Abstract
Background:
Hereditary haemorrhagic telangiectasia (HHT) is a genetic vascular disorder characterised by telangiectases, which cause nasal and gastrointestinal (GI) bleeding, and visceral arteriovenous malformations. Since 2012 bevacizumab, a monoclonal antibody targeting vascular endothelial growth factor, has been a promising treatment for HHT-related bleeding and was evaluated in the phase II BABH study.
Objective:
To follow and describe evolution and treatments of patients with HHT post-BABH study.
Design:
This study is a 1-year, multi-centre descriptive study.
Methods:
We collected clinical (nose and GI bleeding, red blood cell transfusions) and biological (haemoglobin and ferritin levels) data and treatment information.
Results:
Of 22 patients included across 4 centers, 15 received bevacizumab. Among them, 12 (86%) had a >50% decrease in the number of RBC units transfused 3 months post-treatment. Mean haemoglobin levels increased from 83.08 to 105.98 g/L.
Conclusion:
Bevacizumab effectively reduces RBC transfusions and is efficient for treating severe bleeding in patients with HHT.
Trial registration:
This trial was registered with the ClinicalTrials.gov Identifier #NCT06039124.
Keywords
Introduction
Hereditary haemorrhagic telangiectasia (HHT; OMIM#187300) is a rare genetic vascular disorder characterised by recurrent epistaxis, cutaneous and mucosal telangiectasias, and visceral arteriovenous malformations that affect many organs, including the lungs, gastrointestinal (GI) tract, liver and brain. 1 In patients with HHT and severe bleeding related to epistaxis or GI bleeding, we conducted a phase II clinical trial (BABH study) 2 to evaluate the efficacy of intravenous bevacizumab in decreasing the number of red blood cell (RBC) units transfused. Because bevacizumab is already widely prescribed in patients with HHT and severe bleeding, we designed the BABH study with a short follow-up period of 3 months after the end of the treatment to be able to unblind patients at the end of the follow-up phase of the study and treat those in the placebo group with bevacizumab if treatment was indicated.
Even though the main outcome in the BABH study was not achieved because the trial was underpowered, patients with HHT receiving bevacizumab experienced a clinically relevant decrease in the number of RBC units transfused (44% decrease in the treatment group vs 4% in the placebo group), particularly those with severe GI bleeding and/or high bevacizumab concentration. Furthermore, as highlighted by Al-Samkari, 3 all the evidence from prospective4–6 and retrospective7–11 studies is in favour of using bevacizumab in HHT. In this context, our objectives in this descriptive study were to describe the evolution in the number of RBC units transfused in patients treated or retreated with bevacizumab after the end of the BABH study, and to highlight the limitations of randomised trials for rare diseases.
Materials and methods
Patients and data collection
The local research ethics committee approved this study, and patients were informed in accordance with national rules. The 24 patients included in the BABH study, randomised and treated with either placebo or bevacizumab (ratio 1:1) from 27 September 2017 to 19 November 2019, were informed about this 1-year follow-up study and were able to oppose the use of their data. In the BABH study, they received six infusions of treatment 14 days apart and then stopped the treatment and had a 3-month follow-up before their inclusion in the BEST study. Treatment with bevacizumab was possible as soon as patients had completed the study.
The group of patients who were treated with bevacizumab in the BEST study and who initially received placebo during the BABH study were named the placebo-beva group and those who received bevacizumab during both studies were named the beva-beva group.
Effectiveness measures
The number of RBC units transfused in the 3 months on treatment were compared with the number of units transfused in the 3 months pretreatment and the 3 months posttreatment.
Baseline haemoglobin (Hb) was calculated as the average of all haemoglobin values measured in the 3 months prior to bevacizumab initiation and compared with mean on-treatment haemoglobin and 3 months after treatment. For patients treated right after the end of the BABH study, some data might overlap between the after period of BABH and the before period of BEST.
Treatment
Treatment modalities were decided by each centre, but the national consensus is for bevacizumab at a dose of 5 mg/kg every 14 days, with a total of six injections (induction regimen) and then 5 mg/kg every month or 2 months as maintenance therapy in most patients. Both the haemoglobin levels and the number of RBC units transfused were collected and gathered over a 3-month period before, during and after the induction treatment in both groups.
Tolerance and toxicity
Monitoring was carried out in accordance with national pharmacovigilance rules for off-label drugs.
Statistics
The patient characteristics were summarised by means of descriptive statistics (number, mean, standard deviation, median, minimum and maximum for the quantitative variables, and numbers and percentages for the qualitative variables).
Qualitative variables were compared using a Chi-squared test and quantitative variables were compared using Kruskal–Wallis test.
All time-related computations were performed with days as the time unit (e.g. 3 months = 84 days), for homogeneity. Statistical analyses were performed using SAS 9.3 Institute Inc., Cary, NC, USA. Statistical significance was defined as p < 0.05.
Results
Of the 23 patients who completed the BABH study, 22 were included, the 23rd having been lost to follow-up (Figure 1). Six of the 11 patients initially in the bevacizumab group were retreated because of a good response during the BABH study, 2 had a prolonged effect and were still improved (but not yet retreated), and 2 patients did not improve during the BABH study and were not retreated.

Flow chart.
Nine of the 12 patients who received the placebo in the BABH study were treated with bevacizumab alone (no local treatment) in the 12-month follow-up period. The three other patients were not treated with bevacizumab: two had improved after local laser treatment and were not eligible for a bevacizumab treatment during the time of the study and one was treated with bevacizumab, but the treatment was initiated outside of the 12-month follow-up period. During the study period, 15 of the 22 patients included received bevacizumab treatment (induction regimen) and of these, all but 12 subsequently received at least one injection of maintenance treatment (initially every 2 months, with frequency adapted to clinical improvement) during the study period. Of them, at the beginning of treatment; all had epistaxis and 8 had GI bleeding, 6/9 in the placebo-beva group and 2/6 in the beva-beva group. The median time between the end of the BABH study (3-month treatment and 3-month follow-up) and the bevacizumab treatment, was 22 days (9–266) in the placebo-beva group (n = 9) and 133.5 days (45–171) in the beva-beva group (n = 6).
The baseline characteristics of the 15 patients treated with bevacizumab after the end of the BABH study are summarised in Table 1.
Patients’ baseline characteristics before treatment.

Efficacy
Haemoglobin (Figure 2): Mean haemoglobin level increased after bevacizumab treatment from 83.08 to 105.98 g/L in the entire population and from 79.6 to 107.2 g/L and 88.3 to 104.4 g/L in the placebo-beva and beva-beva groups, respectively.
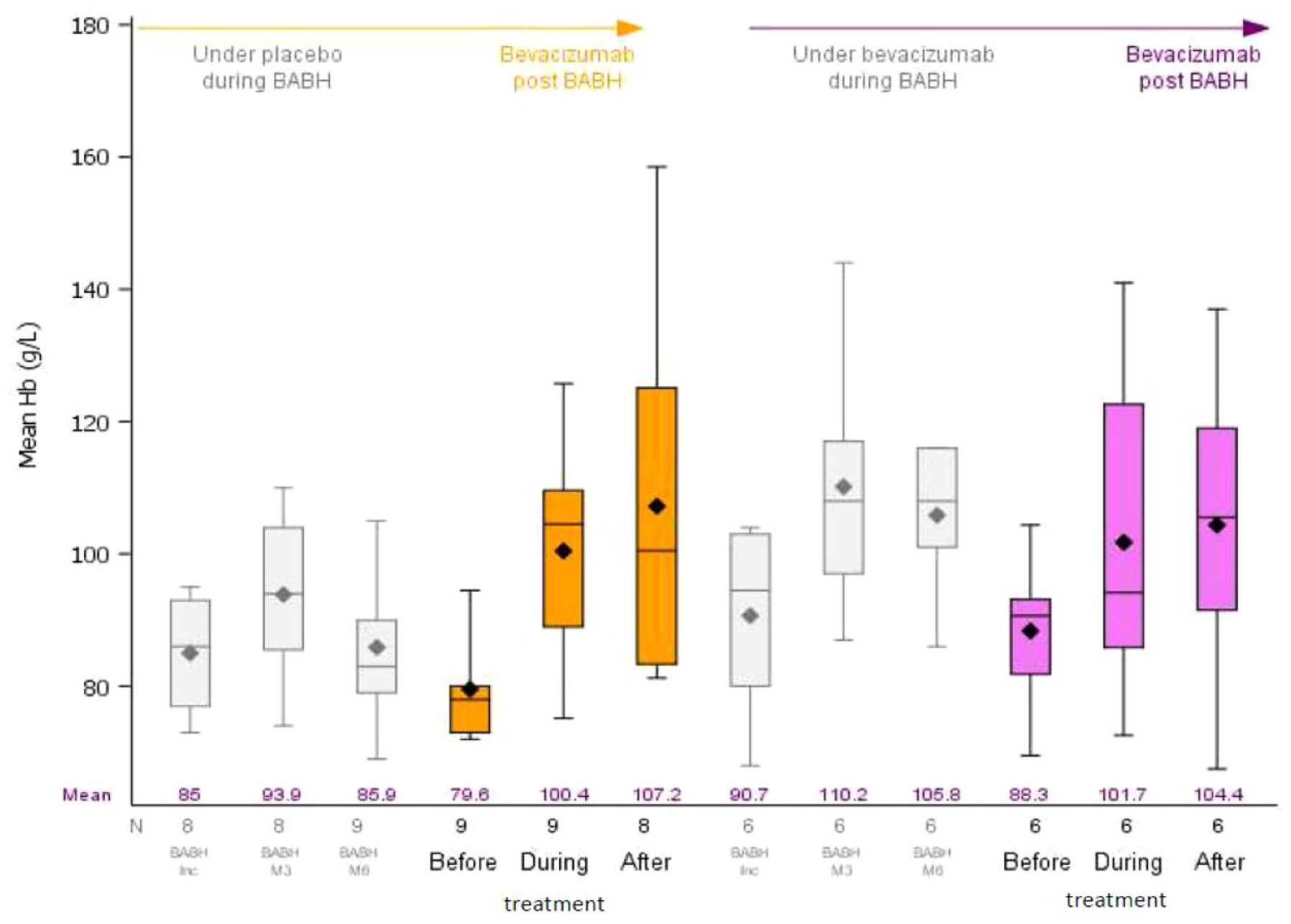
Haemoglobin levels in the 3-month periods before treatment, during treatment and after treatment. As a reminder, we provided the data for the BABH study (in grey) for these patients. There might be some overlap between the BABH-after period and the BEST-before period for patients being retreated right after the end of the study. For haemoglobin, during the BABH study, there was only one measure per timepoint.
RBC transfusions (Figure 3): Of the 15 patients treated with bevacizumab, the mean number of RBC units transfused decreased after bevacizumab treatment in both groups, from 9.22–1.13 to 8.00–3.83 in the placebo-beva and beva-beva groups, respectively. Overall, 12 patients (86%) had a decrease of more than 50% in the number of RBC units transfused 3 months after the beginning of the treatment. The mean and median differences in the total number of RBC units transfused after versus before treatment were −5.79 (SD = 4.34) and −6.5 (−11.6) (p = 0.0018).

Number of RBC units transfused in the 3-month periods before treatment, during treatment and after treatment. As a reminder, we provided the data for the BABH study (in grey) for these patients. There might be some overlap between the BABH-after period and the BEST-before period for patients being retreated right after the end of the study.
Safety
No serious adverse events occurred, and no treatment was discontinued prematurely during treatment the study period.
Discussion
Our results underline the efficacy of bevacizumab treatment and the rapidity of response in patients with HHT, particularly in those with GI bleeding. Due to randomisation imbalance, 67% of the patients with GI bleeding during the BABH study were in the placebo group. 2 The bevacizumab treatment was administered shortly after the end of the follow-up period of the BABH study in most patients, and the response of these patients to bevacizumab treatment in the BEST study demonstrated a marked and quick reduction in the number of RBC units transfused, as well as a clinically relevant increase of almost 30 g/L in haemoglobin levels after treatment.
In this study, we observed that patients treated effectively in the BABH study continued to improve after the second “induction” treatment of six injections, 14 days apart and maintenance therapy. Because bevacizumab is already widely prescribed in patients with HHT and severe bleeding,7,9 we designed the BABH study with a short follow-up period (3 months after the end of the treatment) to be able to unblind patients at the end of the follow-up phase of the study and treat those in the placebo group with bevacizumab if treatment was indicated. However, the short follow-up time made it impossible to observe how long the treatment remained effective after it had been discontinued. Interestingly, two patients were not retreated within the year, as they were still very much improved several months after the first treatment series. Safety during this 1-year follow-up was good, as already reported.
Randomised trials, while considered as the gold standard for evaluating the efficacy of treatments, can raise particular challenges when studying rare diseases. 12 First, small sample size can reduce the statistical power of the study, as observed in the BABH study, making it challenging to detect significant differences. Second, groups were heterogeneous, with patients experiencing nose and/or GI bleeding, and this was not captured in the randomised study. Third, the choice of the primary endpoint for measuring epistaxis is difficult because the endpoint must be objective and reliable and any criterion whose measurement can be influenced by the study, such as the patient’s measurement of duration of epistaxis or an epistaxis severity score, is partly subjective. ‘Hard’ criteria such as haemoglobin level or the number of RBC transfusions should be preferred as judgment criteria. Fourth, as there are limited treatment options available, it is ethically questionable to design a randomised trial and assign patients to a control group without immediate access to a potentially life-saving treatment. For all these reasons, it is essential that innovative trial designs be developed that use real-world evidence.
The major limitations of the BEST study are that it was a non-randomised study, excluding non-responders included in the BABH study, thus adding a recruitment bias. Furthermore, intravenous iron infusions were not taken into-account, introducing significant confounding issues as iron infusions play a significant role in reducing the need for RBC transfusions. Finally, one patient was lost to follow-up during the study, which may have negatively impacted our study, as the patient is still alive and was likely a very good responder to the treatment, no longer requiring RBC transfusions after the treatment.
Conclusion
This study, combined with all previous studies, clearly supports that bevacizumab reduces the number of RBC units transfused, and is an efficient treatment for patients with HHT and severe bleeding, even though it does not eliminate the need for IV iron supplementation. Furthermore, it highlights the difficulty of achieving well-designed trials due to the complexity of including sufficient numbers of subjects with rare diseases and with an objective primary outcome due to practical and ethical reasons. It also confirms the value of collecting real-life data via high-quality, international registries.
Supplemental Material
sj-doc-1-tah-10.1177_20406207241300828 – Supplemental material for BEST study: one-year descriptive follow-up of bevacizumab treatment in hereditary haemorrhagic telangiectasia post-BABH interventional study
Supplemental material, sj-doc-1-tah-10.1177_20406207241300828 for BEST study: one-year descriptive follow-up of bevacizumab treatment in hereditary haemorrhagic telangiectasia post-BABH interventional study by Sophie Dupuis-Girod, Evelyne Decullier, Sophie Rivière, Christian Lavigne, Vincent Grobost, Vanessa Leguy-Seguin, Hélène Maillard, Thierry Chinet, Anne-Emmanuelle Fargeton, Alexandre Guilhem and Ruben Hermann in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
