Abstract
Introduction:
The choice of BCR-ABL1 tyrosine kinase inhibitors (TKI) for the first line of therapy (LOT) for chronic-phase chronic myeloid leukemia (CML) is tailored to disease risk and patient characteristics like comorbidities, which become more prevalent with age. However, contemporary evaluations of frontline TKI choice and the factors associated with TKI switching in this specific patient population are lacking.
Methods:
We sought to describe TKI use in older patients (age: 66–99 years) with CML in the United States. Using the Surveillance, Epidemiology, and End Results–Medicare-linked database, we identified 810 older (median age: 75 years, interquartile range: 70–80 years) patients diagnosed during 2007–2015.
Results:
Imatinib was the most common frontline TKI (63.1%) throughout the study period, but its utilization as such decreased from 76% in 2010 to 47% in 2015. Most patients (65.3%) used only one TKI, but 12.5% of the 281 patients who switched from frontline TKI received ⩾4 LOT. Among the 167 patients switching from frontline imatinib, 18.6% eventually returned to imatinib with nearly all as the third LOT, supporting its favorable safety profile and indicating that the initial switch from imatinib might have been premature. Older patients within our cohort, white patients and those with greater comorbidity were less likely to switch from frontline TKI. Diagnosis year, geographic region, and surrogates for socioeconomic status and healthcare access had no impact on TKI switching.
Conclusion:
As expected, our findings highlight the frequent use of imatinib as the treatment option for older CML patients despite the availability of second-generation TKIs.
Introduction
BCR-ABL1-directed tyrosine kinase inhibitor (TKI) therapy is the standard of care for patients with chronic-phase chronic myeloid leukemia (CML). Imatinib, a first-generation (1G) TKI, was approved by the United States (US) Food and Drug Administration in 2001 and has changed the landscape of CML treatment. 1 Since the approval of imatinib, several later-generation TKIs have been approved for the treatment of CML, including the second-generation (2G) TKIs, such as dasatinib in 2010, nilotinib in 2010, and bosutinib in 2017, for frontline treatment.2–4 The 3G TKI ponatinib was approved in 2012 for patients not benefiting from other TKIs and for CML characterized by a T315I tyrosine kinase–domain mutation. 5 Although 2G TKIs do not show improved survival when compared with imatinib in the frontline setting, patients receiving them are more likely to achieve earlier and deeper responses as well as to become eligible for treatment discontinuation. 6 Initial TKI therapy is well tolerated and beneficial in many patients, but at least one-third of patients will require subsequent use of one or more different TKIs. 7 Sequencing of TKIs has become common in clinics and the proper selection of TKIs is likely to improve outcomes and resource utilization. However, the optimal consecutive use of TKIs is unclear and understudied, particularly in the third-line setting or beyond.
The choice of TKI is in part dictated by its side-effect profile, which is an especially important consideration for the near half of patients with CML in the United States who are diagnosed at age 60 years or older. 8 Insurance coverage options and quality, income and cost issues, and other older patient-specific factors are also likely to influence treatment choices. 9 “Real-world” analyses describing the contemporary practice patterns of older CML patients in the United States are limited. The five currently approved TKIs have become the standard of care and comprise a complex treatment landscape for older patients, who were not well represented in the prospective TKI trials that excluded many patients due to comorbidities or did not evaluate the entire scope of treatment patterns.10–14 Other studies evaluating the frontline TKI choice of US providers described CML patients in general and not specifically the older adult population for whom comorbidity is more likely to influence this decision.11,15–19 Little is known about the utilization of the second or third line of therapy (LOT) after discontinuation of frontline TKI among older patients with CML as the timeframe of many studies did not incorporate patients who were able to receive multiple LOTs. Furthermore, little is known regarding the factors that are associated with TKI switching in older patients with CML, including age, comorbidities, insurance/socioeconomic status, and healthcare access. To better understand the spectrum of contemporary TKI use among older patients with CML, we leveraged the Surveillance, Epidemiology, and End Results (SEER)–Medicare-linked database to longitudinally evaluate TKI use patterns, sequencing as well as factors associated with TKI switching among this understudied group of patients.
Methods
Data source
Using the SEER–Medicare-linked database, we assembled a population-based cohort of CML patients who initiated TKIs after CML diagnosis. The SEER-Medicare database links patient-level information on incident cancer diagnoses reported to the SEER registries with a master file of Medicare enrollment and claims for inpatient, outpatient, physician services, hospice care, home health agencies, durable medical equipment, and prescription drugs. 20 The SEER registries are population-based and covering approximately 35% of the US population. 21 Ethics approval was not sought for this study because the study did not involve human subjects—we did not obtain data through interventions or interaction with any individual or obtain identifiable private information. The reporting of this study conforms to the STROBE statement. A checklist of the guidelines is submitted as Supplementary Material.
Study population
We identified older adults diagnosed with CML (International Classification of Diseases for Oncology, 3rd edition codes: 9875, 9863) during 2007 to 2015 who fulfilled the following criteria: (1) were aged 66 (to obtain Medicare claims for 1 year before CML diagnosis to assess comorbidities) to 99 (to avoid potential changes in the pattern of care for patients ⩾100 years22,23) years at diagnosis, (2) had known month of diagnosis, (3) were not reported from autopsy or death certificate only, (4) had continuous Medicare fee-for-service Parts A/B and Part D coverage from 1 year and 3 months, respectively, before diagnosis to end of follow-up (change in Medicare status, death, or 12/31/2016, whichever came first), (5) initiated TKI therapy after CML diagnosis, and (6) had been followed for more than 3 months after CML diagnosis. Diagnosis year and month from were obtained from SEER with the first day of the month used to identify the date of diagnosis. To ensure that patients initiated TKIs after CML diagnosis, we searched patient’s Part D claims in the 3 months before diagnosis and excluded those receiving TKIs during that period. A follow-up of ⩾3 months was required to avoid including patients who were close to death and were less likely to receive any CML-directed therapy (Figure 1).

Construction of the study cohort.
Treatments
We used Part D claims to identify TKIs (imatinib, bosutinib, dasatinib, nilotinib, and ponatinib) for treating CML during our study period. We used the dispense date and the days of supply to estimate TKI supply available during follow-up. As patients may switch from one TKI to another and two different TKIs could not be taken on a same day, if a person had overlapping supply of more than one TKI, we assumed the patient started to use the second TKI and discontinued the first one. For example, if a patient had a 30-day supply of imatinib on 1/1/2008 and later a 30-day supply of dasatinib on 1/15/2008, we assumed the patient stopped imatinib on 1/14/2008 and started to use dasatinib on 1/15/2008. If a dispense coverage exceeded the end of follow-up, then the dispense would be censored at the end of follow-up date. A switch from one TKI to another was defined as a new TKI LOT. Duration of a LOT was calculated as the days between first dispense date to the end of last dispense. Time to first switch was measured from the first dispense date to the date of the first switch, and observation periods were censored if patients did not switch by the end of the study period. The standard dose of each frontline TKI was defined as 400 mg daily for imatinib, 100 mg daily for dasatinib, 300 mg twice a day for nilotinib, 400 mg daily for bosutinib, and 45 mg daily for ponatinib.
Statistical analysis
Descriptive statistics were used to summarize baseline patient characteristics and TKI use. Categorical variables were presented using frequencies and percentages, and continuous variables were summarized by median and interquartile range (IQR). Consistent with SEER–Medicare requirement to preserve confidentiality, all categories with 10 or fewer patients were reported as <11. Baseline patient characteristics included age at diagnosis, sex, race, year of diagnosis, marital status, type of residential area (big metropolitan area, metropolitan area, or other), SEER region (Northeast, Midwest, South, or West), modified Elixhauser comorbidity index (described below), history of other malignancies, previous cardiovascular diseases (CVDs), previous pulmonary diseases, previous CVD risk factors (hypertension, hyperlipidemia, diabetes mellitus, and tobacco use), low-income subsidy (a marker for reduced out-of-pocket cost sharing and lower socioeconomic status), percentage of population below poverty at the census tract level (a proxy for neighborhood socioeconomic status), and receipt of influenza vaccination in the 12 months prior to CML diagnosis (an indicator for access to the healthcare system 24 ). To assess comorbidities, CVD, pulmonary diseases, CVD risk factors, we used inpatient, outpatient, and carrier claims within 12 months prior to CML diagnosis that appeared on any inpatient claims or at least two outpatient/physician claims more than 30 days apart. 25 As we created individual indicators of prior CVD, pulmonary diseases, and CVD risk factors, a modified Elixhauser score was developed by removing those already included in the CVD and pulmonary disease-related indicators from the original Elixhauser score.25,26 Univariate and multivariable logistic regression models were used to compare likelihood of choosing imatinib as the frontline TKI between the youngest (age 66–69 years) and oldest (age 85–99 years) patients. Taking time to switch into consideration, we used a multivariable Cox proportional hazards model with death and change of insurance as competing risks to assess impact of frontline TKI agent and baseline patient characteristics on switching. All analyses were conducted using SAS, version 9.4 (SAS Institute Inc., Cary, NC) with two-sided tests and a type-I error of 5% as the threshold for statistical significance.
Results
We identified 810 patients with newly diagnosed CML initiating a TKI after CML diagnosis and followed them for a median of 2.84 (IQR: 1.47–4.72) years since TKI initiation. The median age was 75 (IQR: 70–80) years with 52.0% female and 87.0% white (Table 1).
Characteristics of 810 older patients with chronic myeloid leukemia who received BCR-ABL1 tyrosine kinase inhibitors, 2007–2015.

Cardiovascular disease risk factors included hypertension, hyperlipidemia, diabetes mellitus, and tobacco use.
Of the five TKIs studied, imatinib was used by 72.3% of patients, followed by dasatinib (38.8%) and nilotinib (28.8%). Bosutinib, the latest available TKI during the study timeframe, was used by 38 (4.7%) patients. Ponatinib was the least-used TKI with only 16 patients (2.0%) ever-receiving it and nearly all patients using it as third or later LOT. In addition, 24.9% and 7.9% patients received two and three different TKIs, respectively. Only 15 (1.8%) patients received ⩾4 different TKIs with <11 receiving all five TKIs.
A majority of patients received one (65.3%) or two (21.1%) LOTs. However, some patients returned to a previously abandoned TKI as a subsequent LOT and accounts for why the number of LOT may exceed the number of TKIs used for a given patient. Only 35 (12.5%) patients had received ⩾4 LOT, with some (<11) receiving up to 7 LOT. The median duration of frontline TKI was 18.4 months (range: 5.1–38.8) with a median duration that varied across frontline agents, ranging from 12.7 months for dasatinib to 21.9 months for imatinib. The median duration of second, third, and fourth LOT was 10.5 (range: 2.7–28.8), 8.0 (range: 2.8–23.7) and 4.5 (range: 0.4–11.8) months, respectively.
Frontline TKI choice and patterns
The median time from diagnosis to the initiation of frontline TKI therapy was 46 (IQR: 30–83) days, with 78.3% of patients initiating TKI within 90 days after diagnosis. The proportion of patients intiating TKI within 90 days slightly increased from 79.5% of patients diagnosed in 2007 to 86.7% of those diagnosed in 2015. The median time to initiation was similar among imatinib (44 days, IQR: 28–80 days) and dasatinib (46 days, IQR: 31–74 days) users, with a longer median time to initiation (54 days, IQR 36–117 days) among nilotinib users; correspondently, the proportion of patients intiated within 90 days was 79.1%, 84.0%, and 65.8% among imatinib, dasatinib, and nilotinib users, respectively (
As expected, imatinib (63.1%) was the most frequently used frontline TKI in this older patient population (Figure 2), and the use of imatinib as frontline TKI reached a peak in 2009 then decreased during 2010–2015 (Figure 3). During the same time, use of dasatinib and nilotinib, especially dasatinib, as the frontline TKI increased. Among those diagnosed in 2015, 41.7% patients received dasatinib as frontline TKI, which was nearly the same proportion as imatinib (46.7%). The oldest patients in our study population (age 85–99 years at diagnosis) were more likely to receive frontline imatinib

Proportion of patients by BCR-ABL1 tyrosine kinase inhibitors and line of therapy among older patients with chronic myeloid leukemia, 2007–2015.

Proportion of patients by frontline BCR-ABL1 tyrosine kinase inhibitors and year of diagnosis among older patients with chronic myeloid leukemia, 2007–2015.
TKI switching and patterns
A total of 281 (35.1%) TKI users eventually switched to another TKI. About half (
Among these 281 switched patients, 59.4% and 29.2% received imatinib and dasatinib as frontline TKI, respectively. Nearly all patients switching from frontline imatinib received a 2G TKI as the second LOT, with 58.7% and 38.3% switching to dasatinib and nilotinib, respectively. Only 5.3% of patients used bosutinib or ponatinib as the second LOT (Figure 2). Of 113 patients who switched from a frontline 2G TKI, 60.8% received imatinib as the second LOT. Patients who switched from frontline dasatinib mainly received imatinib (59.8%) and nilotinib (31.7%) as their second LOT. Among patients switching from frontline nilotinib, 61.3% switched to imatinib and no more than 10 patients switched to either dasatinib or bosutinib. Among 110 patients who received a third LOT, imatinib (30.9%) was the most common, followed by nilotinib (27.3%), dasatinib (21.8%), and bosutinib or ponatinib (20.0%). The most common TKI sequences for patients who received up to four LOT are presented in Table 2.
Most common treatment sequence by total number of lines of therapy with BCR-ABL1 tyrosine kinase inhibitors among older patients with chronic myeloid leukemia, 2007–2015.
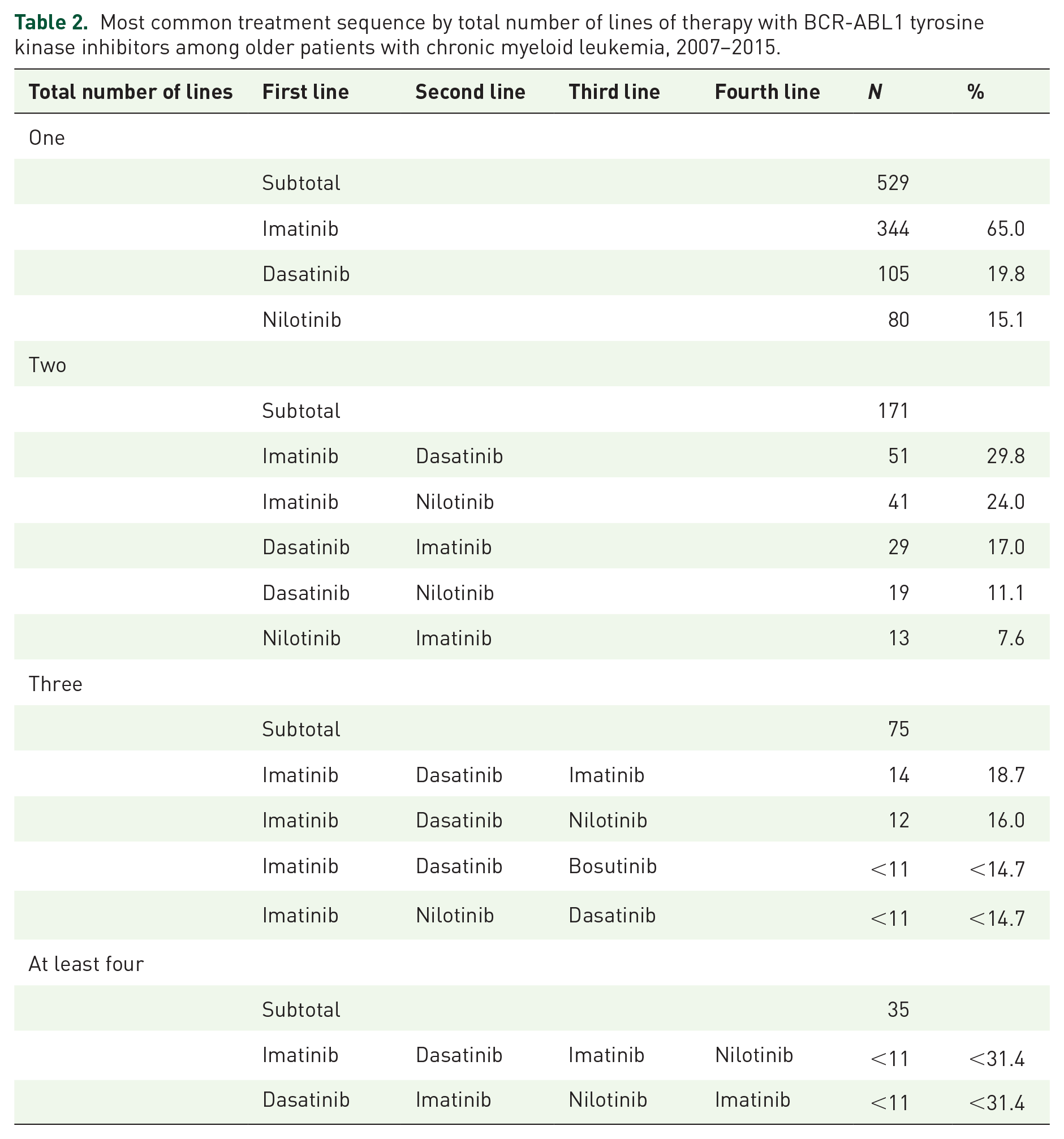
Of the 71 patients who switched from imatinib as first LOT and received at least 3 LOTs, 43.7% returned to imatinib. Only 28.6% and 27.2% of patients who were treated with frontline dasatinib or nilotinib and received at least 3 LOTs subsequently used these 2G TKIs. Of 42 patients who switched back to the frontline TKI agent, more than 90% used them as the third LOT.
Similar proportions of patients among those who switched (85.1%) and who did not switch (86.4%) received a standard initiation dose (
Predictors of switching
Multivariable analysis showed that patients who were on frontline dasatinib were more likely to switch to a second LOT than those on imatinib (hazard ratio (HR) = 1.90, 95% confidence interval (CI) = 1.35–2.66;
Factors associated with switching of BCR-ABL1 tyrosine kinase inhibitors among older patients with chronic myeloid leukemia, 2007–2015.
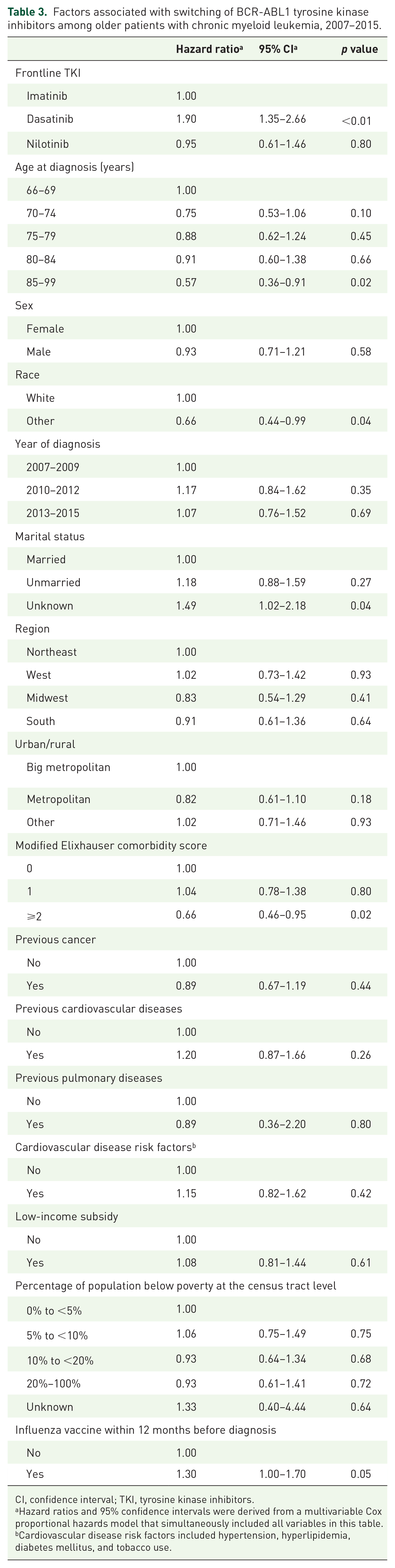
CI, confidence interval; TKI, tyrosine kinase inhibitors.
Hazard ratios and 95% confidence intervals were derived from a multivariable Cox proportional hazards model that simultaneously included all variables in this table.
Cardiovascular disease risk factors included hypertension, hyperlipidemia, diabetes mellitus, and tobacco use.
Discussion
In this large, contemporary, population-based study, we report the real-world patterns of TKI use among 810 older patients with CML. As expected, the prevalence of relevant comorbidities in our cohort was noteworthy; 26% of patients had underlying CVD and 86% had at least one CVD risk factor. Only 3% of patients had underlying pulmonary disease. Even though imatinib was the most common frontline and ever-used TKI, its utilization as frontline therapy declined during the study period as the use of 2G TKIs increased.
Among older patients, there is an increased prevalence of comorbidities,8,9 as well as more common chronic medication use that augments the potential for drug–drug interaction and increases the likelihood of therapy toxicities. 27 These issues are relevant in the context of the toxicity profiles associated with TKIs. Imatinib may lead to superficial edema and nausea, with other common toxicities including musculoskeletal discomfort, rash, fatigue, diarrhea, and headache in half of patients but is associated with less major toxicities than the other TKIs.4,28,29 The more potent 2G TKIs have more prominent side effects with dasatinib associated with pleural and pericardial effusions and pulmonary hypertension,2,28 nilotinib with arterio-occlusive events (AOEs) and pancreatitis, 3 and bosutinib with gastrointestinal toxicity. 4 The most potent 3G TKI ponatinib has the highest risk among all TKIs for serious AOEs. 30 Although limited in number, some retrospective studies have reported a higher rate and severity of toxicities for older CML patients treated with imatinib,4,31 dasatinib, 32 and bosutinib, 4 when compared with younger patients.
Other studies of the broader (and younger) population leveraging alternative registry- and claims-based data sets have demonstrated similar findings regarding the patterns of frontline TKI choice, specifically imatinib being the preferred frontline TKI with a progressive decrease after the availability of 2G TKIs.10–12,19,33 When evaluating special populations like veterans, similarities to our findings are noted. An analysis of 468 patients diagnosed with CML during 2006–2015 using the Veterans Affairs cancer registry noted that imatinib was the most common frontline TKI throughout the study period and accounted for 79% of all new frontline TKI prescriptions during the latter half of the study period; dasatinib and nilotinib were used by 13% and 8% of patients, respectively, with older age, higher comorbidity, and living in a rural area associated with lesser frontline 2G TKI use. 33 Similar to our results, updated results from the SIMPLICITY study, which explored the management of >1000 patients (one-third aged >65 years) with chronic-phase CML, showed that there was a shift from the use of imatinib as the frontline TKI to 2G TKIs from 2009 to 2015. 19 Furthermore, one-third of patients discontinued/switched from frontline TKI with the most common reason for such being intolerance. 19 We could not investigate the reasons for switching in our study due to the lack of such information in the SEER–Medicare database, but the observation that 60.8% of the 113 patients who switched from a frontline 2G TKI received imatinib as the second LOT may be explained by intolerance to 2G TKI rather than refractory/relapsed disease.
Studies that evaluated TKI utilization using US health plan claims data and retrospective cohorts of patients from multiple integrated healthcare systems have reported similar 20%–30% rates of eventual switching from frontline TKI with dasatinib being the preferred second LOT.10,12 These findings are consistent with other analyses of the general CML population, 7 but our study confirms the same pattern among older CML patients and enhances an understanding of the “real-world” practices beyond frontline TKI choices and beyond first year of therapy.14,34 A progressive decrease in median time on TKI with increasing LOT was observed. Few patients who switched from a frontline 2G TKI eventually returned to it during a later LOT. In contrast, nearly 20% of patients who switched from frontline imatinib eventually returned to imatinib, suggesting that in retrospect the toxicities or other issues prompting the initial switch might have been tolerable or manageable, respectively. Despite the 2G TKIs dasatinib and nilotinib being approved for frontline therapy the same year (2010), dasatinib was more commonly used than nilotinib. In addition, although approved for use in 2012, ponatinib was rarely used in our older patient population, likely reflecting providers’ concern over its proven risk of AOEs in a population with a greater prevalence of underlying CVD and CVD risk factors.
Non-white patients were less likely to switch from their frontline TKI. Prior analyses of older US patients participating in fee-for-service Medicare have demonstrated that white patients were more likely than their non-white counterparts to visit a specialist. 35 In addition, a previous SEER-based analysis of older patients with CML found that, although the survival of white patients significantly improved in the first 5 years after imatinib became available, this was not the case for black patients. 36 These observations have been attributed to differences in supplemental insurance coverage, socioeconomic status, and general access to healthcare. In our study, older patients and those with higher comorbidity burden as measured by a modified Elixhauser comorbidity score were less likely to switch from frontline TKI as well. Most patients in our study received frontline TKI at the recommended dose for the treatment of chronic-phase CML, but “non-standard” doses did not influence the likelihood of switching. We did observe, however, that patients who had the dose of their frontline TKI eventually decreased were less likely to switch with dasatinib-treated patients constituting the bulk of this observed difference. This may suggest that there was an attempt by providers to continue with lower doses as opposed to a TKI switch in response to TKI-related side effects.
Our study was subject to limitations inherent in the use of claims data. The SEER–Medicare database does not include detailed clinical, laboratory, and pathologic information for us to assess patient disease risk or phase and understand the reason for TKI switching, which in addition to tolerability issues could include lack of response and disease progression. Our study sample was limited to patients with continuous Medicare fee-for-service coverage, thus our results may not be generalizable to Medicare beneficiaries enrolled in Medicare Advantage plans. The introduction of generic imatinib preparations in 2016 significantly decreased the cost for this TKI and may potentially influence prescription practices for CML patients. We did not investigate the effect of generic imatinib preparations as this was beyond the scope of our study, which involved patients diagnosed with CML up to 2015 and followed-up to the end of 2016. Finally, bosutinib was approved for frontline use in 2017, which is beyond our study period and, hence, was not commonly used by our patients. Considering its acceptable toxicity profile and clinicians’ increasing familiarity with this 2G TKI, bosutinib may play a more prominent role as a frontline TKI and subsequent LOT for older CML patients in more recent years.
Despite these possible limitations, our study sample was large and population-based, and we incorporated data on a broad range of patient characteristics including sociodemographic factors and comorbidities. In addition, we were able to assess the healthcare received by patients longitudinally and comprehensively, following up patients well beyond frontline TKI therapy and beyond the first year after CML diagnosis.
In conclusion, despite the potential for pulmonary toxicity and AOEs with dasatinib and nilotinib, respectively, these TKIs were used in a substantial proportion of older CML patients. Even though imatinib utilization decreased over time, it remained an important treatment option for this patient population in the frontline setting. Furthermore, one-quarter of patients who moved onto second LOT received imatinib after frontline use of a 2G TKI. One-third of TKI users switched to another TKI, with one in five patients switching from imatinib eventually returning to imatinib, indicating that the initial switch from this 1G TKI might have been premature. Based on our findings during the era with multiple 2G TKIs, imatinib continues to play an important role in the management of older patients with CML.
Supplemental Material
sj-doc-1-tah-10.1177_20406207211043404 – Supplemental material for Contemporary practice patterns of tyrosine kinase inhibitor use among older patients with chronic myeloid leukemia in the United States
Supplemental material, sj-doc-1-tah-10.1177_20406207211043404 for Contemporary practice patterns of tyrosine kinase inhibitor use among older patients with chronic myeloid leukemia in the United States by Rory M. Shallis, Rong Wang, Jan P. Bewersdorf, Amer M. Zeidan, Amy J. Davidoff, Scott F. Huntington, Nikolai A. Podoltsev and Xiaomei Ma in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The authors acknowledge the efforts of the Applied Research Program, National Cancer Institute; the Office of Research, Development and Information, Centers for Medicare and Medicaid Services; Information Management Services, Inc.; and the SEER Program tumor registries in the creation of the SEER–Medicare database. The interpretation and reporting of the SEER–Medicare data are the sole responsibility of the authors. The collection of the California cancer incidence data used in this study was supported by the California Department of Public Health as part of the statewide cancer reporting program mandated by California Health and Safety Code Section 103885; the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) Program under contract N01 PC 35136 awarded to the Northern California Cancer Center, contract N01 PC 35139 awarded to the University of Southern California, and contract N02 PC 15105 awarded to the Public Health Institute; and the Centers for Disease Control and Prevention’s National Program of Cancer Registries, under agreement #U55/CCR921930 02 awarded to the Public Health Institute. The ideas and opinions expressed herein are those of the author(s) and endorsement by the State of California, Department of Public Health, the National Cancer Institute, and the Centers for Disease Control and Prevention or their contractors and subcontractors are not intended nor should be inferred.
Author’s note
This work was presented in part at the 62nd American Society of Hematology Annual Meeting on 7 December 2020.
Conflict of interest statement
A.M.Z. received research funding (institutional) from Celgene, Acceleron, Abbvie, Otsuka, Pfizer, Medimmune/AstraZeneca, Boehringer Ingelheim, Trovagene, Incyte, Takeda, and ADC Therapeutics. A.M.Z. had a consultancy with and received honoraria from AbbVie, Otsuka, Pfizer, Celgene, Ariad, Incyte, Agios, Boehringer Ingelheim, Novartis, Acceleron, Astellas, Daiichi Sankyo, Cardinal Health, Seattle Genetics, BeyondSpring, and Takeda. A.D. received research funding from Celgene Corp. N.A.P. consulted for and received honoraria from Alexion, Pfizer, Agios Pharmaceuticals, Blueprint Medicines, Incyte, Novartis, Celgene, Bristol Myers Squib (BMS), CTI BioPharma, and PharmaEssentia. N.A.P. received research funding (all to the institution) from Boehringer Ingelheim, Astellas Pharma, Daiichi Sankyo, Sunesis Pharmaceuticals, Jazz Pharmaceuticals, Pfizer, Astex Pharmaceuticals, CTI biopharma, Celgene, Genentech, AI Therapeutics, Samus Therapeutics, Arog Pharmaceuticals, and Kartos Therapeutics. X.M. received research funding (institutional) from Celgene/BMS and is a consultant for BMS. None of these relationships were related to the development of this work. Other authors have nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Frederick A. Deluca Foundation.
Ethical approval
The Yale Human Investigation Committee determined that this study did not directly involve human subjects.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
