Abstract
Objective
To assess the spectrum of patients who developed recurrent acute transfusion reactions (TRs) and to characterize these recurrent TRs.
Methods
This retrospective study included patients who developed ≥2 acute TRs between April 2017 and March 2020 in a tertiary medical centre.
Results
Among 216 TRs that occurred after 2024 transfusions in 87 patients, 66 (75.9%) patients reported a history of transfusions before the first TR and 70 (80.5%) patients received further transfusions after the last TR; with the same type of TR and reaction to the same type of blood product observed in 59 (67.8%) patients and 56 (64.4%) patients, respectively. TRs were most commonly associated with packed red blood cell (PRBC) transfusions and a febrile non-haemolytic transfusion reaction (FNHTR) was the most common type of TR. However, leukocyte reduced (LR) PRBCs were less common than LR platelets among transfusions with TR (22.7% [27/119] versus 75.0% [57/76], respectively) and premedication was prescribed before 196 of 216 (90.7%) transfusions with TR.
Conclusion
Most patients with recurrent TRs received repeated transfusions in addition to transfusions with TR. Instead of considering premedication, an increase in the use of LR might be the strategy to reduce the recurrence of TR.
Introduction
Blood transfusion is an effective strategy to replenish blood or blood components that may be insufficient or lost from the body secondary to numerous causes. Although it can be lifesaving, a transfusion may be associated with certain risks, which could be direct hazards (the mechanisms for post-transfusion injury are clearly traceable to the blood transfusion) or indirect hazards (those associated with transfusion based on epidemiological evidence). 1 A transfusion reaction (TR) is a direct hazard that is an undesirable transfusion-induced response or effect; with acute TR being defined as a reaction that occurs within 24 h of the administration of blood or blood components. 2 The incidence of acute TRs is <2.0%.3,4 Acute TRs include acute haemolytic transfusion reaction (AHTR), febrile non-haemolytic transfusion reaction (FNHTR), allergic reaction, anaphylaxis, transfusion-related acute lung injury (TRALI), transfusion-associated circulatory overload (TACO), transfusion-associated dyspnoea and transfusion-transmitted microbial contamination.5,6
At Kaohsiung Veterans General Hospital, a subgroup of patients who received repeat transfusions tended to develop more than one episode of TR. These patients were usually seriously ill or undergoing several therapeutic interventions including chemotherapy and surgery, among others. Therefore, TRs may not only lead to confusion among healthcare providers and prevent the initiation of appropriate treatment, but they also make these fragile patients much more uncomfortable. Additionally, TRs cause wastage of transfusable blood and necessitate extensive laboratory workups. Therefore, the careful evaluation of patients with recurrent TRs is important. This current study investigated this specific group of patients and the TRs observed in this patient population.
Patients and methods
Collection of patients and transfusion reaction record
Per the hospital policy at Kaohsiung Veterans General Hospital, Kaohsiung, a tertiary medical centre in Southern Taiwan, a transfusion note is necessary for hospitalized patients undergoing blood transfusion. The transfusion note records the patients’ vital signs at the commencement of transfusion, 15 min after transfusion initiation and at the end of transfusion; and the type and volume of blood product used and premedication administered before transfusion. Patients who developed abnormal vital signs or dyspnoea, chills or fever during transfusion were diagnosed with a positive TR by the assisting nurse and were treated for the TR accordingly. Then a positive TR was recorded in medical chart, but the TR type was not classified by the hospital system.
This retrospective study analysed the transfusion notes obtained from the hospital information bank of Kaohsiung Veterans General Hospital, Kaohsiung between April 2017 and March 2020. All patients with ≥2 acute TRs during this period were included in the study. Details of their transfusion notes were recorded and based on chart review, demographic data including age, sex, underlying disease, type of filter used for transfusion, personal transfusion history and survival were obtained. All of this information was de-identified. Following a thorough chart review of patient details, TRs were categorized as AHTR, FNHTR, allergic reaction, anaphylaxis, TRALI, TACO, transfusion-associated dyspnoea and transfusion-transmitted microbial contamination.5,6 TRs were classified as ‘others’ if specific categorization was difficult.
The study was approved by the Institutional Review Board of Kaohsiung Veterans General Hospital (no. 21-CT5-32[210513-1]) and it also granted a waiver of documentation for signed informed consent. This study conforms to the STROBE guidelines. 7
Transfused blood products
All transfused blood products including packed red blood cells (PRBC), platelets, cryoprecipitate, and plasma (frozen or fresh frozen) were obtained from the local blood centre affiliated with the Taiwan Blood Services Foundation. Platelet products were obtained following apheresis from a single donor or from concentrates separated from whole blood. A total of 16 cryoprecipitate and 15 frozen plasma transfusions were undertaken, both of which were included as plasma transfusions.
Filtration of blood products and premedication before transfusion
Both PRBC and platelets could be pre-storage leukocyte reduced (LR) blood cell products or not. A transfusion filter is required for all blood product transfusions. 8 The transfusion set was usually a Y type set with a 170–260 micron filter, without the function of leukocyte removal. An alternative LR filter can remove 99% leukocytes in the blood. 9 The decision to perform LR transfusion was at the clinician’s discretion. The goal of LR transfusion could be achieved by pre-storage LR blood cell products or adoption of an LR filter. Indications for blood transfusion, irradiation of blood products, premedication administered before transfusion, management and the decision to continue transfusion after diagnosis of TR were all determined clinically.
Methodology
To control for the effect of repeated transfusions in increasing the risk of TR, the median number of transfusions per TR (defined by the transfusion number per TR for all patients arranged in order from smallest to largest) was used to classify patients into two groups as follows: patients with fewer transfusions per TR than the median number of transfusions per reaction were categorized into the high TR occurrence group; and those in whom transfusions per TR were greater than the median number of transfusions per reaction were categorized into the low TR occurrence group.
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 17.0 (IBM Corp., Armonk, NY, USA). Comparisons of mean age and median interval between the two TR occurrence groups were undertaken using the Mann–Whitney
Results
This retrospective study included 87 patients (51 females and 36 males). Table 1 summarizes their baseline clinical and demographic data. The majority of patients (54 of 87; 62.1%) were aged 18–65 years. Of the 87 patients, 51 (58.6%) had active cancer and the reason for transfusion was related to cancer. Among the 51 patients with active cancer, 11 patients had haematological malignancies and they received more transfusions than patients with other cancer types during the study period (mean number of transfusions, 53.5 versus 18.7, respectively).
Baseline demographic and clinical characteristics of patients (

Data presented as mean ± SD (range), median (range) or
40 of 49 females ≥18 years old at transfusion had a previous history of pregnancy.
All with comorbidities, including hypertension, liver cirrhosis, diabetes mellitus, chronic renal insufficiency.
Including one incomplete abortion, one idiopathic thrombocytopenic purpura, one post-kidney transplant haematuria, one systemic lupus erythematosus with pulmonary haemorrhage, one anaemia and end stage renal disease, two plasmapheresis due to myasthenia gravis and six miscellaneous/unclassified.
A review of patients’ transfusion history revealed that 66 (75.9%) patients had received transfusions at Kaohsiung Veterans General Hospital before the transfusion associated with the first reported TR during the study period; 57 (65.5%) patients had received at least one transfusion between two transfusions with TRs; and 70 (80.5%) patients had a transfusion history after the last TR at Kaohsiung Veterans General Hospital (Table 1). The type of recurrent TR remained the same across all TR episodes in 59 (67.8%) patients: 35 FNHTRs, 23 allergic reactions and one TACO. Fifty-six (64.4%) patients had TRs always associated with the same type of blood product in each episode (41 PRBC, 11 platelet and four plasma transfusions); and 26 of them were only transfused with the type of product that could have led to a TR. Notably, 34 (39.1%) patients were deceased at the last follow-up at Kaohsiung Veterans General Hospital. The survival rates for patients with active cancer and those without active cancer were 47.1% (24 of 51 patients) and 80.6% (29 of 36 patients), respectively. But the cause of death was not attributable to the TR in any of the deceased patients.
During the study period, 87 patients received 2024 transfusions, including 961 PRBC, 731 platelet and 332 plasma transfusions (Figure 1a). A total of 216 TRs following 2024 transfusions were observed (10.7%). Figures 1b and 1c show the types of TRs, their associated blood products and their occurrence rates. The most common TR was FNHTR (50.5%), followed by allergic reactions (38.0%), others (7.4%), TACO (2.8%) and anaphylaxis (1.4%). Analysis of TR types showed that allergic reactions were the most common TR associated with plasma and platelet transfusions (81.0% and 60.5%, respectively), whereas FNHTR (71.4%) was the most common TR associated with PRBC transfusions (Figure 1d). Following the diagnosis of TR, 149 (69.0%) transfusions were completed without blood product wastage (Figure 1e).
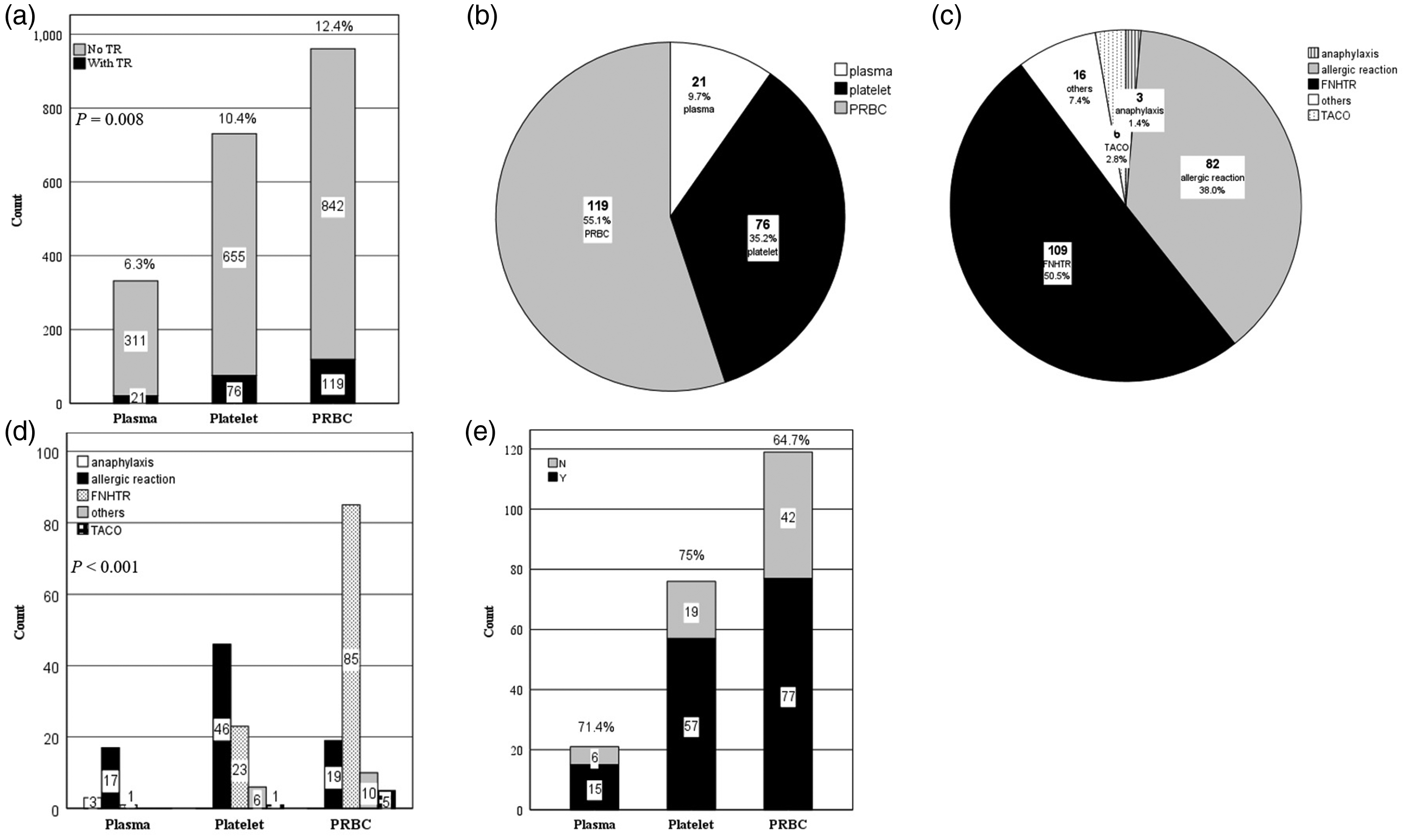
Distribution of blood products associated with transfusion reactions (TRs) and types of TRs in patients (
In this current study, 46 patients with 110 TRs were categorized into the high TR occurrence and 41 patients with 106 TRs into the low TR occurrence groups, according to the median of 5 transfusions per TR. Table 2 summarizes the clinical and demographic data of the patients stratified according to the TR occurrence. There were no significant differences in age, sex distribution or active cancer between the two groups. Patients in the high TR occurrence group had a higher survival rate than the low TR occurrence group (71.7% [33 of 46 patients] versus 48.8% [20 of 41 patients],
Comparison of patients (

Data presented as mean ± SD (range), median or
Between-group comparisons undertaken using the Mann–Whitney
PRBC, packed red blood cells; NA, not applicable.
Figure 2 shows the classification of the TRs and related blood products in the two groups. The TR occurrence of PRBC, platelet and plasma transfusions in the high TR group was 29.4% (65 of 221), 35.6% (32 of 90) and 30.2% (13 of 43) respectively, whereas 7.3% (54 of 740), 6.9% (44 of 641) and 2.8% (eight of 289) for PRBC, platelet and plasma transfusions, respectively, were noted in the low TR group. Plasma transfusions caused fewer TRs than PRBC and platelet transfusions in the low TR occurrence group (
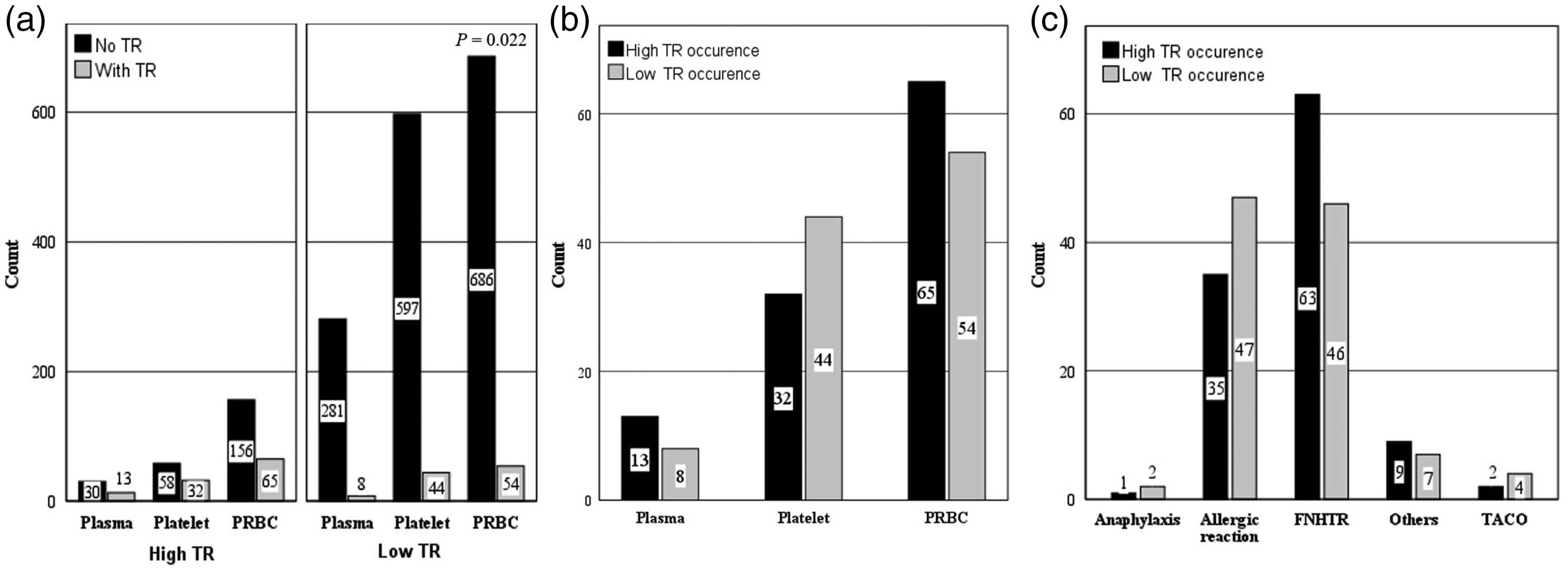
Comparison of patients (

Summary of the type of filters used and the use of leukocyte-reducing (LR) cell products during 216 transfusion reaction (TR) episodes in patients (
Premedication to prevent TRs was prescribed in 196 of 216 (90.7%) transfusions associated with TRs; antihistamines (192 of 216 [88.9%] transfusions, either alone or combined with other drugs) were the most commonly prescribed agents (Table 3).
Summary of the premedication used and management during 216 transfusion reaction (TR) episodes in patients (

Data presented as
Antipyretics used were all acetaminophen except one intravenous ketorolac.
CTM, chlorpheniramine.
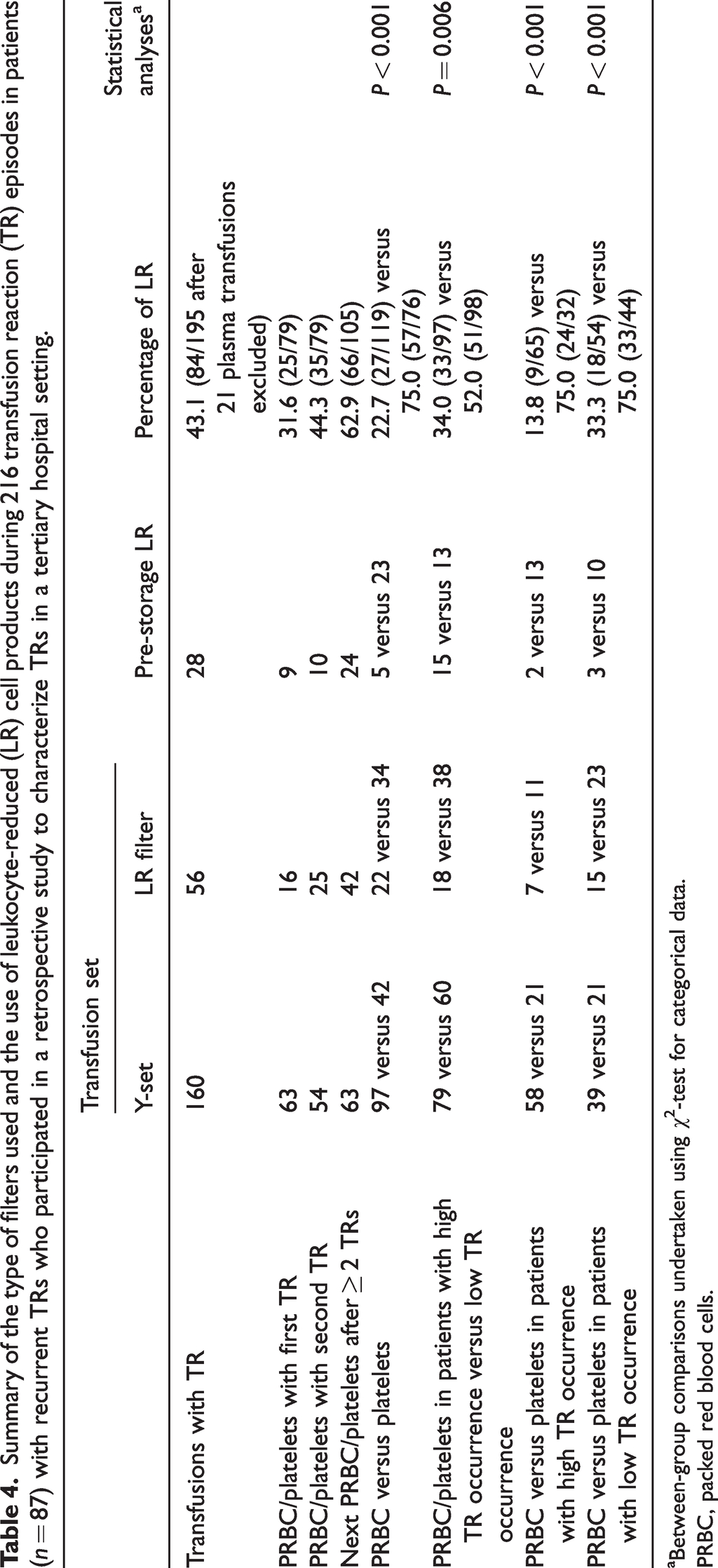
Table 4 and Figure 3 show the transfusion sets and LR cell products used for the 216 transfusions associated with TR. The offered rate of LR cell products (either platelets or PRBC) by either LR filter or pre-storage LR was 43.1% (27 of 119 PRBC and 57 of 76 platelet transfusions, respectively), which was gradually increased after recurrent TRs: 31.6% (25/79), 44.3% (35/79) and 62.9% (66/105) at the first TR, second TR and after ≥2 TRs respectively. Subgroup analysis showed that LR cell products were used more frequently in patients who received platelet transfusions (75.0%; 57/76) than in those who received PRBC (22.7%; 27/119) (
Summary of the type of filters used and the use of leukocyte-reduced (LR) cell products during 216 transfusion reaction (TR) episodes in patients (
Between-group comparisons undertaken using χ2-test for categorical data.
PRBC, packed red blood cells.
Discussion
This current retrospective study investigated the characteristics of transfusions associated with TRs in patients who developed recurrent TRs. Because 149 (69.0%) of 216 transfusions were completed after the occurrence of TR and 70 (80.5%) of 87 patients received further transfusions after the last transfusion with TR, most TRs did not seem to be a serious clinical obstacle. Analysing the risk factors of recurrent TRs was limited in this current study because of the lack of comparative data from patients who did not experience recurrent TRs. As a control group was not available, intra-group data were compared instead and a high TR occurrence group was identified. The current study demonstrated a lower likelihood of a positive transfusion history before the first TR (56.5% versus 97.6%,
The occurrence of TR was reported to be 1.8% in an intensive care unit 3 and 0.62% and 0.24% in paediatric and adult populations, respectively. 4 It is not surprising that the TR occurrence in this current study was 10.7% (216 of 2024) because the study population consisted of patients experiencing recurrent TRs. Some authors have reported that allergic reactions represent the most common TR,4,11 in contrast to FNHTR reported by a few others.12–14 The TR occurrence contributed by different types of blood products is also variable.4,11–15 These variations are attributable to differences in the premedication used, patient characteristics, product manufacturing processes, storage times, use of LR and the reporting and monitoring systems used in different studies. In this current study, the occurrence of FNHTR associated with PRBC, platelet and plasma transfusions was 8.7%, 3.1%, and 0.3% (85 of 981 PRBC, 23 of 731 platelet and one of 332 plasma transfusions), respectively; and that of allergic reactions associated with PRBC, platelet and plasma transfusions was 1.9%, 6.3% and 5.1% (19 of 981 PRBC, 46 of 731 platelet and 17 of 332 plasma transfusions), respectively. These findings were similar to those previously reported, 13 although the TR rates were lower in the previous study (FNHTR associated with PRBC, platelet and plasma transfusions were 0.9%, 0.3% and 0.2%, respectively; and allergic reactions associated with PRBC, platelet and plasma transfusions were 0.3%, 0.9% and 0.9%, respectively). Additionally, 59 of 87 (67.8%) of the current patients experienced the same type of TR and the recurrent TR related to the same type of blood product in 56 of 87 (64.4%) patients. In this current study, allergic reactions were the most common TR associated with plasma and platelet transfusions (81.0% and 60.5%, respectively), whereas FNHTR (71.4%) was the most common TR associated with PRBC transfusion. A previous study reported that FNHTR accounted for 76.2% of TRs with PRBC transfusion and allergic reactions accounted for 73.68% and 91.9% TRs with plasma and platelet transfusions, respectively. 11 The relatively high percentage of the same TR type and the same blood product type to cause TR for the individual patient, and the strong correlation between specific TR types and blood products, suggest that the high predictability of TR types and products associated with TR may aid in the prevention, diagnosis and management of TR.
Routine premedication is controversial and not the standard of care for TR prevention. A previous study reported a FNHTR rate of 0.09% in 80.0% of patients who received transfusions after routine antipyretic premedication; however, premedication did not prevent other more severe TRs. 16 A Cochrane review of three randomized controlled trials reported no evidence to support the administration of premedication to minimize the risk of non-haemolytic transfusion reactions. 17 The British Committee for Standards in Haematology Blood Transfusion Task Force recommends antipyretic premedication for patients with recurrent FNHTR, no antihistamines or steroid premedication for mild recurrent allergic reactions, and consideration of antihistamine prophylaxis for recurrent moderate or severe allergic reactions. 2 In this current study, 196 of 216 (90.7%) transfusions with TRs were premedicated. But whether premedication reduced the severity of these TRs was difficult to evaluate. Randomized trials are necessary to investigate the role of premedication in patients with recurrent TRs. However, considering the low risk and relatively low cost of premedication in Kaohsiung Veterans General Hospital, it is deemed acceptable to administer prophylactic medication despite its unproven efficacy.
The benefits of LR include a reduction in the incidence of FNHTR and immunization against human leukocyte antigens and human platelet antigens, which may cause refractoriness to platelet transfusion and prevention of cytomegalovirus transmission.
18
A previous reported that universal pre-storage LR reduced the FNHTR rate by 47.1% and 93.1% for RBC and platelet transfusions, respectively.
19
Another study reported that the FNHTR incidence associated with RBC transfusion decreased from 0.37% to 0.19% (
In conclusion, most TRs were mild and patients safely received transfusions even after recurrent TRs in the current study population. Some patients show an innate tendency to develop TRs and further studies are warranted to definitively establish the characteristics of these high-risk patients. These current results highlight that a large percentage of TRs occurs in association with the first transfusion and a shorter interval between the two TRs in patients with a high TR occurrence. Furthermore, the prevalence of the same TR type was associated with the same blood product. In addition to the administration of premedication, the use of LR blood products may effectively prevent FNHTR, particularly PRBC transfusions in patients with high TR occurrence. Greater awareness and education are required with regard to the benefits of LR blood cell products for the management of patients who develop recurrent TRs.
Footnotes
Author contributions
Jin-Shuen Chen and Ming-Yun Hsieh conceived of the presented idea. Chun-Hao Yin contributed to data collection. Ming-Yun Hsieh designed and performed the analysis, interpretated the results and prepared the manuscript.
Acknowledgements
The authors thank personnel at the Cancer Centre, Department of Medical Education and the Research Centre of Medical Informatics of Kaohsiung Veterans General Hospital for providing information in response to inquiries and assistance in data processing.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from funding agency in the public, commercial, or not-for-profit sectors.
