Abstract
Background:
The development of tyrosine kinase inhibitor (TKI) therapy has positively impacted the survival rates of patients with chronic myeloid leukemia (CML). It is common in medical practice to adjust the dosage of TKI downward because of TKI-associated adverse events, financial burden, comorbidity, or an attempt at treatment-free remission.
Objectives:
This investigation sought to explore the feasibility of employing a reduced dosage of TKI for treating CML.
Design:
This was a retrospective study.
Methods:
Patients with CML in its chronic phase who had been on a reduced dose of TKI for a minimum of 3 months for various reasons in a practical clinical environment, irrespective of molecular response, were included. Regular molecular monitoring was performed, and changes in adverse events were recorded after dose reduction.
Results:
This research included a total of 144 participants. Upon reducing the dosage, 136 of 144 patients achieved major molecular response or deeper, and 132 of 144 achieved molecular response 4 (MR4). Following a median observation period of 16 months, the calculated 1- and 2-year survival rates free from MR4 failure were estimated to be 96.5% (95% CI: 90.8–98.7) and 90.5% (95% CI: 81.3–95.3), respectively. MR4 failure-free survival was better in patients with longer MR4 durations (⩾34 months) before dose reduction (
Conclusion:
Lowering the TKI dose can effectively preserve a deep molecular response over time while relieving adverse events caused by TKIs.
Introduction
The introduction of tyrosine kinase inhibitors (TKIs) has markedly enhanced the survival outlook for patients diagnosed with chronic myeloid leukemia (CML). As a direct consequence, patients with chronic CML now enjoy a life expectancy that closely mirrors that of people without CML. 1 The majority of patients need lifelong exposure to TKIs. Nonetheless, prolonged administration of TKIs is associated with sustained adverse events (AEs), which adversely influence patients’ well-being and potentially result in significant disease and fatality.2–5 Furthermore, constant TKI exposure entails significant financial burdens for both patients and society at large. For these reasons, TKI dose reductions in patients with CML are often considered a means of addressing such problems in real-life practice. 6
At present, achieving treatment-free remission (TFR) is recognized as a novel long-term therapeutic aim for patients with CML. Several clinical studies and practical experiences have demonstrated that approximately 40–60% of patients who experienced a deep molecular response (DMR) are capable of attaining TFR, and success in discontinuing TKI therapy is notably influenced by the length of time on TKI treatment and the period of sustained DMR before the cessation of TKI.7–10 Consequently, guidelines for TKI discontinuation have been established by international specailists.11,12 However, it is worth noting that approximately 50% of patients experience molecular relapse after stopping TKI therapy and only a percentage of patients can meet the criteria recommended by guidelines for discontinuing TKI therapy. 13
Most recently, some investigators have attempted to reduce the TKI dose before stopping it altogether within clinical trial settings. Several clinical studies and practical evidence have confirmed that administering TKIs at a lower dose can still sustain a major molecular response (MMR) and that tapering TKI doses could enhance the likelihood of achieving TFR over direct discontinuation.14–18 The De-Escalation and Stopping Treatment with Imatinib, Nilotinib, or sprYcel (DESTINY) study was designed to assess the outcomes of a stepwise TKI dose decrease before stopping it in patients with a stable MMR or MR4 (molecular response 4 logs beneath an internationally accepted level) after receiving standard TKI doses for at least 3 years.14,15 In this study, the TKI dosage was reduced to half for 1 year, followed by complete cessation for the next 2 years. The study revealed a 7% molecular recurrence (loss of MMR) during the reduced-dose period and a TFR rate at 2 years after stopping treatment of 72% (95% CI: 64–80%) in the MR4 group. The authors concluded that a gradual decrease in treatment before cessation could enhance the effectiveness of TFR strategies. A Chinese multicenter, retrospective trial included 108 patients who received a reduced dose of TKI for different reasons.
16
The results showed that for patients who achieved MMR at the onset of dose reduction, the observed molecular recurrence-free survival rates at 1 and 2 years were 96.7% and 95.1%, respectively. A detailed retrospective analysis involving 26 CML patients who underwent a regimen of reduced-dose TKI prior to stopping the treatment revealed a 5-year TFR of 47.5% in the group receiving the standard dose and 58.8% in the group receiving the reduced dose. This indicates that a TKI dose de-escalation regimen before cessation does not compromise the likelihood of attaining TFR in patients with CML.
17
In a practical review by Claudiani
Lowering the TKI dosage presents a practical strategy for lessening TKI-related adverse effects without sacrificing its effectiveness. However, data on the effect of reducing TKI dosage in patients diagnosed with CML are insufficient. Additional research is required to validate the efficacy of reduced-dose TKIs in sustaining DMRs and reducing adverse reactions. Hence, we undertook a retrospective single-center cohort study on CML patients who had been on a low-dose regimen in a real-world setting for different reasons including AEs, DMR achievement, financial stress, and comorbidities, aiming to provide more evidence about the effectiveness of low-dose TKIs.
Materials and methods
Patients
Clinical information from patients with chronic phase CML who underwent a reduction in dosage of imatinib or any second-generation TKIs (including dasatinib, nilotinib, and flumatinib) between March 2010 and August 2023 at West China Hospital of Sichuan University was retrospectively collected, irrespective of their molecular response. Those who terminated their treatment after the dose was reduced were also included. Excluded from this were patients who were diagnosed with either the accelerated or blast phase of CML or who were treated with a low-dose TKI for less than 3 months. The TKI dosage decreased for reasons such as AEs related to the TKI, economic challenges, existing health conditions, or favorable outcomes from the therapy. Follow-up began on the date the TKI dosage was lowered, with the latest update occurring in November 2023.
TKI treatment
In this research, the criteria for low dosages of TKIs were set as doses under 400 mg per day for imatinib, under 100 mg per day for dasatinib, under 600 mg per day for nilotinib, and under 600 mg per day for flumatinib, with these doses being maintained for a minimum of 3 months. The dose reduction schedule was determined by the severity of side effects, the depth of molecular response, the physician’s advice, and the preference of the patient. If patients experienced adverse reactions of grade 3 or above, as determined by the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0 (published in 2017 by the United States National Cancer Institute), following a dose reduction, they were instructed to discontinue the present TKI and transition to another TKI. 19 Patients with molecular recurrence were administered standard-dose TKIs or switched to another TKI, the choice of which was influenced by how well they tolerated their present TKI and the possible risks involved in switching to another TKI. For patients who achieved a sustained DMR during the reduction of TKI, an attempt to discontinue treatment was made.
Molecular response definitions
Every 3 months, molecular assessments were carried out through quantitative polymerase chain reaction in accordance with the European LeukemiaNet recommendations after dose reduction.11,20 The outcomes were expressed in terms of the
Statistical analysis
Quantitative data are presented as median (range), while qualitative data are shown as frequencies (percentages). Patients were censored upon their most recent clinical check-up. The likelihood of surviving without losing MR4 status under low-dose TKI treatment, termed MR4 failure-free survival, was calculated. This calculation was done through the Kaplan–Meier survival estimates and is presented alongside a 95% confidence interval (CI). To explore potential determinants of MR4 failure-free survival, a univariate analysis was performed using the Kaplan–Meier approach, with group comparisons made through the log-rank test. Statistical significance was acknowledged for
Results
Patient characteristics
In all, 144 CML patients in the chronic phase who underwent low-dose TKI treatment were included in this study, with a sex distribution of 66 females and 78 males and a median age at diagnosis of 45 years (range, 14–77). Using the Sokal score, 69 patients (47.9%) were classified as low risk, 40 (27.8%) as intermediate risk, and 9 (6.3%) as high risk, and Sokal score data were missing for 26 patients. The EUTOS Long-Term Survival (ELTS) score was available for 118 patients: 84 were low risk, 26 were intermediate risk, and 8 were high risk. All patients (100%) carried the e13a2 or e14a2 variant of the
Baseline characteristics of the CML patients enrolled (

CML, chronic myeloid leukemia; F, female; M, male; MMR, major molecular response; MR, molecular response; MR4, molecular response 4 logs beneath an internationally accepted level; NA, not available; TKI, tyrosine kinase inhibitor.
Low-dose TKI treatment
The proportions of patients receiving different TKIs were as follows: imatinib was being taken by 50 patients (34.7%), dasatinib by 68 patients (47.2%), nilotinib by 20 patients (13.9%), and flumatinib by 6 patients (4.2%), as shown in Table 1. All patients treated with imatinib have an initial dosage of 400 mg daily. For those on dasatinib, the starting dosages were distributed as follows: 50 mg/day for 5 patients, 100 mg/day for 61 patients, and 150 mg/day for 2 patients. With nilotinib treatment, 12 patients began with a dose of 600 mg/day, while 8 started at 800 mg/day. Flumatinib’s initial dosages were 600 mg/day for five patients and 400 mg/day for one patient. A median imatinib dosage of 300 mg/day was observed in 35 patients, 250 mg/day in 6 patients, and 200 mg/day in 9 patients. Seventeen patients had a median dasatinib dosage of 75 mg/day, 50 had a median dosage of 50 mg/day, and 1 had a median dosage of 25 mg/day. The median dosage of nilotinib for two patients was 500 mg/day, for nine patients it was 400 mg/day, and for another nine patients, the median dosage was 300 mg/day. For flumatinib, a median dosage of 400 mg/day was administered to three patients, 300 mg/day to one patient, and 200 mg/day to two patients. Overall, 72 (50%) patients experienced a 50% or greater reduction in their TKI dose. At dose reduction, 62 (43.1%) patients were receiving first-line TKI treatment, 81 (56.3%) patients were receiving second-line treatment, and 1 (0.7%) was receiving third-line treatment. The majority of patients (94, 65.3%) underwent TKI dose reduction due to AEs. Treatment included imatinib for 26 patients, dasatinib for 52 patients, nilotinib for 13 patients, and flumatinib for 3 patients. The remaining patients received a reduced dose of TKI due to the desire to stop TKI after achieving a stable DMR, financial burden, and comorbidities. The details are shown in Table 2.
Causes for dose de-escalation by medication.

TKI, tyrosine kinase inhibitor.
Adverse events
Pleural effusion was the leading AE causing TKI dose reduction, affecting 30 patients, representing 31.9% of the cases; in particular, all 30 patients were treated with dasatinib. Twenty (21.3%) patients underwent dose reduction for hematological toxicity (mainly thrombocytopenia and anemia), among whom 8 patients received imatinib and 11 received dasatinib. The remaining common AEs were dermatological disturbances in 19.1% of patients, renal failure in 12.8% of patients, and hepatic toxicity in 11.7% of patients. AEs according to medication are detailed in Table 2. After dose reduction, more than half of the AEs (61.3%) were alleviated to some degree, with 36% completely disappearing. The frequencies of patients with improved TKI-associated AEs are presented in Table 3.
Number of patients with lower-grade adverse events after TKI dose reduction.
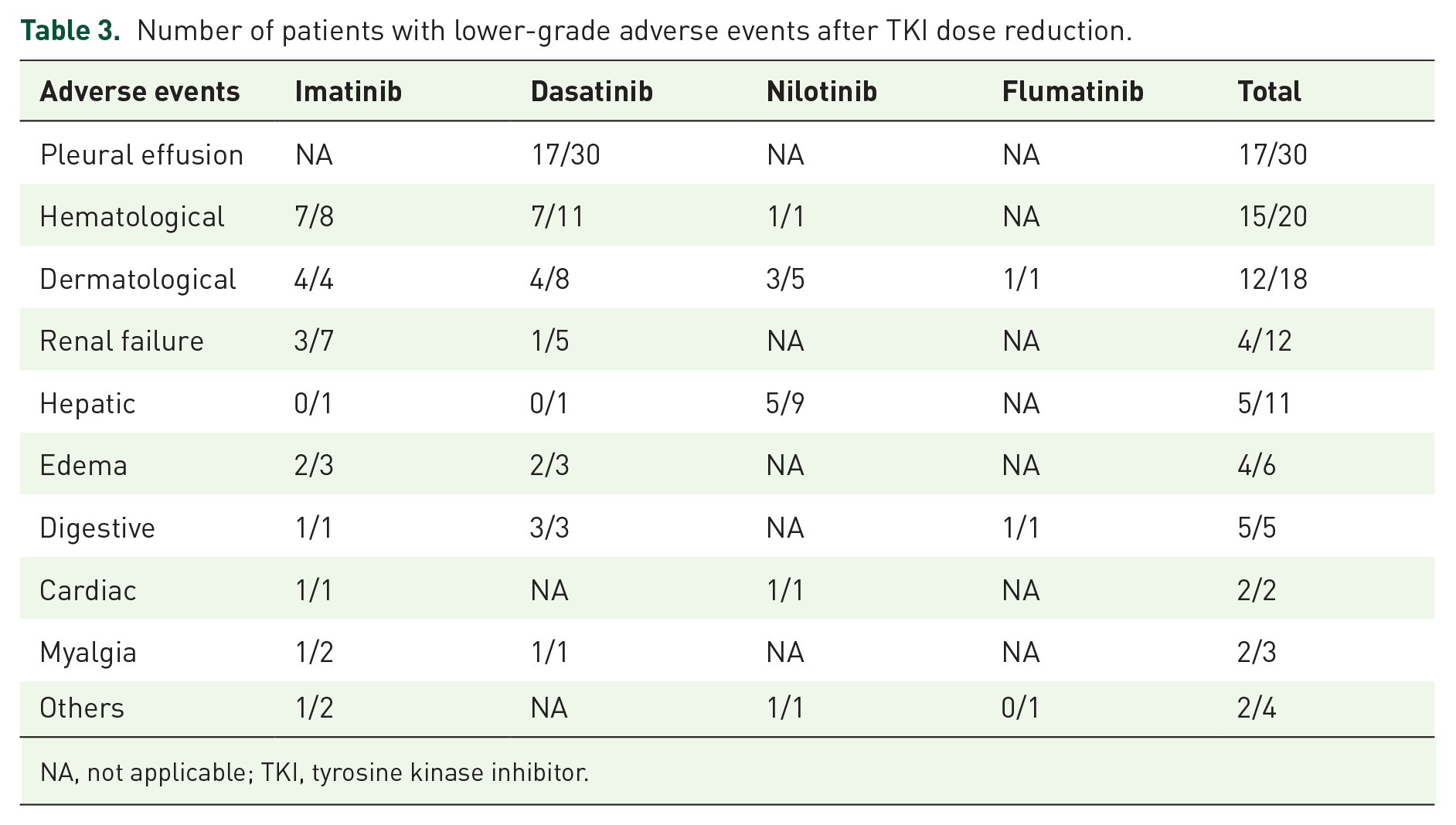
NA, not applicable; TKI, tyrosine kinase inhibitor.
Due to severe AEs in patients even after reducing the dose of TKI, 10 patients discontinued their current low-dose TKI and transitioned to an alternative TKI. All 10 patients were treated with dasatinib and experienced low-dose treatment due to AEs, which were not alleviated after dose reduction. The adverse effects associated with discontinuing TKI included pleural effusion (
Molecular response
None of the 136 patients who had reached MMR at the point of TKI dose reduction reported a loss in MMR over a median follow-up duration of 16 months (range, 3–163) after lowering the TKI dose. In addition, among the eight patients who had not yet achieved MMR at dose reduction, five additional achieved MMR or a deeper molecular response for the first time during dose reduction, with three achieving MR4 or a deeper response.
A total of 122 (92.4%) out of 132 patients who achieved MR4 or deeper at dose reduction experienced maintenance in MR4 or deeper during low-dose TKI treatment. The estimated 1- and 2-year MR4 failure-free survival rates were 96.5% (95% CI: 90.8–98.7) and 90.5% (95% CI: 81.3–95.3), respectively (Figure 1). There are no statistical differences in MR4 failure-free survival among different TKIs (

MR4 failure-free survival for 132 patients who achieved MR4 prior to dose reduction.

MR4 failure-free survival after TKI dose reduction: (a) MR4 failure-free survival according to gender, (b) MR4 failure-free survival according to median age, (c) MR4 failure-free survival according to Sokal risk score, and (d) MR4 failure-free survival according to ELTS risk score.
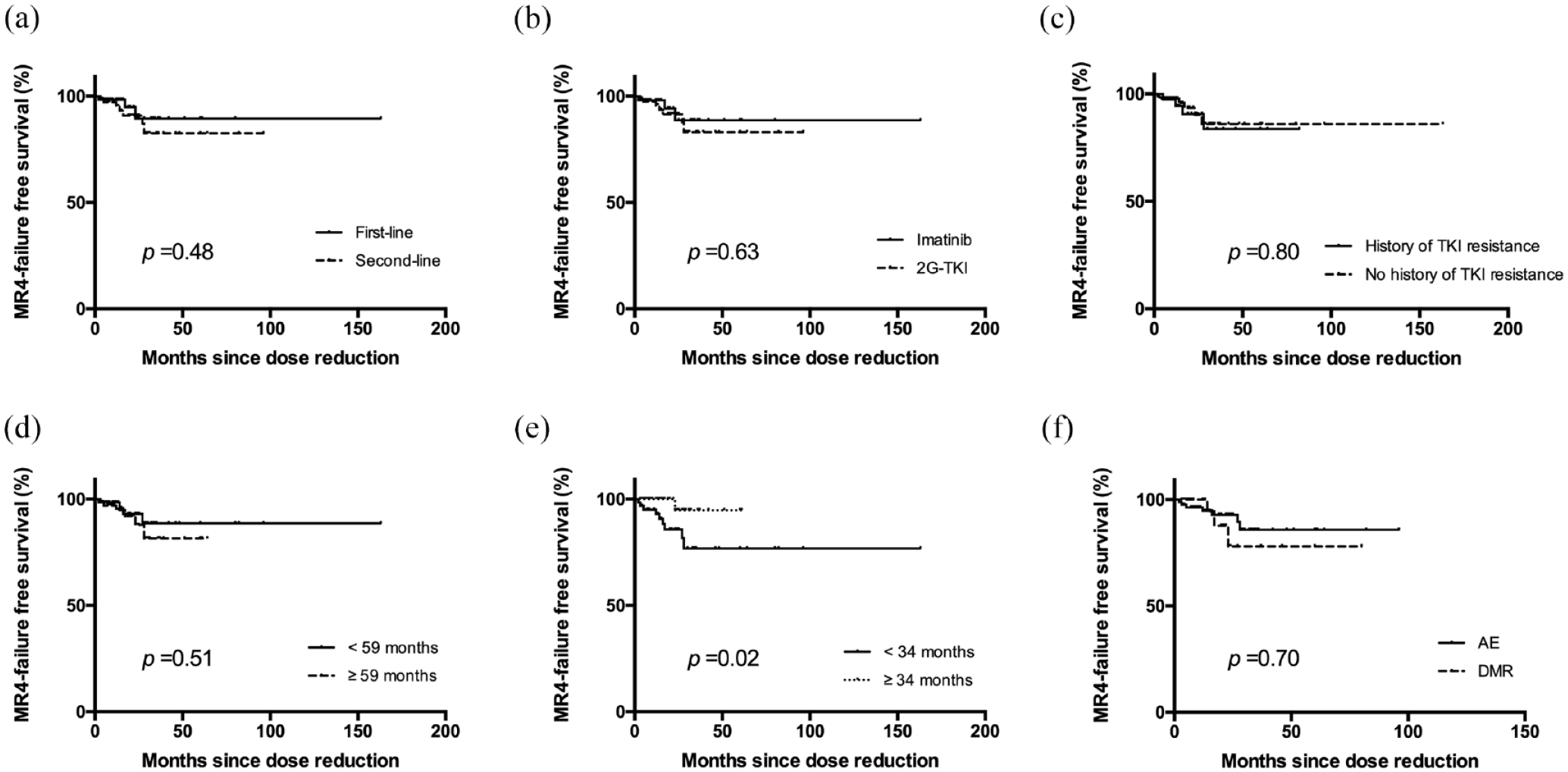
MR4 failure-free survival after TKI dose reduction: (a) MR4 failure-free survival according to treatment lines, (B) MR4 failure-free survival according to medication, (c) MR4 failure-free survival according to whether resistance to previous TKI, (d) MR4 failure-free survival according to median duration of TKI treatment at the onset of dose reduction, (e) MR4 failure-free survival according to median duration of MR4 prior to dose de-escalation, and (f) MR4 failure-free survival according to main reasons for dose reduction.
After a median monitoring period of 16 months on reduced-dose TKI treatment, 10 patients lost MR4. This group comprised three patients who were receiving imatinib and seven patients who were receiving dasatinib. The median interval from the time the dose was reduced to MR4 loss was 15 months (range, 3–28). Among the 10 patients who lost MR4, 6 patients continued with low-dose TKI treatment because they did not lose their MMR, while 2 patients chose to resume full-dose TKI treatment based on their wishes. Another two patients switched from imatinib to dasatinib due to the fear of disease recurrence and concomitant AEs. By the time of the last follow-up, 4 out of these 10 patients had successfully regained MR4.
Two patients who achieved a stable molecular response during low-dose treatment further stopped low-dose TKI therapy, one to pursue TFR, and the other to prepare for pregnancy. Unfortunately, one patient experienced loss of MMR 5 months following the discontinuation of TKI therapy and did not resume TKI treatment because of pregnancy.
Discussion
With the widespread use of TKIs, disease control is achieved in many patients diagnosed with CML. However, lifelong exposure to TKIs correlates with numerous chronic adverse reactions, resulting in TKI dose de-escalation or even the discontinuation of the treatment to alleviate adverse effects, maintain ongoing TKI treatment, and enhance patients’ quality of life. In addition, TKI dose reduction also occurs due to comorbidities or financial stress. Several studies focused on lower doses of TKIs have demonstrated that a decrease in dosage can successfully sustain an MMR and relieve AEs in real-life practice, but more evidence is needed.14–16,18,21
In the present study, 144 CML patients receiving a reduced dose of TKIs in a real-world scenario were analyzed to assess the outcomes of treatment de-escalation. All patients who achieved MMR upon dose reduction maintained their MMR status throughout treatment with lower doses during the follow-up period, and MR4 failure-free survival at 2 years was 90.5%, indicating that low-dose TKI therapy did not compromise efficacy. Similarly, a prospective study included 52 patients who had sustained MR4 or deeper for a minimum duration of 6 months and were treated with low-dose TKI, and the results showed that nearly all participants (50 out of 52) persistently maintained their MR4 status with the reduced TKI dosage.
21
Only 7% of patients underwent molecular recurrence for 12 months in the DESTINY study.
15
A Chinese study reported that the rates of MR4 failure-free survival at 1 and 2 years were 87.8% and 86.1%, respectively.
16
Claudiani
In this manuscript, univariate analysis showed that the loss of MR4 is affected only by the duration of MR4 prior to dose reduction but not by the category of TKI administered during reduced-dose treatment, inconsistent with prior findings reported by Chen
Dose reduction is a commonly used method to alleviate TKI-associated AEs in clinical practice. In the present study, 61.3% of AEs improved after dose reduction. Moreover, in the DESTINY study and study conducted by Chen
TFR represents a significant new target in treatment strategies for CML. For patients who achieve a sustained DMR for a sufficient duration, an attempt at TKI discontinuation can be considered. Typically, the discontinuation of a TKI following a sustained DMR correlates with a 40–60% rate of TFR.7–9 Notably, among patients who stop taking TKIs directly, the majority of molecular relapses, exceeding 80%, occur in the initial 6–8 months post-cessation of TKI treatment, mirroring the scenario seen in patients who first undergo a low-dose TKI regimen before halting the treatment.8,11,18 Claudiani
Despite being a retrospective real-world study, this research has its limitations. The follow-up time was not long enough that no one experienced loss of MMR, so we did not evaluate the factors associated with molecular recurrence. Moreover, the sample size was not large enough, with only a small sample size of patients administered nilotinib and flumatinib, making it difficult to compare the difference in molecular response and remission of AEs among patients receiving different TKIs after dose reduction. In addition, each patient displayed their schedule for dose reduction, making it impossible to identify the best dose reduction strategy to alleviate side effects in this study.
In summary, the data of this retrospective study support the feasibility of lowering the doses of TKIs in patients who have maintained a sustained DMR in a clinical setting. Dose diminution is effective in reducing adverse effects and maintaining a DMR. A longer MR4 duration before dose decrease is associated with a lower loss of MR4. Although the results reported thus far are promising, many issues remain unsolved, and thus, more studies are still needed.
Supplemental Material
sj-tiff-1-tah-10.1177_20406207241259678 – Supplemental material for Successful maintenance of a sustained molecular response in CML patients receiving low-dose tyrosine kinase inhibitors
Supplemental material, sj-tiff-1-tah-10.1177_20406207241259678 for Successful maintenance of a sustained molecular response in CML patients receiving low-dose tyrosine kinase inhibitors by Yan Li, Pu Kuang, Huanling Zhu, Ling Pan, Tian Dong, Ting Lin, Yu Chen and Yunfan Yang in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
