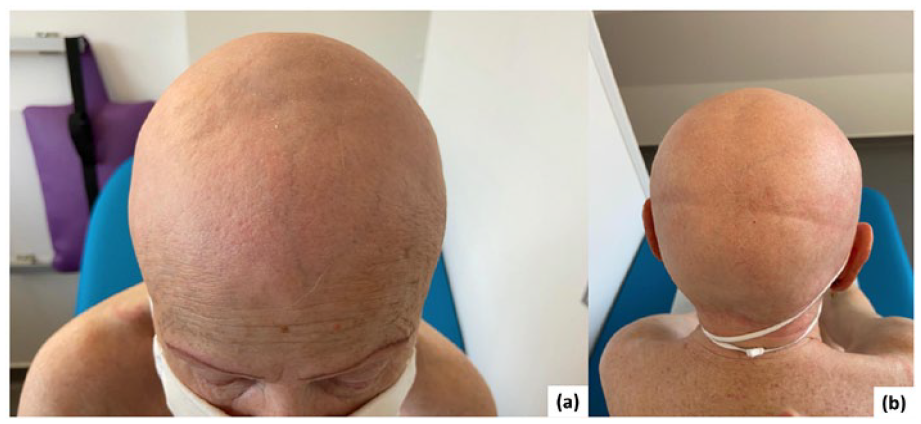
After 12 infusions of mogamulizumab, the patient developed secondary alopecia. (a) Front view. (b) Back view. No images of the first pustular rash are available.
Therapeutic interventions
Following the onset of the itchy pustular rash of the scalp and alopecia after initiation of treatment with mogamulizumab, topical clobetasol propionate, chlormethine, and 5% salicylic acid in petroleum jelly were prescribed ad libitum in October 2019.
Diagnostic assessment
Low levels of CD4+ cells (283 cells/mm3) were first recorded in January 2020 following initiation of routine blood monitoring every 15 days, confirming the lymphopenia observed in August 2019. In April 2020, an anatomopathologic and polymerase chain reaction analysis of the skin biopsy from the scalp determined that the cells were negative for T-cell receptor rearrangement, suggesting the cause of the skin symptoms was an eosinophilic immune response. Histologic analysis revealed lesions of the hair infundibulum that had become vesicular and contained infiltrating eosinophils, neutrophils, and lymphocytes (Figure 2). Extension of lesions into the dermis, sebaceous glands, and arrector pili muscles was also noted. Together, these results verified the suspected diagnosis of EF (rather than SS relapse); based on these findings, alopecia totalis and alopecia areata (AA) were excluded as potential diagnoses.
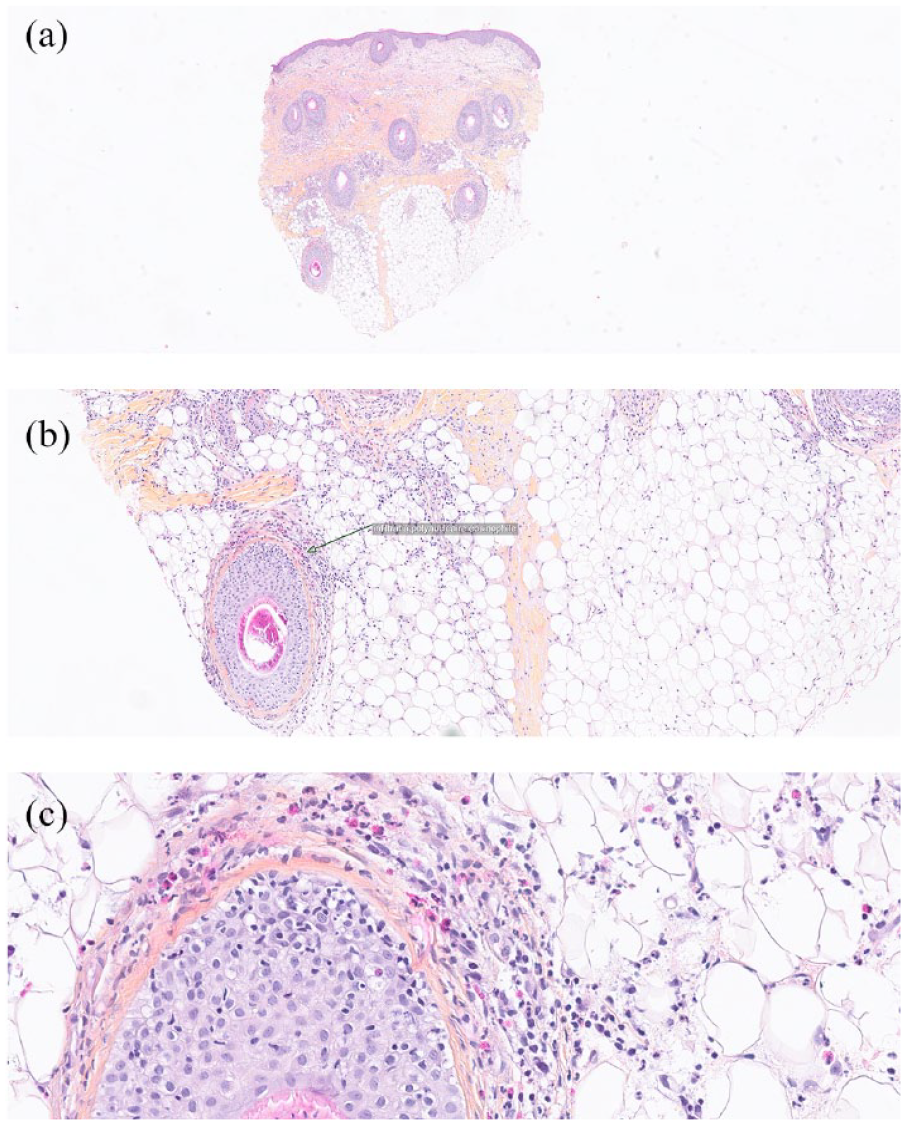
Histologic images of scalp skin tissue biopsy used to help exclude SS recurrence as a potential diagnosis. (a) Edema and perifollicular infiltrate infiltrating the hypodermis: HES-stained tissue (low magnification: 10×). (b) Eosinophil-rich infiltrate dissociating the follicular sheath (indicated by arrow): HES-stained tissue (medium magnification: 20×). (c) Eosinophil-rich infiltrates dissociating the follicular sheath: HES-stained tissue (high magnification: 40×).
Follow-up and outcomes
The patient’s EF responded well to topical clobetasol propionate, chlormethine, and 5% salicylic acid in petroleum jelly. A clear regression of erythema, pustular involvement, and pruritus-like symptoms was observed, but without hair regrowth. Some EF of the scalp has persisted (diffuse erythema without pustules or pruritus); treatment with clobetasol propionate, chlormethine, and 5% salicylic acid in petroleum jelly was stopped in April 2020 due to the symptoms of EF not fully resolving. Clobetasol propionate for ad libitum topical use due to inflammatory lesions was re-prescribed in February 2021.
In May 2021, immune-related hyperthyroidism was diagnosed on the basis of underlying biologic readings [thyroid-stimulating hormone 0.011 mU/L and T4 24.1 ng/L; anti-thyroid peroxidase antibodies four times normal value; two Thyroid Imaging Reporting and Data Systems (TI-RADS) 2 nodules of 7 mm, measured using echography]; thyroid scintigraphy was performed in June 2021 to confirm the diagnosis. The patient received carbimazole 20 mg/day to treat immune-related hyperthyroidism and propranolol 20 mg twice daily from June 2021 to treat tachycardia; this regimen continues to date. CT scan revealed no abnormal findings, and no adenomegaly was noted.
Skin biopsies in July and August revealed superficial and spongiform dermatitis; while the presence of rare, atypical lymphocytes was noted, this was insufficient to confirm SS relapse. In July, the results of a positron emission tomography (PET) scan were negative, with no suspicious hypermetabolic activity. In August 2021, FC revealed that out of 100,000 lymphocytes analyzed per tube, there were no abnormal T lymphoid cells (CD4+/CD8−/CD7−/CD26−/CD3+ low/CD5+/CD2+ low); CD4:CD8 ratio was 7.4. As a result of mogamulizumab treatment, skin CR and normal CD4:CD8 ratios were maintained, and no imaging abnormalities were noted on annual CT and PET scans. Mogamulizumab was discontinued after 32 cycles of treatment in September 2021 due to temporary loss of local reimbursement.
The patient subsequently received maintenance bexarotene 300 mg/m2/day from September 2021, with the aim of sustaining the good therapeutic response achieved with mogamulizumab. Five months following cessation of mogamulizumab (February 2022), of 75,000 lymphocytes analyzed per tube there were no abnormalities in T-cell status (CD4+/CD8−/CD7−/CD26−/CD3+ low/CD5+/CD2+ low); CD4:CD8 ratio was 4.7. PET-CT revealed no suspicious individualizable hypermetabolic activity over the entire explored volume. Due to liver dysfunction, maintenance bexarotene was stopped in March 2022. At the last follow-up in March 2022, the patient had maintained CR as assessed by FC and PET-CT and has ongoing complete alopecia with EF persisting as diffuse erythema without pustules or pruritus. Clobetasol propionate for ad libitum topical use prescribed for the scalp rash has not been used to date.
Discussion
This report details the case of a patient with refractory SS who was treated effectively with mogamulizumab. The patient developed EF and alopecia on the scalp, associated with immune response and lymphopenia, but achieved a long-term blood and skin CR that was sustained even after mogamulizumab withdrawal.
The onset of lymphopenia in this patient is consistent with previous observations of its association with mogamulizumab. 14 As an on-target effect, lymphopenia might be expected to be associated with mogamulizumab efficacy. The effect of mogamulizumab on lymphocytes and the need for continued treatment should be regularly monitored by FC, as evidenced by a retrospective study of 13 patients with CTCL, who experienced a decrease in aberrant T-cell populations even after one dose of mogamulizumab. 21
Immunosuppression, particularly CD4+ T-cell deficiency, is a risk factor for EF.16–18 EF diagnosed in this patient was associated with immunosuppression with a non-infectious inflammation of the pilosebaceous follicles that falls within a clinically and histologically heterogeneous group of skin conditions known as eosinophilic dermatoses.22–25 These eosinophilic skin disorders arise from immune activation mediated by the overproduction of interleukin-5, which is involved in chemotaxis, maturation, and survival of eosinophils.23,26 Upon activation, eosinophils release inflammatory proteins, including cationic proteins (major basic protein, eosinophil cationic protein, and eosinophil peroxidase).23,26,27 Of note, while cationic proteins are antiparasitic, antibacterial, and antiviral, they are also cytotoxic. 28 Indeed, folliculitis has been identified as a potential adverse event associated with mogamulizumab, and perivascular lymphocytic infiltrate with eosinophils has been associated with the occurrence of MAR.6,14
Following mogamulizumab initiation in this patient, lymphopenia preceded the onset of a scalp rash by 2 months. The rash was later diagnosed as EF. Therefore, it may be the case that in this patient, mogamulizumab induced EF by reducing the ALC. Given the patient’s favorable response to mogamulizumab in the presence of a rash, this case supports existing evidence that has suggested that MAR may be associated with a better response to treatment, especially in SS.11,12 MAR commonly exhibits distinct granulomatous/histiocytic, psoriasiform/spongiotic, or lichenoid histologic patterns, and MAR is attributed to chemokine (C–X–C motif) ligand (CXCL) 9 and CXCL11 overexpression.12,29,30 It has been suggested that predominance of CD8+/CD7+ expression distinguishes MAR from the inherent disease pathology.29,30 Indeed, biopsy results in our patient showed that the rash contained diffuse lymphohistiocytic infiltrate and a CD3+/CD8+ high/CD4+ low T-cell profile. It should be noted that, as far as the author is aware, there are no cases of mogamulizumab-associated EF on the scalp in the literature to date, and therefore the underlying pathophysiologic mechanisms of EF remain to be determined.
Other reports have postulated that other conditions associated with immune modulation may also be associated with better outcomes following mogamulizumab treatment.
AA is an autoimmune disease; its exact pathophysiological mechanisms are unknown, but multiple immunomodulators are thought to be involved. 31 A retrospective analysis of AA occurring in patients treated with mogamulizumab from 31 centers of the French Group for the Study of Cutaneous Lymphomas revealed that AA has been reported during mogamulizumab treatment (4 cases of 157 patients). 32 AA may be a consequence of absent Treg-mediated inhibition of an autoimmune attack. 33 Accordingly, it has been suggested that conditions such as AA that are associated with immunomodulation during mogamulizumab treatment could be associated with better clinical outcomes. 32 In the author’s clinical experience, AA or alopecia totalis are excluded as underlying diagnoses for the eosinophilic infiltrate, given that pustular lesions rarely precede development of the alopecia totalis, while AA appears without pre-existing pustular lesions. A possible association between autoimmunity manifestations and long-term complete remission of SS after mogamulizumab treatment has also been reported. 34
Supporting this evidence, there appears to be a connection between EF and positive clinical outcomes in this case. Prospective studies are needed to confirm an association between EF and CR following mogamulizumab. Given the timeline of lymphopenia and EF development in this patient following mogamulizumab treatment, monitoring of rashes in patients with lymphopenia is advised, along with regular FC analyses. Patients with EF and CR must also be monitored for signs of SS relapse. Moreover, the potential pathophysiologic mechanisms behind this connection between previously unreported EF and response to mogamulizumab warrant prospective investigation in a larger patient population.
Patient perspective
Patient’s testimony by phone, 9 December 2022
Overall, the patient reported she was doing well until the discontinuation of mogamulizumab.
Throughout the treatment, the patient reported complete confidence in her care plan. She appreciated the contact with the oncology nurses to manage adverse events. There were no problems with fatigue, except for the thyroid problems she had at the beginning of 2021. She had no other side effects and no issues with putting the corticosteroid creams on her scalp. The patient used to be an elite swimmer and continued to go to the pool twice a week; she reported never feeling socially impacted by the display of dermatosis.
Supplemental Material
sj-jpg-1-tah-10.1177_20406207241235777 – Supplemental material for An occurrence of eosinophilic folliculitis and alopecia associated with a sustained complete response to mogamulizumab in Sézary syndrome: a case report
Supplemental material, sj-jpg-1-tah-10.1177_20406207241235777 for An occurrence of eosinophilic folliculitis and alopecia associated with a sustained complete response to mogamulizumab in Sézary syndrome: a case report by Jean-Matthieu L’Orphelin in Therapeutic Advances in Hematology
Supplemental Material
sj-jpg-2-tah-10.1177_20406207241235777 – Supplemental material for An occurrence of eosinophilic folliculitis and alopecia associated with a sustained complete response to mogamulizumab in Sézary syndrome: a case report
Supplemental material, sj-jpg-2-tah-10.1177_20406207241235777 for An occurrence of eosinophilic folliculitis and alopecia associated with a sustained complete response to mogamulizumab in Sézary syndrome: a case report by Jean-Matthieu L’Orphelin in Therapeutic Advances in Hematology
Supplemental Material
sj-pdf-3-tah-10.1177_20406207241235777 – Supplemental material for An occurrence of eosinophilic folliculitis and alopecia associated with a sustained complete response to mogamulizumab in Sézary syndrome: a case report
Supplemental material, sj-pdf-3-tah-10.1177_20406207241235777 for An occurrence of eosinophilic folliculitis and alopecia associated with a sustained complete response to mogamulizumab in Sézary syndrome: a case report by Jean-Matthieu L’Orphelin in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The author would like to thank the patient and their family for providing consent to use their medical information to make this article possible.
Third-party submissions
This manuscript was submitted by Kyowa Kirin International on behalf of the author. No fees for service or honoraria were paid to the author by Kyowa Kirin per Good Publication Practice guidelines.
Writing assistance
Editorial support in the preparation of this manuscript was provided by Shireene Kalbassi, PhD of Infinity, OPEN Health Medical Communications, with financial support from Kyowa Kirin International in accordance with Good Publication Practice guidelines (www.ismpp.org/gpp-2022); the author retained complete editorial control over the content.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
