Abstract
Background:
Currently, the goal of chronic myeloid leukemia (CML) treatment is normal survival and good quality of life without life-long treatment, namely, “treatment-free remission” (TFR). At present, approximately only 50% of patients with CML with a deep molecular response are able to discontinue tyrosine kinase inhibitor (TKI) without experiencing molecular relapse [MR; loss of major molecular response (MMR)]. In addition, prior interferon (IFN) treatment is associated with a higher rate of TFR.
Methods:
We aimed to evaluate the feasibility of TKI discontinuation in Chinese patients with CML and determine whether IFN could prevent MR when used after TKI discontinuation in patients with 0.0032% <BCR-ABLIS ⩽0.1%. Therefore, we retrospectively analyzed the data of patients with CML who discontinued TKI treatment at our center.
Results:
Forty-nine patients who discontinued TKI therapy after achieving MR 4.5 were included in this study, and the median follow-up time from TKI discontinuation was 27 (7, 75) months. Nineteen patients eventually lost MMR, and the TFR rate of the 49 patients was 67% (95% confidence interval 53.6%, 80.3%) at 12 months. The duration of MR 4.5 ⩾54 months and duration of imatinib ⩾85 months were significantly associated with a higher TFR rate. Of the 22 patients with 0.0032% <BCR-ABLIS ⩽0.1%, 12 received IFN-α treatment. The median IFN-α therapy duration was nine (2, 18) months, and three patients eventually lost MMR. Three patients discontinued IFN-α after 2, 2.5, and 10 months, and maintained MMR for 9, 8, and 11 months after IFN discontinuation, respectively. Of the 10 patients not receiving IFN-α treatment, eight eventually lost MMR. The MR-free survival rate was significantly different between the patients treated with and those treated without IFN-α over 24 months (70.7% versus 15.0%, p = 0.002).
Conclusion:
These results indicate that after TKI discontinuation, IFN-α can be administered to patients with 0.0032% <BCR-ABLIS ⩽0.1%, which may help prevent MR.
Introduction
Chronic myeloid leukemia (CML) is characterized by the presence of the Philadelphia chromosome and its molecular counterpart, the BCR-ABL1 fusion gene, making it a model disease for targeted therapy.1,2 CML accounts for 15% of all adult leukemias, with an annual incidence of 1.6–2 per 100,000 globally.3,4
Treatment with tyrosine kinase inhibitors (TKIs) has greatly improved the survival of patients with CML, with TKI-associated overall survival rates of 85–90% at 10 years.5–8 Although TKIs are well-tolerated compared with the majority of antineoplastic drugs, this chronic therapy may impact patients’ lives through either its associated adverse events (such as peripheral edema and muscle cramps with imatinib, pleural effusions with dasatinib, and vascular events with many), its influence on real-life events (e.g. pregnancy), or the financial burden to patients and society.9–13 More recently, there has been increasing focus on quality of life and avoiding long-term organ toxicities, especially in the identification of strategies to maximize the possibility of stopping TKI therapy resulting in so-called “treatment-free remission” (TFR). 14
Up to 35–68% of patients with CML who are treated with TKIs experience a deep molecular response [molecular relapse (MR) 4.5, defined as BCR-ABL1IS ⩽0.0032%], 14 mainly in those involved in clinical studies of TKI withdrawal. Approximately 11 clinical studies have evaluated patient outcomes following the discontinuation of TKI therapy worldwide, and the 2-year TFR rate after TKI discontinuation is approximately 50%.2,15–20 Therefore, as the interest in and evidence to support treatment discontinuation increases, determining how best to decrease the rate of MR [the loss of the major molecular response (MMR), defined as BCR-ABL1IS ⩽0.1%] after the discontinuation of TKI therapy has become an important area of research. MR or loss of MMR triggers the restarting of TKI therapy in most TKI discontinuation clinical studies,2,15–20 meaning the end of TFR.
Different prognostic factors for success in TFR have been reported. However, longer durations of TKI therapy, deep molecular response, and prior treatment with interferon (IFN)-α have been associated with TFR with a high level of consistency.2,14–20 Therefore, IFN therapy may increase the TFR rate in patients with CML. IFN was the main treatment in the pre-TKI era for patients with CML, and it may play a role in the TKI era. IFN combined with TKIs can improve the rate of TFR.21–23 Several TKI discontinuation studies have shown that patients with low-level BCR-ABL1 positivity after withdrawal eventually lost TFR.16–19 Most likely, if used in patients with positive BCR-ABL1 at low levels, IFN-α may prevent the MR of patients with CML discontinuing TKI treatment. This design may prevent the need to use IFN in all patients with CML and could decrease adverse events.
Therefore, we aimed to evaluate the feasibility of TKI discontinuation in Chinese patients with CML who achieved and sustained MR 4.5, and determine whether IFN-α prevented MR when used after TKI discontinuation in patients with low-level BCR-ABL1 positivity (0.0032% <BCR-ABLIS ⩽0.1%). To the best of our knowledge, this study represents the first investigation into IFN-α treatment after TKI discontinuation according to the levels of BCR-ABL1 through patient monitoring in clinical practice, and is the largest single-institution report on TKI treatment discontinuation in China to date.
Methods
Study design and patients
All patients with CML stopped TKI treatment voluntarily. The medical records of all patients with CML treated with TKIs at Peking University People’s Hospital who subsequently discontinued TKI therapy between 2013 and 2018 were reviewed. Patients with CML who received any TKI treatment in the prior 3 years who had discontinued therapy after a confirmed MR 4.5 (BCR-ABL1IS ⩽0.0032%) that lasted for at least 2 years and had at least 6 months of follow-up data were eligible for this study. The level of molecular response was determined by evaluating BCR-ABL1 positivity, which was measured by real-time quantitative polymerase chain reaction (RT-qPCR) analysis conducted in the laboratory of the Institute of Hematology of Peking University and was reported as the ratio of BCR-ABL1 gene expression to ABL1 gene expression on the International Scale (IS). All patients with CML who discontinued TKI therapy at this hospital voluntarily underwent at least 3 years of follow-up. Fifty-one patients with CML discontinued TKI therapy, and 49 had more than 6 months of follow-up and were included in this analysis. Thirty-three of the 49 patients have participated in several clinical trials, including # STI571-113, registered at www.chictr.org as # CA180-056, CAMN107ECN02, CAMN107A2404, HHGV678-20, and HS-10096-301. All patients provided signed informed consent for their data to be included in this study. Follow-up information was collected until 16 June 2019.
Follow-up
Following the discontinuation of treatment, the patients underwent blood tests that included a whole blood cell count, a peripheral blood smear, tests of hepatorenal function, and tests of BCR-ABL1 levels every month for the first 6 months, followed by testing every 6 weeks until month 12, then every 3 months for at least 3 years. MMR was defined as a blood BCR-ABL1IS level of <0.1%, and MR was defined as the loss of the MMR (BCR-ABL1IS ⩾0.1%).
As part of routine clinical decision-making, patients received further treatment based on the levels of BCR-ABL1IS determined during follow-up. Patients who experienced a MR in any BCR-ABL1 test during follow-up were prescribed TKIs, ideally the same TKI they had previously received before discontinuation. Patients with low-level BCR-ABL1 positivity but who did not meet the criteria for MR, defined as 0.0032% <BCR-ABL1IS <0.1%, received a recommendation to undergo treatment with IFN-α. For the patients who agreed with this recommendation, IFN-α was administered as a three million unit dose hypodermic injection three times per week. This dose was determined based on the methods of a previous study. 21 The treatment continued for at least 12 months if the patient did not experience MR. Patients for whom the first or second positive BCR-ABL1 test indicated a MR were excluded from the analysis. Any adverse events of IFNs were recorded, including influenza-like symptoms, hemocytopenia, and psychiatric disorders.
Statistical analysis
Fisher’s exact test, the Chi-square test, unpaired t-tests, and the Wilcoxon rank-sum test were used, as appropriate, to perform comparisons between two groups. Time to MR was measured from the date of TKI discontinuation to the date of MR, or the date of the last molecular examination for patients who did not undergo MR. MR-free survival was estimated using the Kaplan–Meier method. The MR-free rate was compared between the two groups using the log rank test. Stepwise forward selection multiple logistic analysis for MR by 12 months was performed to determine the effect of the variables examined in univariate analysis. All statistical analyses were performed using SPSS software version 17.0 for Windows (SPSS Inc., Chicago, IL, USA). In all analyses, two-tailed p values < 0.05 were considered statistically significant.
Results
Fifty-one patients with CML at Peking University People’s Hospital discontinued TKI therapy, and 49 had more than 6 months of follow-up and were included in this study. The baseline characteristics and treatment information for the 49 patients are shown in Table 1. The male/female ratio was 28/21, and the median age at discontinuation was 50 (13, 81) years. Two patients were in the accelerated phase (AP), and the remaining patients were in the chronic phase at CML diagnosis. The two CML-AP patients requested to stop TKI treatment, thus, we observed the two patients for drug withdrawal. Patient 1 received 400 mg q.d. of imatinib (bordy surface area (BSA) = 1.3 m2), and reached MMR and MR 4.5 after 12 and 18 months of imatinib treatment, respectively. Patient 2 received second-generation TKI flumatinib (China’s original research second-generation TKI) 24 as first-line treatment, and reached MR 4.5 after 6 months of flumatinib treatment. The Sokal score was calculated for 40 patients (missing data prevented Sokal score calculation for the other nine patients); 26 were classified as low-risk, 12 as intermediate-risk, and two as high-risk. The median time to MR 4.5 was 18 (6, 160) months, and the median MR 4.5 duration before TKI discontinuation was 56 (24, 115) months. Prior to discontinuation, 40 patients were treated with imatinib, and nine were treated with second-generation TKIs. The median duration of TKI therapy before discontinuation was 80 months (38, 201).
Patient characteristics at study entry.

Data are n (%), n/N (%), or median (interquartile range).
TKI, tyrosine kinase inhibitor.
Data were missing for nine patients.
MR 4.5: BCR-ABL1/ABLIS ≤0.0032%.
Outcomes after TKI discontinuation
After TKI discontinuation, the median follow-up duration was 27 (7, 75) months (Table 1). Nineteen patients eventually lost their MMR. The MR-free survival rate was 67% [95% confidence interval (CI) 53.6%, 80.3%] at 12 months and 59.6% (95% CI 45.5%, 73.7%) at 24 months (Figure 1). A total of 17 patients restarted TKI treatment; one was lost to follow-up, and 16 regained an MMR or MR 4.5 within 3 months after restarting treatment. Thirty patients did not lose their MMR, 13 of whom maintained the MR 4.5. Two CML-AP patients did not lose their MMR after TKI discontinuation during 34 and 28.6 months of follow-up, respectively. Two of nine patients (22.2%) with second-generation TKIs lost their MMR, and 17 of 40 patients (42.5%) undergoing imatinib treatment lost their MMR, but the difference was not statistically significant (p = 0.451).

Kaplan–Meier estimates of molecular relapse-free survival after tyrosine kinase inhibitor discontinuation in 49 chronic myeloid leukemia patients.
TKI withdrawal syndrome was noted in 14 (28.6%) of the 49 patients, which was in line with the results of a previous study. 16 Mostly transient, TKI withdrawal syndrome consists of musculoskeletal or joint pain. Thirteen (26.5%) of 49 patients reported grade 1–2 TKI withdrawal syndrome and one (2.1%) patient reported grade 3 TKI withdrawal syndrome. The median time at which the TKI withdrawal syndrome occurred was 2 (1–4) months after TKI discontinuation. The median duration of withdrawal syndrome was 3 (2–6) months.
Factors associated with MR
Because the influence of second-generation TKIs on TFR has not been fully elucidated, 14 the factors influencing the MR-free survival rate at 24 months in the 40 patients who received imatinib alone were analyzed. Age, sex, Sokal score, and time to achieve MR 4.5 were not significantly correlated with the MR, while the duration of MR 4.5 and cumulative duration of imatinib therapy were (Table 2). A 54-month duration of MR 4.5 was the optimal value for dichotomization of this variable according to the receiver operating characteristic (ROC) curve (Figure 2), and the proportions of patients who experienced MR after drug discontinuation were 6/24 and 11/14 (p = 0.01) in the MR 4.5 duration ⩾54 months and <54 months groups, respectively. For two patients, the MR 4.5 duration before drug discontinuation was greater than 24 months, but the specific duration of MR 4.5 could not be determined, thus, these patients were not included in the analysis. There was a significant difference in the MR-free survival rate between the two groups (p < 0.001; Figure 3). Eighty-five months of imatinib therapy was identified as the boundary value according to the ROC curve (Figure 4), and the proportions of patients who underwent MR after drug discontinuation were 6/22 and 11/18 (p = 0.031) in the groups with imatinib therapy durations of ⩾85 and <85 months, respectively. There was a significant difference in the MR-free survival rates between the two groups (p = 0.03; Figure 5). Multivariate Cox regression analysis indicated that after adjusting for the duration of imatinib, a MR 4.5 duration of ⩾54 months was the only independent factor affecting MR-free survival [p = 0.004; odds ratio (OR) 5.34 (95% CI 4.21–6.47)].
Clinical factors associated with the molecular relapse in 40 patients with chronic myeloid leukemia receiving imatinib only before TKI discontinuation.

Data are ratio, n/n, or median (interquartile range).
TKI, tyrosine kinase inhibitor.
Data were missing for six patients.
MR 4.5: BCR-ABL1IS ≤0.0032%.

ROC analysis of duration of MR 4.5 before imatinib discontinuation and rate of molecular relapse after imatinib discontinuation.

Effect of MR 4.5 duration before imatinib cessation on molecular relapse after imatinib discontinuation in 40 chronic myeloid leukemia patients receiving imatinib only. Two patients were not included in this analysis due to data of MR 4.5 not available.
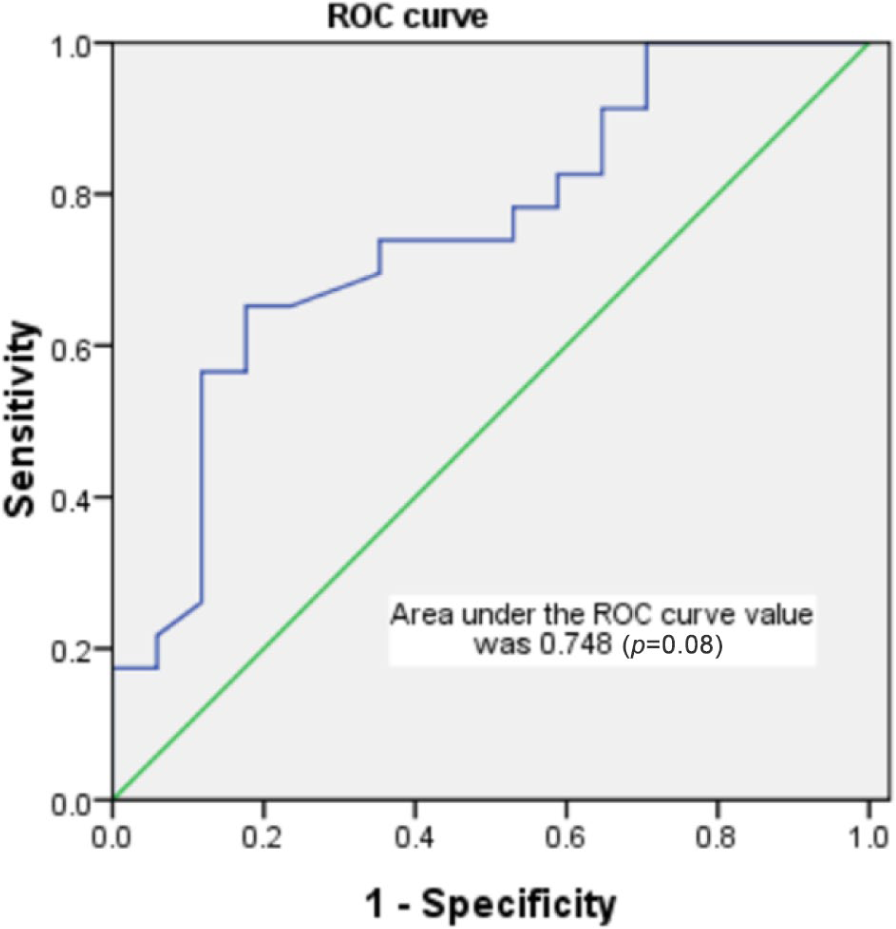
ROC analysis of duration of imatinib therapy and rate of molecular relapse after imatinib discontinuation.

Effect of duration of receiving imatinib on molecular relapse after imatinib discontinuation in 40 patients with chronic myeloid leukemia receiving imatinib only.
Effect of post-TKI discontinuation IFN-α treatment on MR-free survival
A total of 36 patients became BCR-ALB1 positive again after TKI discontinuation, and six patients only once tested BCR-ABL1-positive for low levels (not losing the MMR) and did not receive any interventions. Eight patients lost the MMR (MR) at the first or second positive BCR-ABL1 test and restarted TKI treatment. At the early stage of follow-up, four patients with low-level fluctuating BCR-ABL1 ultimately lost the MMR. Subsequently, patients with low BCR-ABL1 levels who tested positive more than twice received IFN-α treatment. Finally, 12 patients agreed to receive IFN-α treatment and six did not. Three of the 12 patients receiving IFN-α ultimately lost the MMR, and eight out of 10 patients (4 plus 6) not receiving IFN-α ultimately lost the MMR (25% versus 80%, p = 0.03). Among these 22 patients, we analyzed the factors affecting MR after TKI discontinuation.
Twelve of the 22 patients received IFN-α therapy. The median duration of IFN-α therapy was 9 (2, 18) months, and three patients eventually experienced MR. Three patients stopped IFN-α therapy because they tested BCR-ABL1-negative after IFN-α treatment and could not endure frequent injections of IFN-α. These three patients discontinued IFN-α treatment after 2, 2.5, and 10 months, and at their last follow-up visit, they maintained their MMR for 9, 8, and 11 months after discontinuation of IFN-α, respectively. Among the 12 patients receiving IFN-α, one discontinued IFN-α 2 months after MMR loss, and two patients discontinued IFN-α after 2 and 2.5 months because the BCR-ABL1 test was negative and they were reluctant to continue to receive frequent injections. The median duration of IFN-α therapy of the remaining nine patients was 10 (6, 18) months. The two patients with CML-AP received IFN-α intervention after TKI discontinuation. Of the 10 patients who did not receive IFN-α treatment, eight eventually experienced MR. The MR-free survival rate was significantly different between the two groups (70.7% versus 15.0%, p = 0.002; Figure 6). Further analysis showed that of the 22 patients with low-level BCR-ABL1 positivity, 11 had a MR at 24 months after TKI discontinuation. In terms of their clinical characteristics, the patients who did and did not relapse were not significantly different in terms of age, sex, Sokal score, time to achieve MR 4.5, duration of MR 4.5 before TKI discontinuation, and type of TKI, but they were significantly different in terms of whether they received IFN-α therapy and the duration of TKI therapy before discontinuation (Table 3). Multivariate Cox regression analysis showed that after adjusting for the duration of TKI therapy before discontinuation, post-TKI discontinuation IFN-α treatment was the only independent factor affecting MR-free survival [p = 0.011; OR 6.10 (95% CI 4.71–7.48)]. Of note, two additional pregnant women experienced MR 4 months after TKI discontinuation. Because they were pregnant, they did not resume TKI therapy; instead, they were administered IFN-α treatment. A MMR was obtained after 2 months of IFN-α treatment and maintained for 12 months in both patients. Therefore, 14 patients received IFN-α treatment after TKI discontinuation.

Survival without molecular relapse after TKI discontinuation in 22 patients with chronic myeloid leukemia with low level BCR-ABL1 (0.0032% <BCR-ABLIS ⩽0.1%).
Clinical factors associated with the molecular relapse in 22 chronic myeloid leukemia patients with low level BCR-ABL1 gene positive after TKI discontinuation.

Data are ratio, n/n, or median (interquartile range).
Molecular relapse: BCR-ABL1IS >0.1%. MR 4.5: BCR-ABL1IS ⩽0.0032%. Low level BCR-ABL1 gene positive: 0.0032% <BCR-ABL1IS ⩽0.1%.
Data were missing for six patients.
IFN-α, interferon-α; TKI, tyrosine kinase inhibitor.
Most patients tolerated the IFN-α treatment well. Three (21%) of the 14 patients receiving IFN-α treatment developed transient fever after the initial injection of IFN-α and reported grade 1–2 myalgia during the treatment period, and one (7%) patient reported grade 1 arthralgia.
Discussion
To explore the effect of IFN-α treatment on MR-free survival after TKI discontinuation, we retrospectively analyzed the data of 49 patients with CML who discontinued TKI treatment. In this single-center retrospective study, we found that the MR-free survival rate 12 months after TKI discontinuation in patients with CML who had achieved MR 4.5 was 67% (95% CI 53.6%, 80.3%). Stopping TKI therapy appears to be feasible when predefined criteria are applied and standardized molecular monitoring is performed. Moreover, MR 4.5 duration before imatinib discontinuation was much longer in the non-MR than in the MR group (71.2 versus 50 months, p = 0.01) Moreover, in patients with low-level BCR-ABL1 positivity, those treated with IFN-α had a much higher MR-free survival rate at 24 months than those not treated with IFN-α (70.7% versus 15.0%, p = 0.002). The results of our study showed that MR 4.5 duration before TKI discontinuation upon receiving IFN-α treatment after TKI discontinuation affected MR-free survival.
In this study, the results showed that an MR 4.5 duration ⩾54 months before imatinib discontinuation and an imatinib therapy duration ⩾85 months were associated with prolonged MR-free survival, while the patient’s age, sex, Sokal score, and time to achieve MR 4.5 were not associated with MR. The STIM study found that patients with low Sokal scores who received imatinib for more than 50 months were more likely to maintain a MMR after TKI discontinuation. 15 The STOM-IM study similarly found that the duration of MR 4.5 was related to maintenance of a MMR after TKI discontinuation, but the Sokal score had no significant impact in that study. 25 The EURO-SKI study also found that patients who took imatinib for >5.1 years and had duration of deep remission of more than 3.1 years had a lower rate of MR. 16 Above all, the results of our study were similar to those of previous clinical studies in that a longer duration of MR 4.5 and TKI therapy results in a higher rate of TFR. The difference was mainly in the cut-off value of duration of MR 4.5 and TKI therapy, which may be due to the different selection of sensitivity and specificity.
TKI discontinuation is feasible and safe for patients with longer durations of deep molecular response and TKI therapy. In our study, the MR-free survival rate at 12 months was 67%, and the patients experiencing MR almost regained MMR after resuming TKI treatment within 3 months. In the largest EURO-SKI study, no loss of complete hematological remission or progression due to accelerated-phase CML or blast phase were reported, and the median time to regaining the MMR after resuming TKI therapy was 2.8 months. 16
Once the feasibility of TKI discontinuation is established, it will be important to determine whether there is a way to decrease the MR rate after TKI discontinuation. Currently, there are no adequate interventions to achieve this aim. However, since IFN-α treatment leads to long-term remission in patients with CML,26,27 we compared MR-free survival with and without the administration of IFN-α after TKI discontinuation in patients with low levels of BCR-ABL1 and a maintained MMR. Our study showed that the MR-free survival rate at 18 months in patients treated with IFN-α was much higher than that in patients not treated with IFN-α (70.7% versus 15.0%, p = 0.002). We also assessed the effect of IFN-α treatment in a small group of patients. Specifically, in our study, three patients treated with IFN-α discontinued treatment after 2, 2.5, and 10 months, and did not lose their MMR for 9, 8, and 11 months after the discontinuation of IFN-α treatment, respectively. Therefore, even a short course of IFN-α may help decrease the MR rate. In the German CML study IV, 20 patients with CML received an IFN-α/imatinib induction treatment followed by temporary IFN-α maintenance therapy, and 13 patients remained MR-free for a median of 7.9 years after imatinib discontinuation. Of the 10 patients who discontinued IFN-α, nine remained MR-free. 22 The interim analysis of TIGER study demonstrated that nilotinib plus pegylated IFN-α induction and peg-IFN maintenance therapy increased the rates of MR 4.5 and may translate into higher rates of TFR. 23 Above all, IFN treatment could help the MR-free survival rate after TKI discontinuation. However, in these two clinical studies, IFN was used in combination with TKI as an induction and maintenance therapy. In contrast to these studies, IFN was used in post-TKI discontinuation patients with CML who were positive for BCR-ABL1 at low levels (0.0032% < BCR-ABLIS ⩽0.1%) in the present study, and this increased the MR-free survival rate significantly. This design avoids the use of IFN in all patients with CML and decreases the occurrence of adverse events.
The use of IFN-α in the treatment of CML may be due to the ability of IFN to inhibit tumor cell proliferation and induce tumor cell apoptosis. 28 In addition, Mahon et al. noted that some patients who achieved a cytogenetic response to IFN-α treatment were able to discontinue the treatment without disease relapse. 26 Furthermore, these patients had significantly increased CD8+ T cells and natural killer cell counts, indicating that IFN may increase the number of killer immune cells. Moreover, IFN-α-treated patients present a unique pattern of clonal γδ T cells, which is not observed in TKI-treated patients with CML, 27 which may suggest that IFN works by increasing the number of killer immune cells. In a 2010 study, Burchert et al. showed that IFN-α therapy is associated with an increase in the expression of leukemia-associated antigen proteinase 3 and the induction of proteinase-3 specific CTLs, 21 which may provide further rationale for how IFN-α may help reduce the rate of MR.
To the best of our knowledge, this study is the first to use IFN in post-TKI discontinuation in patients with CML according to the BCR-ABL1 levels in the monitoring test after TKI discontinuation, and this treatment with IFN-α is associated with a decreased rate of MR in patients with CML with low levels of BCR-ABL1. However, the sample size in this study was small, and the follow-up time was short. The appropriate duration of INF treatment is currently unknown. Furthermore, this study was not randomized. Therefore, we plan to design a prospective randomized trial to test our hypothesis in future.
Conclusion
The results of this study indicate that TKI discontinuation is feasible in Chinese patients with CML after sustained MR 4.5. Moreover, after the discontinuation of TKI therapy in these patients, early intervention with IFN-α treatment while patients are in a state of low-level BCR-ABL1 positivity may help maintain the MMR and prolong survival without TKI treatment.
Footnotes
Acknowledgements
The authors thank the faculty members who assisted with this study.
Author contributions
Jun Kong, Hao Jiang, and Xiao-jun Huang designed the study, analyzed and interpreted the data, and wrote the manuscript. Ya-zhen Qin performed qPCR experiments.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Capital Characteristic Clinic Project Foundation (Z181100001718126), National Science and Technology Major Project (2017ZX09304021), and grant 2016YFE0202800 from the National Key R&D Program.
