Abstract
Background:
Adult T-cell leukemia/lymphoma (ATL) is a neoplasm with a high prevalence in certain regions such as southwestern Japan, the Caribbean, sub-Saharan Africa, South America, and Australia. In Colombia, the seroprevalence of human T-cell lymphotropic virus type 1 (HTLV-1) has been reported in specific populations, but there is limited information about the clinical course and management of ATL in the country.
Objectives:
To describe the clinical characteristics, treatment patterns, and outcomes of patients diagnosed with HTLV-1-positive ATL in a high-complexity healthcare institution in Colombia and compare these findings with reports from other geographic regions.
Design:
Observational retrospective cohort study conducted in a single high-complexity healthcare institution in Cali, Colombia, including patients diagnosed between 2011 and January 2022.
Methods:
This study presents an observational retrospective cohort of patients diagnosed with HTLV-1-positive ATL and managed at a high-complexity institution in Cali, Colombia. Eligible patients were adults diagnosed with ATL and HTLV-1 seropositivity, receiving treatment between 2011 and January 2022. Demographic, clinical, and treatment-related variables were collected and analyzed using descriptive and survival analyses.
Results:
Thirty-three patients diagnosed with ATL between 2011, and January 2022 were identified and included in the study. Most patients were female (52%) with a median age of 54 years. Acute presentation was the most common (64%), and most patients were identified at Ann Arbor stage 4. Treatment mainly consisted of various chemotherapy protocols, with cyclophosphamide, doxorubicin hydrochloride, vincristine sulfate, and prednisone (CHOP)-like regimens being the most frequently used. However, the overall response rate to chemotherapy was low, and most patients experienced complications and toxicities associated with treatment.
Conclusion:
This study provides insights into the clinical course and management of ATL in a Colombian population. The findings highlight the predominance of acute presentations, advanced disease stages at diagnosis, and challenges in achieving a complete response with conventional chemotherapy. Further research is needed to improve treatment strategies, identify prognostic markers, and develop more effective therapies for ATL patients in Colombia.
Background
Adult T-cell leukemia/lymphoma (ATL) is a neoplastic disease that affects the southwestern region of Japan, the Caribbean, sub-Saharan Africa, South America, and Australia. 1 It is classified into four clinical presentations: acute, lymphoma, chronic, and smoldering. 2 The acute, lymphoma, and chronically aggressive forms (chronic-type ATL with unfavorable prognostic factors) require differential treatment with intensive therapy to achieve longer survival time and a better rate of partial and complete response to chemotherapy.3,4
In Colombia, the seroprevalence of human T-cell lymphotropic virus (HTLV) was identified as 1% for human T-cell lymphotropic virus type 1 (HTLV-1) and 0.7% for HTLV-2 among the pre-Colombian Emberá and Paez tribes.5,6 Additionally, a combined seroprevalence of 0.24% for HTLV-1 and HTLV-2 (using the chemiluminescent microparticle immunoassay method on the automated Architect System platform) was reported in a study analyzing 77,117 donor records over a 6-year period. 7
Although most individuals infected with HTLV-1 remain asymptomatic, approximately 5% progress to ATL after a prolonged clinical latency. Historically, this latency period had been described as approximately 30 years8–10; however, recent studies suggest a higher average age at diagnosis. Some references report latency periods extending to four to five decades, indicating variability influenced by both host and viral factors.11–13 These discrepancies underscore the need to reassess earlier estimates and incorporate modern epidemiological trends to better understand the development of ATL.
In our country, a study was conducted to investigate the geographic epidemiology of lymphoid neoplasms in Cali and southwestern Colombia, with a particular focus on ATL. The study found that 5 out of 75 non-Hodgkin lymphoma cases were positive for HTLV-1 proviral sequences by PCR in tissue specimens, despite none of the patients having undergone serological testing for HTLV. Additionally, in Tumaco, Colombia, 356 samples from native volunteers were analyzed for antibodies against HTLV-1/2 using an enzyme-linked immunosorbent assay (ELISA) and confirmed by Western blot. Of these, 18 individuals (5.1%) tested seropositive for HTLV-1 antibodies. 14 Despite the high seroprevalence of HTLV-1 in our population, very few cases of ATL have been reported in the local literature.15,16
Aggressive variants of the disease, despite high-intensity chemotherapy, have an average survival of only 8 months.2,15 This study describes the progression of patients diagnosed with HTLV-1-positive leukemia/lymphoma in a region with a high prevalence of the virus. These patients were managed at a highly complex healthcare institution in Cali, Colombia. The main objective is to compare the clinical outcomes of these patients with those from other geographical regions, considering the low prevalence of the disease.
Materials and methods
This is an observational study of a retrospective cohort of patients diagnosed with ATL, who were managed at a single clinical center: Fundación Valle del Lili.
The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement, ensuring comprehensive and transparent reporting of observational research.17,18
The eligibility criteria included patients over 18 years of age, diagnosed with adult T-cell lymphoma/leukemia based on compatible immunohistochemistry findings and HTLV seropositivity (was determined using an ELISA test that simultaneously detects the presence of antibodies against HTLV-1 and HTLV-2), and Shimoyama criteria, diagnosed between 2011 and January 2022, that received treatment at the institution. The data were obtained from medical records.
Variables
Variables of demographic characteristics of the subjects and clinical characteristics prior to treatment were collected, including disease classification, Lugano staging, functional status, among others. Additionally, variables related to therapy were collected, such as type of treatment, number of cycles, complications, and outcomes.
Statistical analysis
Ten percent of the data was randomly taken and compared against the source documents, with no evidence of inconsistencies. An exploratory analysis of the data was performed to verify and correct extreme values. In a descriptive analysis of the clinical and pathological characteristics, measures of central tendency and interquartile ranges were used, based on the distribution of each variable, determined by the Shapiro–Wilk test.
The Kaplan–Meier method was used to estimate overall survival, considering the event of death, and survival was evaluated according to the received therapy. Univariate and multivariate Cox regression analysis was used to determine factors associated with survival.
Results
Thirty-three patients with ATL between 2011 and 2022, were included. Regarding the sociodemographic description, most of the patients were female (52%), with a median age at diagnosis of 54 years (see Table 1). The most common cities of origin were Cali, accounting for 36% of cases, and Buenaventura, accounting for 30% of cases. Among the patients, 42% (14) did not report any comorbidity, while among those who did, the most frequent comorbidity was hypertension, present in 33% of cases. Among the four patients who had an oncological comorbidity, three were in remission from solid neoplasms, including two with prostate adenocarcinoma and one with colon adenocarcinoma. One patient had stage IIB prostate adenocarcinoma and active disease at the time of diagnosis of HTLV-1-positive leukemia/lymphoma.
Characteristics of patients with HTLV-1 and ATL between 2011 and 2022 (n = 33).

ATL, adult T-cell leukemia/lymphoma; HTLV-1, human T-cell lymphotropic virus type 1.
Figure 1 illustrates the presenting symptoms of the patients in this study. It should be noted that some patients reported multiple symptoms, resulting in their inclusion in more than one category. Constitutional symptoms emerged as the most prevalent, followed by the presence of lymphadenopathy in cervical, axillary, or inguinal regions. Gastrointestinal symptoms encompassed abdominal pain, chronic diarrhea, nausea, and vomiting. Neurological manifestations included headache, visual disturbances, and syncope. Specific masses and lesions were identified in the mediastinum, liver, and as lytic bone lesions. Additionally, a subset of patients presented with symptoms associated with opportunistic infections, notably strongyloidiasis and salmonellosis.

Symptoms at hospital admission.
The most common presentation of the disease was the acute presentation, representing 64% of the patients. Most of the patients (74%) were identified at stage Ann Arbor 4 at the time of diagnosis, but with good reported functionality, as 85% were classified as ECOG 0 or 1.
Coinfection with other pathogens were identified in 10 patients, including hepatitis B, syphilis, and tuberculosis, all with a low proportion. Five patients showed coinfection with Epstein–Barr virus (EBV) by immunohistochemistry, of whom three had an acute clinical presentation and two had a lymphoma type.
Regarding the biological markers at the time of diagnosis, the median albumin level in the included patients was 3.3 mg/dL. It was found that 64% (21) of the patients had hypoalbuminemia. On the other hand, 48% (16) had hypercalcemia, with 11 having an acute presentation and 5 having a lymphomatous presentation. According to the clinical presentation, the median peripheral lymphocyte count in lymphomatous disease was 1590 cells/µL, while in acute form, it was 9474 cells/µL.
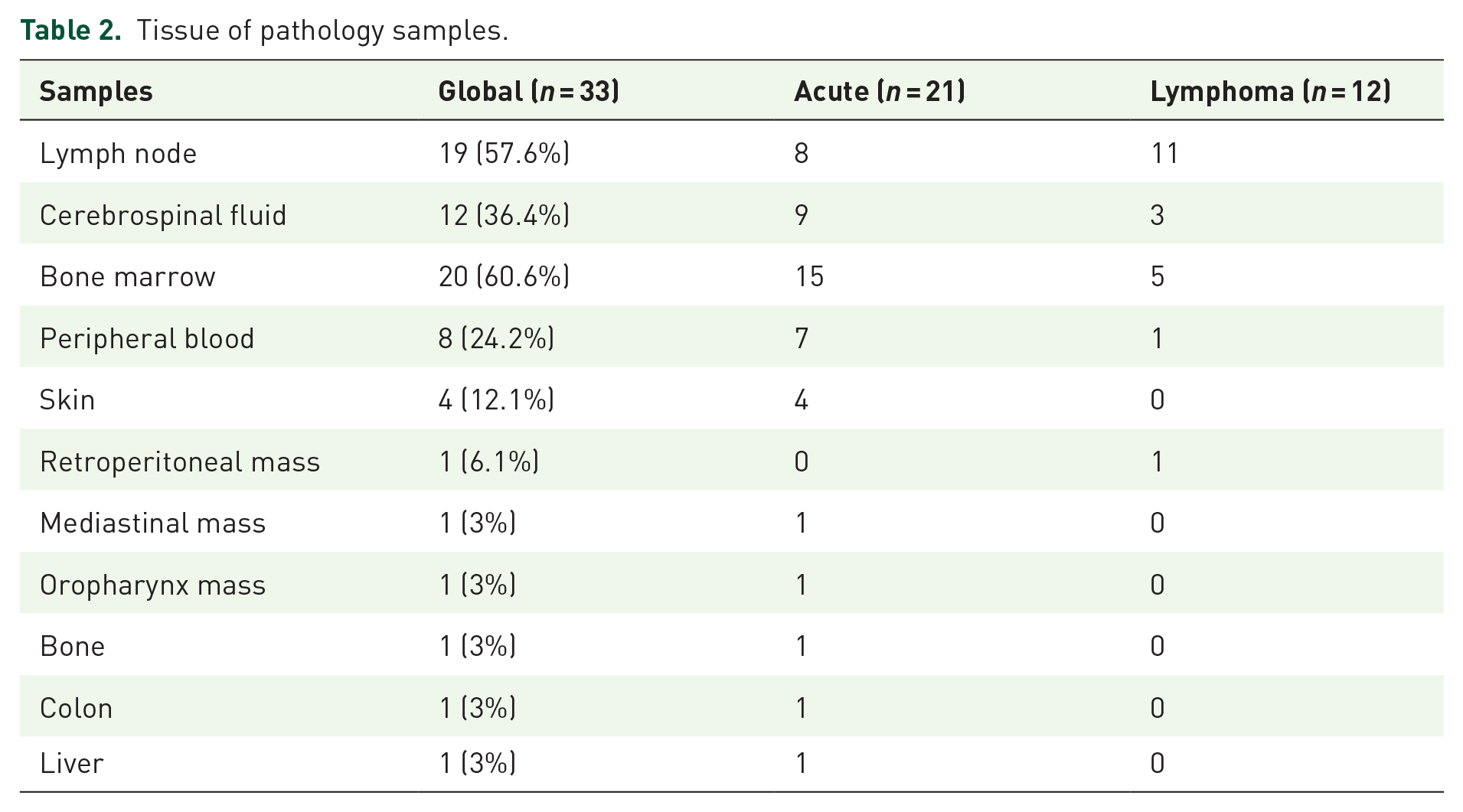
The histopathological diagnosis was performed on various tissues, as described in Table 2. The most frequent samples were lymph nodes (57.6%), bone marrow (60.6%), cerebrospinal fluid (36.4%), and peripheral blood (24.2%). Of the 33 patients, 26 (78.7%) had some form of extranodal involvement, with the majority presenting with an acute form of the disease and extension to other tissues such as skin, bone, colon, and liver.
Tissue of pathology samples.
Regarding the results of immunohistochemistry, the most prevalent immunophenotype was CD4+, CD3+, and CD5+. Only three patients were double positive (CD4 positive and CD8 positive). CD25 was expressed in 78.7% (26) of the patients, and CD30 was expressed in 25% of the patients (Table 3).
Results of immunohistochemical characterization from the pathology reports.

Immunohistochemistry frequency distribution. Green: Most frequent; Yellow: Intermediate frequency; Orange: Low frequency.
Ninety-one percent (30) of the patients received disease-specific management with different chemotherapy protocols. Sixty percent of them received cyclophosphamide, doxorubicin hydrochloride, vincristine sulfate, and prednisone (CHOP) or CHOP-like protocols, with CHOP being the most common, followed by etoposide phosphate, prednisone, vincristine sulfate, cyclophosphamide, and doxorubicin hydrochloride (EPOCH) and modified DAEPOCH (see Table 4). Five patients received modified LSG15 (vincristine, cyclophosphamide, doxorubicin, prednisolone, etoposide, carboplatin, methotrexate, and folinic acid). Ten patients received second-line chemotherapy due to primary refractoriness or relapse after achieving complete response. The most common second-line regimens were DHAP (three patients) and modified LSG15 (two patients).
Treatment protocols used in HTLV-1 positive ATL.

ATL, adult T-cell leukemia/lymphoma; CHOP, cyclophosphamide, doxorubicin hydrochloride, vincristine sulfate, and prednisone; EPOCH, etoposide phosphate, prednisone, vincristine sulfate, cyclophosphamide, and doxorubicin hydrochloride; HTLV-1, human T-cell lymphotropic virus type 1.
Only four patients achieved complete response. Two patients achieved complete response in the first line, one receiving HYPERCVAD (cyclophosphamide, vincristine, doxorubicin, dexamethasone which alternate with cycles of high-dose methotrexate and high-dose cytarabine) and the other receiving CHOP. Another patient achieved complete response in the second line after receiving DAEPOCH and modified LSG15. The fourth patient achieved complete response in the third line after receiving CHOP, modified LSG15, and the MATRIX (methotrexate, cytarabine, thiotepa, and rituximab) protocol.
As of now, three patients are still alive. One of them underwent allogeneic hematopoietic stem cell transplantation (from an HLA-identical donor with negative serology for HTLV-1) and has remained disease-free for 5 years. The second patient was not eligible for transplantation due to being over 65 years old and also had active management for prostate cancer. The third patient has indolent progression with refractory cutaneous involvement despite multiple lines of treatment. The patient who passed away was a candidate for transplantation, but it could not be performed due to the inability to mobilize progenitor cells and the unavailability of an allogeneic donor.
During the follow-up period, 73% of the patients experienced febrile neutropenia associated with treatment, with isolation of Gram-negative microorganisms in 36% of cases. No patients in this cohort had fungal isolates in blood cultures, and only one case of SARS-CoV-2 infection and one case of Mycobacterium tuberculosis infection were reported. Nineteen patients developed acute renal failure. Chemotherapy had to be modified or suspended due to infections (21%) and critical illness with or without multiple organ failure (27%) in 64% of the patients. Among the patients, 75.8% (25) experienced neutropenia with a count below 500 cells/µL, 84.8% (28) had thrombocytopenia, 54.5% (18) had elevated total bilirubin, 84.8% (28) had elevated alanine aminotransferase, and 87.9% (29) had elevated alkaline phosphatase due to treatment toxicity. The most frequent electrolyte imbalance was hypokalemia in 84.8% (28) of the patients, followed by hyponatremia in 69.7% (23) and hypercalcemia in 63.6% (21). During the follow-up, 91% of the patients died, with 75.7% attributed to disease progression and 15.1% lacking data on the cause of death (see Table 5).
Complications during the follow-up period.

After 12 months of follow-up, the median overall survival was 5 months (IQR 2–9 months). At 5 months of follow-up, less than 50% of patients were alive, and at 9 months, only 25% remained alive (see Figure 2).

Kaplan–Meier analysis of overall survival. Time in months.
The observed time at risk was 135 months, and there were 0.17 deaths per patient-month among those diagnosed with ATL.
No statistically significant change in survival was found according to the Ann Arbor classification at diagnosis (p = 0.211; Figure 3).

Kaplan–Meier analysis of survival according to Ann Arbor staging. Time in months.
Regarding the ATL-PI (Prognostic Index for Adult T-cell Leukemia/Lymphoma), 3 the following findings were observed: the mean albumin level for the evaluated population was 3.3 mg/dL (range 2.86–3.57), the calcium level was 10.1 mg/dL (range 9.0–14.5), and the average peripheral lymphocyte count was 4180/mm3 (range 1815–17,455). In this population, none of these factors influenced survival in both univariate and multivariate analyses. Even after performing the variable iteration process, only the variable “Ann Arbor classification” remained significant.
Discussion
This study shows the clinical and epidemiological behavior of aggressive presentations (acute leukemic and lymphomatous) of HTLV-1-positive leukemia/lymphoma in a population with a high prevalence of HTLV-1 infection. It highlights the poor prognosis and short survival associated with these presentations. It represents the largest sample of ATL patients published in the country. Prior to 1990, only 14 cases of ATL had been reported at the Hospital Universitario del Valle. All cases had a poor prognosis and originated from the Colombian Pacific region (Buenaventura, López de Micay, Cali, Barbacoas, Tumaco). 16 In our case, all patients also originated from the Pacific region, with residence in Valle del Cauca (75.1%, Cali: 36%, Buenaventura: 30%), Cauca (18%), and Nariño (6.1%), which is consistent with the global literature where HTLV-1 infection has a high prevalence in the southwestern Pacific coast of Colombia, specifically in Tumaco, with studies reporting prevalences of up to 5.3%.19,20
Coinfection with other pathogens were identified in 10 patients in this study. It has been reported that patients infected with HTLV are at higher risk for other sexually transmitted infections. 21 However, in this series, no differences were observed in coinfection rates for hepatitis B and syphilis compared to the general Colombian population.22,23 Additionally, coinfection with M. tuberculosis, which has a higher prevalence in the Colombian population, was less frequent.24,25 In this study, the most frequent coinfection was due to EBV.
In a retrospective analysis of 253 Latin American ATL patients (including 17 Colombian patients diagnosed and managed in Cali), the majority (n = 211; 83%) had aggressive ATL (lymphomatous: n = 127, 50%; acute: n = 83, 33%), and 31 (12%) had indolent ATL (chronic: n = 26, 10%; smoldering: n = 5, 2%). In this series, the most frequent presentation was the acute form, representing 64% of the patients, with no chronic or indolent variants identified.
The age of presentation in our study (54 years) was older compared to early local reports from Colombia (38.5 years) but aligned more closely with findings from other Latin American countries.16,26,27 A similar trend has been observed in Japan, the country with the highest number of ATL cases worldwide. According to a nationwide survey of ATL conducted in Japan and published in 2011, the mean age at diagnosis has steadily increased over the decades. In the first survey (cases before 1980), the mean age was 52.7 years, rising to 61.1 years in the ninth survey (1996–1997) and further to 66.0 years in the most recent report (range: 19–94, median: 67 years). 13 This shift likely reflects the impact of improved public health measures and screening programs designed to reduce vertical transmission of HTLV-1. Key interventions include nationwide prenatal screening and counseling for HTLV-1-positive mothers, as well as recommendations to avoid breastfeeding, which have significantly decreased the rate of new infections in younger populations.11,13,28 These changes underscore the role of healthcare infrastructure, increased public awareness, and targeted interventions in altering the epidemiological patterns of ATL globally.
For the acute variants, poor prognostic factors have been described, including functional status, age over 40 years, presence of more than three lesions, and hypercalcemia. Each factor has an odds ratio for mortality of 3.4, 2.1, 1.8, and 1.5, respectively. 29 In our cohort, there was a higher risk of mortality despite having an adequate functional status (ECOG 0 in 67%) and average calcium levels within the normal range.1,30 The average age was 54 years, and the majority presented with advanced disease (Ann Arbor stage 4 in 79%). Additionally, the presence of EBV infection, which can accelerate organ involvement, was observed in 15% of the patients. These factors explain the high mortality rate in the included patients. 31
Regarding the immunophenotype, literature describes that patients are positive for multiple T-cell antigens, such as CD5, CD2, CD3, TCR-α/β associated with CD38, CD25, and HLADr, with CD3+/CD4+ being the most common phenotype, while CD4+/CD8+ or CD3+/CD8+ are rare. 32 In our cohort, most patients were positive for CD5 (91%), CD3 (91%), CD4 (100%), and CD25 (78.7%), with CD8+ being positive in only 6% of cases, which are consistent with the literature.
The presence of CD25 positivity is important as it plays a fundamental role in the transition from HTLV-1 infection to ATL. 33 In some cases, ATL expresses CD30, opening up the possibility of using anti-CD30 drugs like Brentuximab as a rescue treatment. However, this medication was not available in the country during the evaluation of these patients, 34 and thus, it was not used in first-line or salvage treatment in the positive population.
The Latin American cohort from the GELL group reported that the first-line treatment was high-intensity multiagent chemotherapy in 159 subjects (63% of the sample), with only 2 patients able to undergo allogeneic hematopoietic stem cell transplantation (one in the first line and the other in the second line). The most used protocol was CHOP or CHOP-like in 117 patients, followed by CHOEP in 40 patients, with overall response rates of 29%, 12%, and 17% for complete, partial, and overall response, respectively. 35 In our study, most patients received first-line management with high-intensity multiagent chemotherapy (91%). Sixty percent received anthracycline-based protocols (CHOP, EPOCH, DAEPOCH), and five patients received the modified LSG15 protocol as first-line treatment. In our study, 91% of the patients received disease-specific management with different chemotherapy protocols. Sixty percent received CHOP-like protocols, with CHOP being the most common, followed by EPOCH and DAEPOCH (see Table 4).
The LSG15 protocol (VCAP-AMP-VECP) is the gold standard in Japan, the country with the most publications on the disease to date.26,36–38 This therapy has shown treatment response in 81% of patients who received the regimen, with a complete response rate of 35.5%, a median overall survival of 12.7 months, and a 3-year overall survival rate of 24% for those who received VCAP-AMP-VECP. However, Colombia does not have Ranimustine or Vindesine, so the modified LSG15 protocol does not include them and instead replaces Ranimustine with Carmustine, which is also a nitrosourea. Additionally, Vindesine is replaced with vincristine, as both are vinca alkaloids. In this study, five patients received the modified LSG15 protocol as first-line treatment, and two patients received it as second-line treatment. However, only two patients achieved a complete response, both in the second-line setting. None of the patients who received the modified LSG15 protocol as first-line treatment achieved a complete response. It is not clear if the change from Ranimustine to Carmustine and from Vindesine to vincristine could explain the variation in these results.
In a retrospective study, most patients were Afro-Caribbean (77%), originating from Haiti, Jamaica, and other Caribbean islands. The median survival time and 4-year overall survival were 4.1 months and 10% for the acute presentation, and 10.2 months and 4.2% for the lymphomatous variant, respectively. 26 In other series including patients from Argentina, Bolivia, Chile, Colombia, Ecuador, Mexico, Paraguay, and Peru, the 1-year overall survival was 24% for the acute variety and 44% for the lymphomatous variety. 36 In our series, the prognosis was also unfavorable, with a median survival time of 5 months. Only one patient was able to undergo consolidation with allogeneic bone marrow transplantation from an HTLV seronegative donor and is still alive to date.
Limitations
This study has several limitations. First, the retrospective nature of the analysis limits the ability to establish causal relationships, as the data were collected from existing records and were not specifically designed to address the current research questions. Additionally, being a single-center study, the findings may not be generalizable to other populations or clinical settings, as the practices and characteristics of the population may differ.
Conclusion
In this retrospective study of patients diagnosed with ATL at a high-level institution in Cali, Colombia, a poor prognosis and short survival were observed in these aggressive variants of the disease.
Most patients had acute presentations of the disease and were identified at advanced stages at the time of diagnosis. Treatment mainly consisted of chemotherapy, with CHOP-like protocols being the most used. However, the rate of complete response was low, and most patients experienced complications and treatment-related toxicities, often severe.
The prognosis for patients with this disease is bleak, with short survival and a low response rate to intensive chemotherapy. Despite the different treatment protocols used, most patients did not achieve a complete response, limiting treatment options and the possibility of hematopoietic stem cell transplantation.
These findings underscore the imperative to enhance therapeutic approaches for ATL, particularly for aggressive variants of the disease. The development of more efficacious and individualized treatment strategies is crucial to improve survival rates and mitigate treatment-associated complications in affected patients. While factors associated with mortality have been established in various populations, there remains a pressing need for more comprehensive data from larger cohorts across Latin America. Such data could provide a more accurate and nuanced understanding of the disease in this region.
Given that this study represents the largest sample of ATL patients published in Colombia, it emphasizes the importance of further research and awareness about this disease in the country. Additionally, the need to implement early detection programs and prevention of HTLV-1 infection, especially in areas with high prevalence such as the southwestern region of Colombia and the Pacific coast, is highlighted.
In conclusion, this study provides important clinical and epidemiological insights into ATL in Colombia, emphasizing the need for more effective therapies to improve the quality of life and survival of patients affected by this disease.

