Abstract
Background:
Droplet digital polymerase chain reaction (ddPCR) is an exact method of measurement.
Objectives:
We conducted this study to identify the prognostic factors for successful treatment-free remission in patients with chronic-phase chronic myeloid leukemia who discontinued tyrosine kinase inhibitors (TKIs). We also aimed to validate ddPCR for predicting molecular relapse.
Design:
This is a prospective, multicenter study.
Methods:
We enrolled patients treated with TKIs for at least 3 years with a confirmed sustained deep molecular response (DMR) for at least 1 year. TKI was re-administered in patients who experienced the loss of major molecular response (MMR).
Results:
A total of 66 patients from five institutions in South Korea were enrolled. During a median follow-up period of 16.5 months, 29/66 (43.9%) patients experienced molecular relapse; the probability of molecular relapse-free survival (RFS) at 6 or 12 months after TKI discontinuation was 65.6% or 57.8%, respectively, with most molecular relapses occurring within the first 7 months. All patients who lost MMR were re-treated with TKI, and all re-achieved MMR at a median of 2.8 months. E14a2 transcript type (p = 0.005) and longer DMR duration (⩾48 months) prior to TKI discontinuation (p = 0.002) were associated with prolonged molecular RFS and with sustained DMR. Patients with both e13a2 transcript type and detectable BCR::ABL1 (⩾MR5.0) by ddPCR at the time of TKI discontinuation showed shorter duration of molecular RFS (p = 0.015).
Conclusion:
Our data suggest that transcript type and BCR::ABL1 transcript levels on ddPCR should be taken into consideration when deciding whether to discontinue TKI therapy.
Keywords
Introduction
Due to the development of tyrosine kinase inhibitors (TKIs), chronic myeloid leukemia (CML) patients can expect near-normal life expectancy. 1 However, ongoing lifelong treatment with TKIs can significantly reduce quality of life, lead to off-target effects, such as life-threatening cardiovascular or liver toxicity, and represent a significant financial burden.2,3 Therefore, successful treatment-free remission (TFR) has become a new therapeutic goal, and large, prospective data has shown encouraging results. In the Stop Imatinib (STIM) study, molecular recurrence-free survival (RFS) at 60 months was 38% [95% confidence interval (CI): 29–47%] with no disease progression among patients in whom BCR::ABL1 transcripts were not detected for >2 years. 4 Multiple subsequent clinical trials have demonstrated the possibility of successful TFR in patients with a deep molecular response (DMR) sustained for several years, and approximately 40–60% of patients could successfully discontinue TKIs, regardless of the type of TKI. 5 Recent European LeukemiaNet guideline recommended that chronic-phase CML patients with typical BCR::ABL1 transcripts who have been treated > 5 years and sustained a DMR for more than 2 years could attempt the discontinuation of TKIs. 6 However, the prognostic factors for selecting appropriate candidates for TKI cessation remain controversial. The results of the large clinical trials identified longer duration of TKI treatment or DMR as important prognostic factors for successful discontinuation of TKI treatment.7,8 Some studies also suggest that a low Sokal risk score or interferon pre-treatment is associated with a higher TFR rate.7–9 Experimental studies emphasized immunologic profiles, including increases in natural killer cells and decreases in regulatory T cells, as having significant roles in successful TFR.10,11
Droplet digital polymerase chain reaction (ddPCR) is a sensitive method for quantifying BCR::ABL1 transcript levels. ddPCR has been used for measuring gene expression and detecting mutations and minimal residual disease in various hematologic malignancies. 12 Limited data showed that CML patients with undetectable or lower levels of BCR::ABL1 by ddPCR at the time of TKI discontinuation achieved successful TFR.13–15 However, further evidence is needed to prove the role of ddPCR in identifying suitable candidates for TFR.
Here, we prospectively examined the prognostic factors for successful TFR in patients with Philadelphia chromosome-positive, chronic-phase CML who had been treated with TKIs. We also validated the role of ddPCR in predicting successful TFR.
Methods
Patient characteristics
This prospective, multicenter trial enrolled patients with Philadelphia chromosome-positive, chronic-phase CML in five Korean centers from August 2019 to August 2020. The eligible patients were 18 years or older and had received any TKI as first, second, or higher lines of treatment. Patients were treated with standard doses of TKIs for at least 3 years and had confirmed, sustained DMR for at least 1 year as measured by reverse transcription-quantitative polymerase chain reaction (RT-qPCR). To be included, the patients needed at least three RT-qPCR results confirming a sustained DMR during the 1 year before study entry. Other inclusion criteria were Eastern Cooperative Oncology Group performance status ⩾2 and absence of severe dysfunction in any primary organ.
We excluded patients with a history of allogeneic stem-cell transplant, prior attempt(s) to discontinue TKI treatment, reduced TKI dose due to abnormal laboratory findings, participation in other clinical trials, or diagnosis indicating active concomitant malignancy.
Study procedures
Molecular response was assessed by RT-qPCR using peripheral blood samples obtained at the time of TKI discontinuation, 1 month later, and then every 2 months up to 1 year. ddPCR was performed at the time of TKI discontinuation, 1 month, and 5 months later after TKI discontinuation. RT-qPCR assays with 4.5-log sensitivity and ddPCR assays with 5.0-log sensitivity were performed in the central laboratory (LAS, Gimpo, Korea). The loss of major molecular response (MMR), which indicates molecular recurrence, was determined after two consecutive assessments within 1 month. TKI was re-started in cases of MMR loss. Patients were re-administered the same TKI that had been taken at the time of TKI discontinuation. For these patients, molecular response was assessed every 2 months by RT-qPCR until MMR was re-established.
ddPCR procedure
RNA (1 µg) was reverse-transcribed using QXDx iScript Advanced Reverse Transcriptase in QXDx BCR::ABL1 %IS Kit (Bio-Rad Pleasanton, CA, USA). After reverse transcription, 16 µL of cDNA was transferred to a ddPCR plate containing ddPCR Master Mix. The samples were then split into two wells per sample (data to be merged during analysis), and the plate was sealed for droplet generation. After droplet generation, ddPCR amplification was performed using the BCR::ABL1 ddPCR Amplification Thermal Cycling Protocol. After the polymerase chain reaction (PCR) was complete, the PCR plate was transferred to a QX200 Droplet Reader (Bio-Rad). For reading, the QuantaSoft Software 1.7.4 (Bio-Rad) was set to RED (rare events detection) for experiments, FAM signal for BCR::ABL1 copies (target gene) and HEX signal for ABL1 copies (internal control gene
PCR for BCR::ABL1 transcript type
To determine the BCR::ABL1 transcript type, we used the method described by Goh et al. 16 For the detection of e14a2 or e13a2, primers specific for e11 of the BCR gene and a3 of the ABL1 gene were chosen. PCR was performed according to the manufacturer’s instructions. The PCR reaction consisted of 1 µL cDNA, 1X PCR buffer, 1.5 mM MgCl2, 0.2 mM dNTP, 2.5 U Taq DNA polymerase, and primers in a final volume of 25 µL. PCR was performed in duplicate under the following cycling conditions: pre-denaturation at 94°C for 3 min, 30 cycles at 94°C for 30 s, 60°C for 30 s, and 72°C for 30 s, followed by a final extension at 72°C for 5 min. Amplified products were visualized by ethidium bromide-stained agarose gel electrophoresis. We used the following amounts of cDNA for each technique.

ddPCR, droplet digital polymerase chain reaction; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; PCR, polymerase chain reaction.
Outcomes
The primary endpoint was to identify molecular RFS, which is the length of time from the date of TKI discontinuation to the date at which loss of MMR, death, or censoring was recorded. The secondary endpoint was to find prognostic factors affecting molecular recurrence and evaluate safety and disease progression after TKI discontinuation. We also assessed the time to re-achieve MMR after restarting TKI in patients who lost MMR.
Statistical analysis
Previously, STIM and Korean KID trials reported that CML patients who discontinued TKI achieved approximately 40% and 60% molecular RFS at 12 months, respectively.7,17 Thus, we assumed that the null proportion was 0.4 according to the STIM trial and that the alternative proportion was 0.6 according to the KID study. According to the Cancer Research and Biostatistics SWOG statistical tools using these values (null proportion = 0.4, alternative proportion = 0.6, α error = 5%, and β error = 10%), 53 patients were required to analyze TFR.
The paired T-test was used to compare the BCR::ABL1 values measured by RT-qPCR and ddPCR, and the Kaplan-Meier method was used to analyze molecular RFS. The duration of MMR before stopping TKI was defined as the period of BCR::ABL1IS ≤0.1%, based on the RT-qPCR results before TKI discontinuation. The duration of DMR before stopping TKI was defined as the period of BCR::ABL1IS ≤0.01% (molecular response at 4.0-log reduction, MR4.0) before TKI discontinuation. The duration of sustained DMR was defined as the date of TKI discontinuation to the date of the loss of MR4.0. Median values for age, TKI treatment duration, the duration of MMR before stopping TKI, and the duration of DMR before stopping TKI were used as cut-off values for each variable. Statistical analyses were performed using IBM SPSS, version 23.0 (IBM, Armonk, NY, USA). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 18
Results
Patient characteristics
Sixty-six patients from five institutions in South Korea were finally enrolled in this study between August 2019 and August 2020 (Figure 1). The median follow-up period was 16.5 months (range, 9.8–22.5 months), and the median age of the patients was 49 years. The number of patients who were treated with first-line TKI and second or higher lines at the time of TKI discontinuation were 44/66 (66.7%) and 20/66 (30.3%), respectively. The TKI that most patients had received at the time of study enrollment was nilotinib (57.6%). All patients had been treated with standard doses of TKIs (imatinib 400 mg once daily, dasatinib 100 mg once daily, nilotinib 300 mg twice daily, or radotinib 300 mg twice daily). The patients’ characteristics are shown in Table 1.
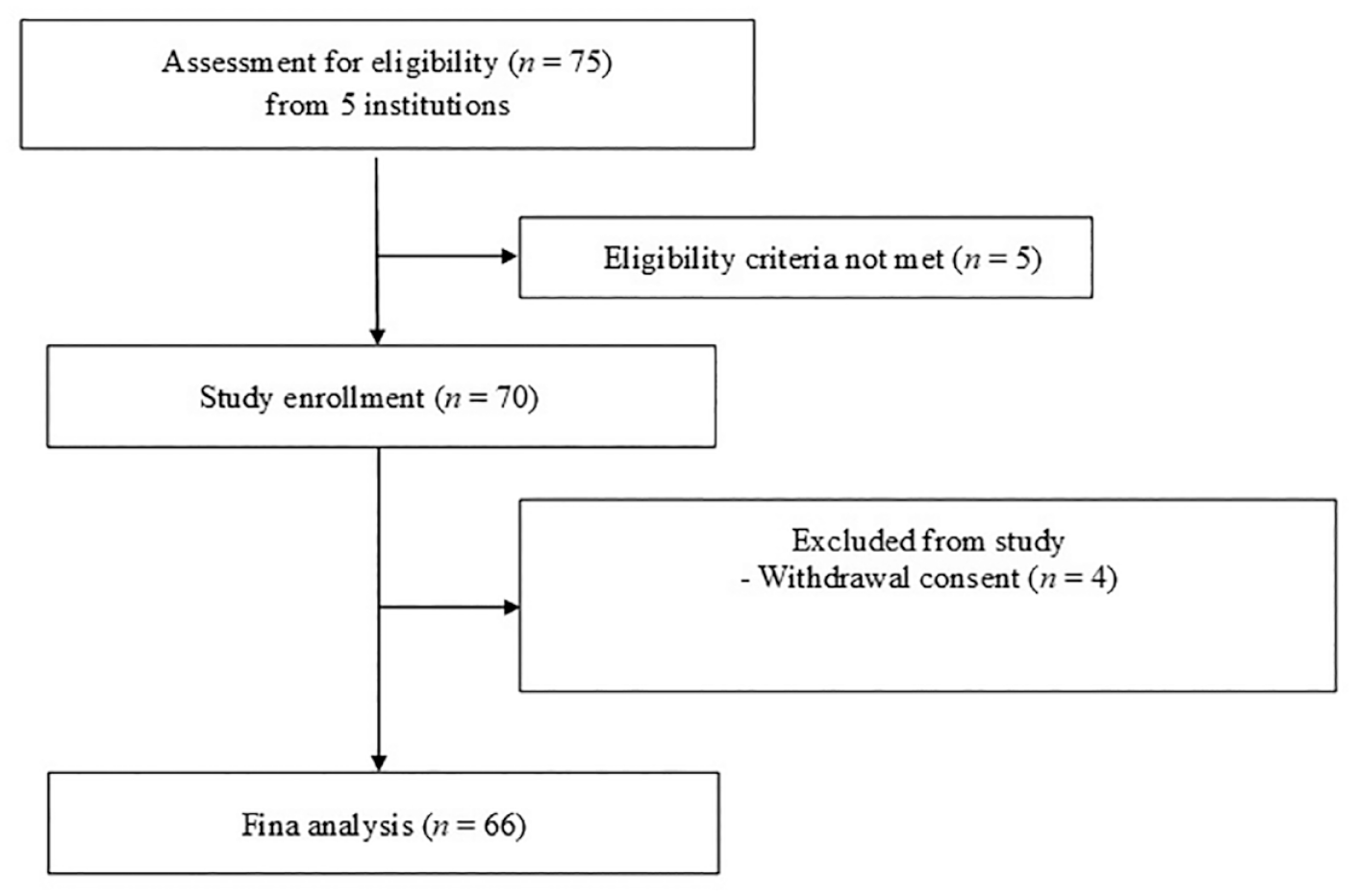
CONSORT flow diagram.
Baseline characteristics of the patients.

ECOG, Eastern Cooperative Oncology Group performance status; TKI, tyrosine kinase inhibitor.
Outcomes of TKI discontinuation
During the follow-up period, 29/66 (43.9%) patients experienced molecular relapse, and 35/66 (53%) patients maintained MMR (two patients were not evaluated). The median duration of molecular relapse after TKI discontinuation was 3.4 months (range, 1.0–21.3 months), mostly within the first 7 months (86.2%). One patient experienced late molecular relapse, occurring 21.3 months after radotinib was stopped. Thus, the probability of molecular RFS was 65.6% at 6 months and 57.8% at 12 months [Figure 2(a)]. The probability of sustained DMR after TKI discontinuation was 46.0% at 6 months and 33.3% at 12 months [Figure 2(b)].
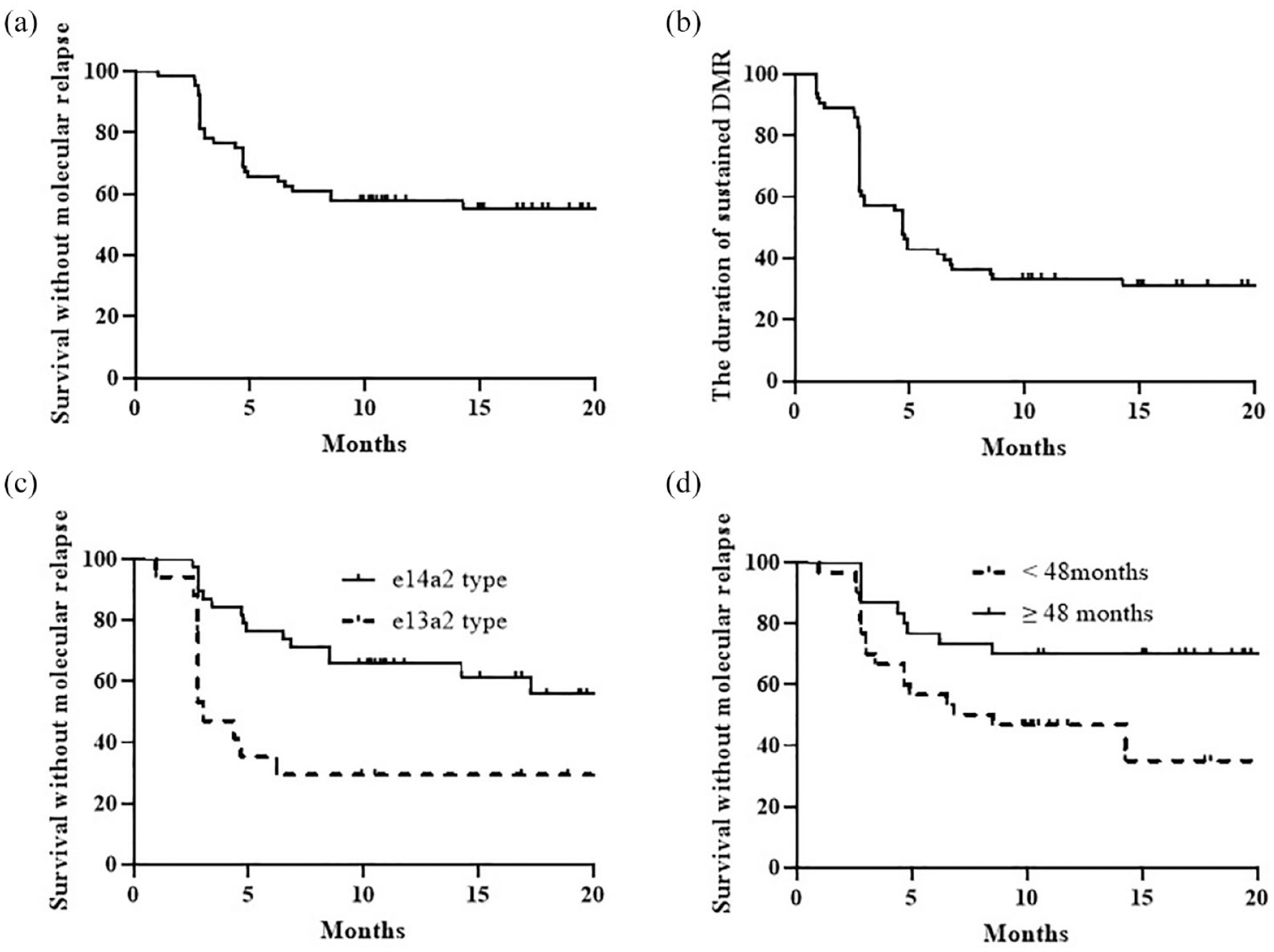
The probability of molecular relapse (a) and sustained DMR after TKI discontinuation (b). The probability of molecular relapse according to BCR::ABL1 transcript type (c) and duration of DMR (d).
The prognostic indicators for prolonged molecular RFS in univariate analysis were decreased initial white blood cell count (p = 0.042), e14a2 transcript type [p = 0.002; Figure 2(c)], longer MMR duration before stopping TKI (p = 0.012), undetectable transcripts on ddPCR [<MR5.0 (molecular response at 5.0-log reduction)] at the time of TKI discontinuation (p = 0.041), and longer DMR duration before stopping TKI [p = 0.012, Figure 2(d); Table 2]. In multivariate analysis, e14a2 transcript type and longer DMR duration before TKI discontinuation (⩾48 months) were prognostic factors for prolonged molecular RFS [hazard ratio (HR) = 3.569, 95% CI: 1.462–8.717, p = 0.005 for transcript type; HR = 2.867, 95% CI: 1.177–6.983, p = 0.002 for DMR duration; Table 2]. The favorable factors in multivariate analysis for sustained DMR after TKI discontinuation were also e14a2 type and longer DMR duration before TKI discontinuation (HR = 4.164, 95% CI: 2.057–8.429, p < 0.001 for transcript type; HR = 3.091, 95% CI: 1.530–6.240, p = 0.002 for DMR duration; Table 3).
Prognostic factors for molecular RFS after TKI discontinuation.
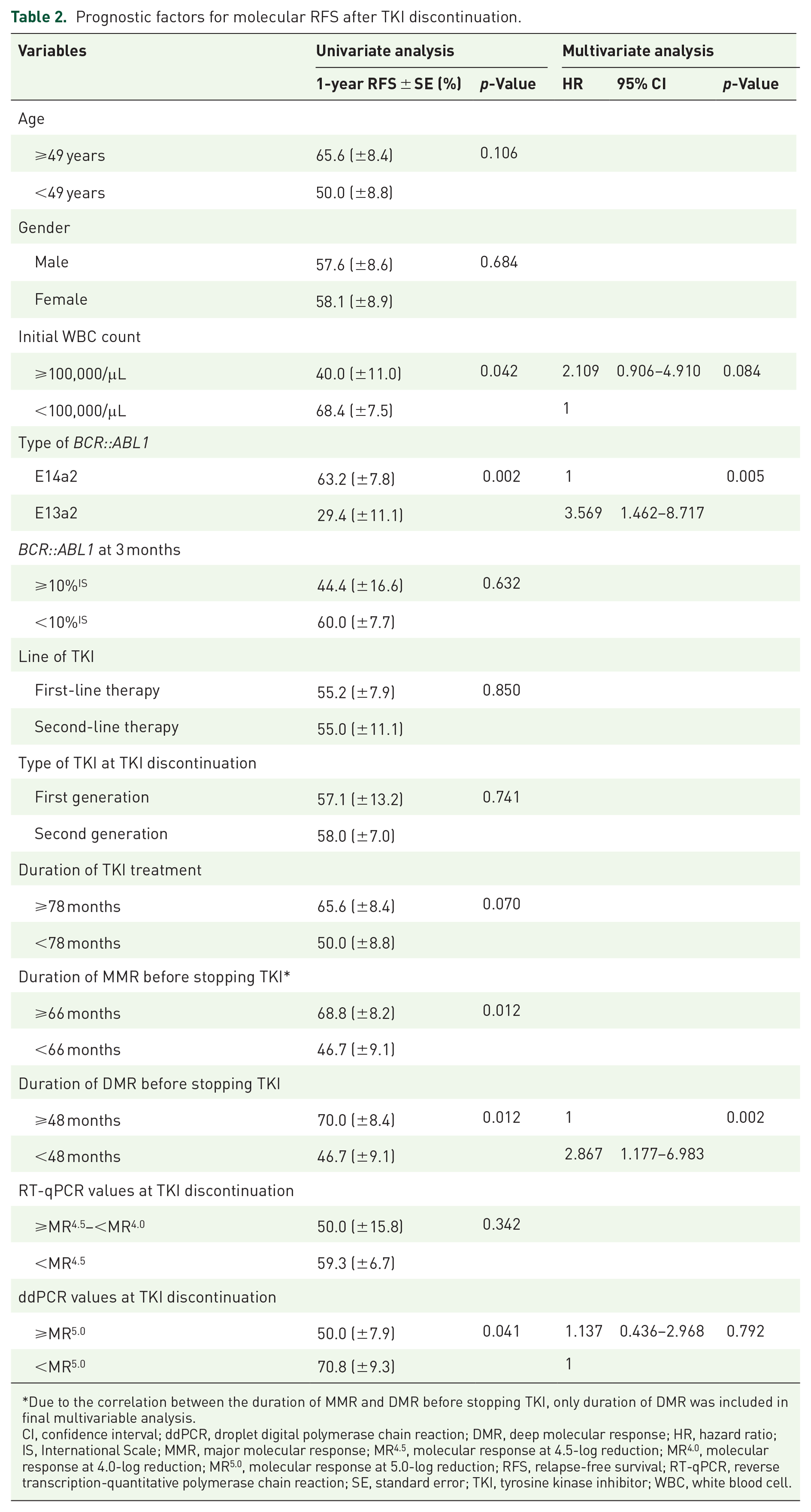
Due to the correlation between the duration of MMR and DMR before stopping TKI, only duration of DMR was included in final multivariable analysis.
CI, confidence interval; ddPCR, droplet digital polymerase chain reaction; DMR, deep molecular response; HR, hazard ratio; IS, International Scale; MMR, major molecular response; MR4.5, molecular response at 4.5-log reduction; MR4.0, molecular response at 4.0-log reduction; MR5.0, molecular response at 5.0-log reduction; RFS, relapse-free survival; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; SE, standard error; TKI, tyrosine kinase inhibitor; WBC, white blood cell.
Prognostic factors for a sustained DMR after TKI discontinuation.
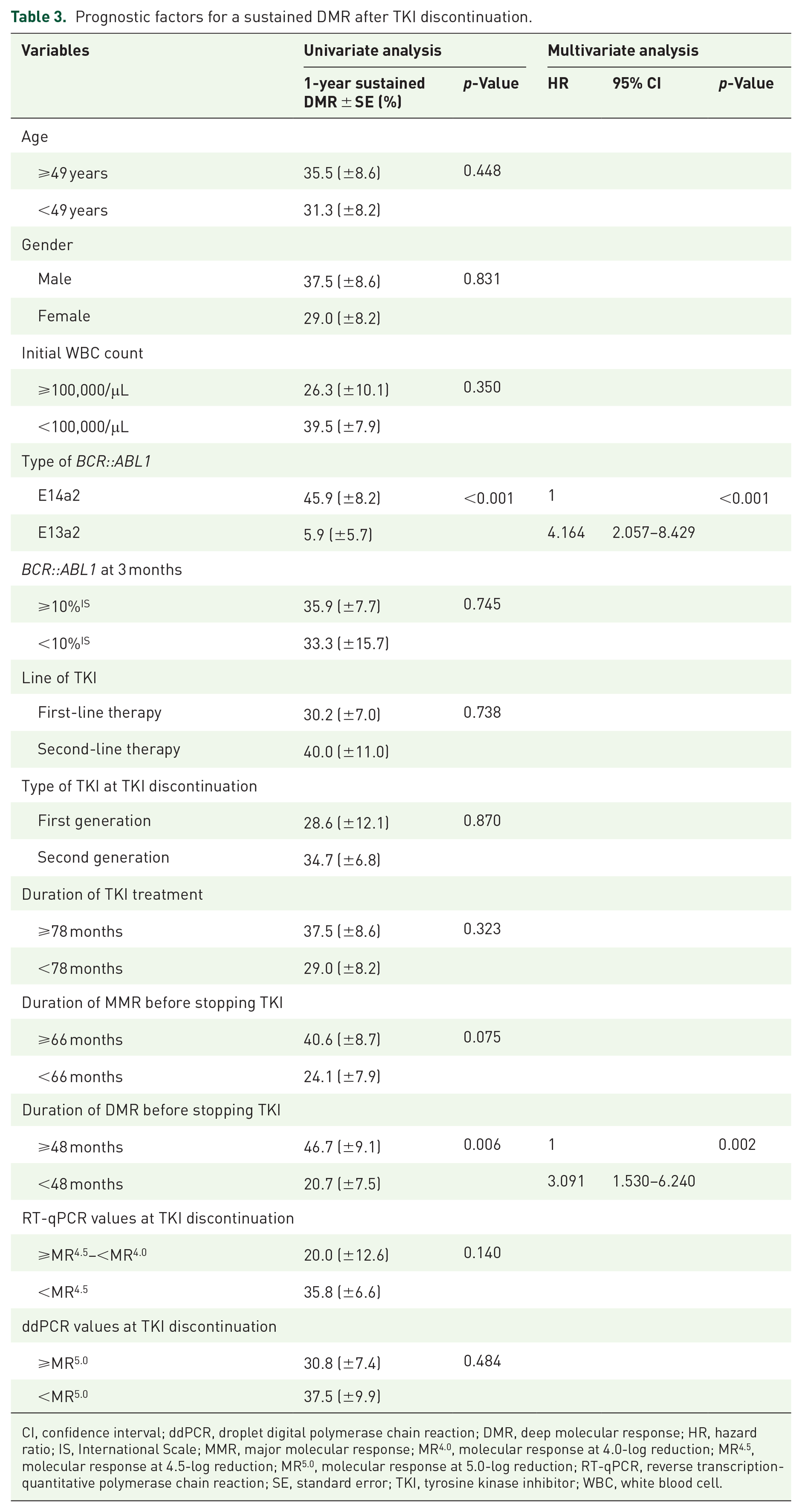
CI, confidence interval; ddPCR, droplet digital polymerase chain reaction; DMR, deep molecular response; HR, hazard ratio; IS, International Scale; MMR, major molecular response; MR4.0, molecular response at 4.0-log reduction; MR4.5, molecular response at 4.5-log reduction; MR5.0, molecular response at 5.0-log reduction; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; SE, standard error; TKI, tyrosine kinase inhibitor; WBC, white blood cell.
TKI treatment was re-initiated in patients who lost MMR and MMR was re-achieved in all of these patients within a median time after TKI re-administration of 2.8 months (range, 1.0–7.1 months). No patients progressed to the accelerated or blast phase.
Outcomes of ddPCR
Undetectable BCR::ABL1 transcript (<MR5.0) on ddPCR at the time of TKI discontinuation was a favorable prognostic factor for molecular RFS in univariate analysis (1-year molecular RFS: 70.8% for undetectable BCR::ABL1 versus 50.0% for detectable BCR::ABL1, p = 0.041); however, this association was not observed in multivariate analysis (Table 2). When considering the combination of transcript type and detection of BCR::ABL1 by ddPCR, patients who were both e13a2 type and had detectable transcripts on ddPCR (⩾MR5.0) at the time of TKI discontinuation showed significantly worse probability for molecular RFS than other groups [1-year molecular RFS: 21.4% for e13a2 type and detectable transcripts on ddPCR versus 62.5% for e14a2 type and undetectable transcript on ddPCR versus 64.0% for others; Table 4 and Figure 3(a)]. In multivariate analysis, both e13a2 type and detectable transcript on ddPCR (⩾MR5.0) were statistically significant factors for shorter molecular RFS (HR = 0.331, 95% CI: 0.119–0.924, p = 0.035 for e14a2 and undetectable transcript on ddPCR; HR = 0.275, 95% CI: 0.108 – 0.699, p = 0.007 for others). Shorter DMR duration before TKI discontinuation (<48 months) was also a significant factor for shorter molecular RFS (HR = 2.478, 95% CI: 1.058–5.803, p = 0.037) (Table 4). Similarly, multivariate analysis showed that the factors associated with a shorter DMR after TKI discontinuation were both e13a2 transcript type and undetectable BCR::ABL1 by ddPCR (HR = 0.272, 95% CI: 0.112–0.659, p = 0.004 for e14a2 and undetectable transcript on ddPCR; HR = 0.230, 95% CI: 0.102–0.518, p < 0.001 for others). In addition, a shorter DMR duration before TKI discontinuation (<48 months) was associated with a shorter DMR (HR = 2.675, 95% CI: 1.330–5.381, p = 0.006) [Figure 3(b)].
Effect of BCR::ABL1 type and ddPCR value at the time of TKI discontinuation on molecular RFS after TKI discontinuation.

Due to the correlation between the duration of MMR and DMR before stopping TKI, only duration of DMR was included in final multivariable analysis.
CI, confidence interval; ddPCR, droplet digital polymerase chain reaction; DMR, deep molecular response; HR, hazard ratio; IS, International Scale; MMR, major molecular response; MR 4 , molecular response at 4.0-log reduction; MR4.5, molecular response at 4.5-log reduction; MR5.0, molecular response at 5.0-log reduction; RFS, relapse-free survival; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; SE, standard error; TKI, tyrosine kinase inhibitor; WBC, white blood cell.

The probability of molecular relapse (a) and sustained DMR (b) according to transcript type and detectability by ddPCR at time of TKI discontinuation.
In subgroup analysis according to the various factors, undetectable BCR::ABL1 on ddPCR (<MR5.0) was an important factor for longer molecular RFS in patients with longer TKI treatment duration (⩾78 months) or long MMR duration before stopping TKI (⩾66 months) (Supplemental Table 1).
When comparing BCR::ABL1 values detected by ddPCR and RT-qPCR, there was no significant difference at the time of stopping TKI and 1 month after stopping TKI, but there was a difference 5 months after TKI discontinuation (Figure 4; Supplemental Table 2).
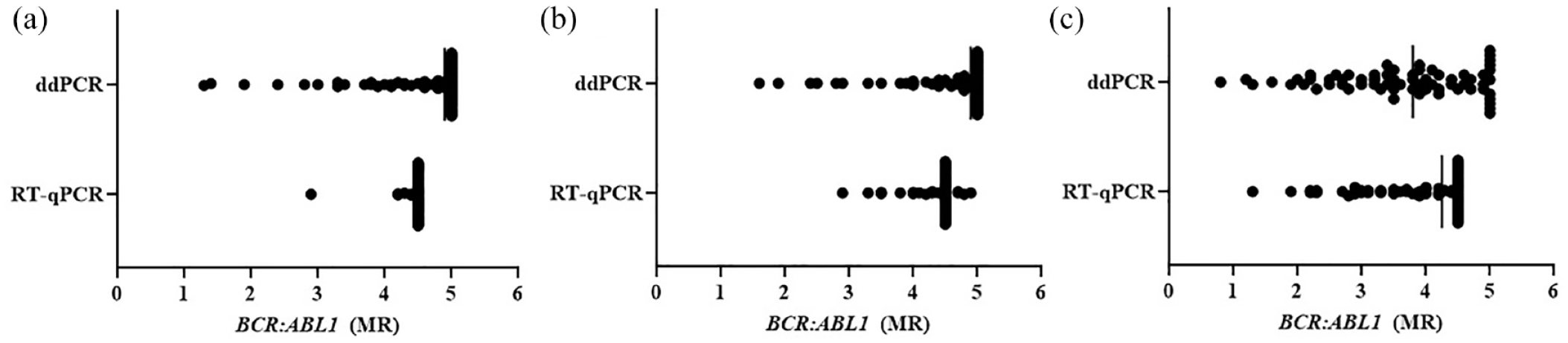
Comparison between RT-qPCR and ddPCR. (a) At the time of TKI discontinuation (mean MR4.5 for RT-qPCR versus MR4.5 for ddPCR, p = 0.931), (b) 1 month after TKI discontinuation (mean MR4.4 for RT-qPCR versus MR4.5 for ddPCR, p = 0.246), and (c) 5 months after TKI discontinuation (mean MR3.8 for RT-qPCR versus MR3.6 for ddPCR, p = 0.017).
Discussion
We conducted this study in hopes of establishing the prognosis of patients who discontinue TKI and the associated factors for successful TFR. We found that (1) the probability of the continuous discontinuation of TKIs was 57.8% at 12 months, (2) the prognostic factors for successful TKI cessation were e14a2 type and longer DMR duration (⩾48 months), and (3) ddPCR could be a sensitive method for predicting successful TFR. Especially, patients with e13a2 type and detectable BCR::ABL1 on ddPCR (⩾MR5.0) at the time of stopping TKI showed poor TFR outcomes.
The present study showed that 65.6% of the patients sustained MMR at 6 months and 57.8% at 12 months after TKI cessation, with most molecular relapses occurring within the first 7 months. These results confirm the results of previous studies, which showed that 40–60% of chronic-phase CML patients can successfully discontinue TKIs. 5 No patient experienced disease progression, and all patients re-achieved MMR within a median of 2.8 months after restarting TKI. Although our criteria for TKI discontinuation were less stringent than criteria used in previous large clinical trials, our findings suggest that patients who receive first- or second-generation TKIs for at least 3 years and demonstrate DMR for at least 1 year could be considered as candidates for discontinuation of TKI treatment.7,19
Prognostic factors for successful TFR were e14a2 type and longer DMR duration before stopping TKI (⩾48 months). This was not related to patient age, the line of treatment, TKI treatment duration, or the type of TKI, which were taken into consideration as clinical parameters (Table 2). The favorable prognosis of the e14a2 transcript type was reported in several earlier studies.20–25 Previous imatinib data showed that patients expressing the e14a2 type achieved higher and more rapid molecular responses than e13a2-expressing patients.20,24,25 Additional reports support that the e14a2 transcript correlates positively with sustained DMR and potential to achieve TFR.21,24 Retrospective studies also found that patients with e14a2 transcripts had higher rates of TFR success compared to those with e13a2 variants.22,23 Little is known about functional relevance according to transcript type. One possible hypothesis is that the increased tyrosine kinase activity of the e13a2 transcript may have affected the results. 20 Immunological control of minimal residual disease due to different immunogenicity is also proposed for the clinical benefit of the e14a2 variant.26,27 Another possibility is that RT-qPCR artifacts may influence clinical outcomes. Recently, a study comparing amplification according to the transcript type found that shorter e13a2 transcripts may be amplified more efficiently than e14a2 by RT-qPCR, possibly leading to an underestimation of the quantified values of the e14a2 transcript.28–31 However, some reports showed better or similar molecular responses in patients expressing the e13a2 transcript than in patients expressing the e14a2 transcript. Thus, more data are needed to understand the significance and role of BCR::ABL1 transcript type.32,33
Based on our experience and accumulated data, we can tentatively conclude that a longer DMR duration (⩾48 months) before TKI discontinuation is a predictive indicator for successful TFR. In the EURO-SKI trial, patients with longer DMR durations with a cut-off of 3.1 years before TKI cessation had a higher probability of TFR (p = 0.0029). 8 A retrospective United States trial suggested that the relapse rate for patients with MR4.5 >2 years in duration was 32%, whereas for those with MR4.5 for <2 years, the relapse rate was 82%. 34 Similarly, Takahashi et al. 35 reported that DMR duration >24 months was a relevant factor for sustained DMR. 35 Furthermore, the TRAD trial revealed that patients with MR4.0 for more than 7.7 years had a 6-month RFS rate of 89.5%, whereas the rate for the comparison group was 36.3% (p = 0.003). 36 Our results were comparable to those reported in previous studies, even though a statistically significant relationship between the duration of TKI treatment and successful TFR was not demonstrated in our study. Other studies also suggested that the duration of TKI treatment was a significant factor for TFR, and the duration of DMR could be a factor dependent on the total treatment period.8,34,36 However, the EURO-SKI trial placed greater importance on duration of DMR than on total treatment duration. 8 Based on this evidence, a durable, deep response is a significantly relevant indicator for successful TFR. In the present study, we used the median DMR duration cut-off value of 48 months. However, the appropriate cut-off value is expected to be different for different patient cohorts and further research is needed to understand this variation more precisely.
We performed ddPCR with a sensitivity of MR5.0, and it proved to be a useful method for detecting minimal residual disease that could not be detected by RT-qPCR. Due to the increased sensitivity of ddPCR, BCR::ABL1 was detected in numerous samples by ddPCR but not by RT-qPCR in this study (Supplemental Table 2). In this study, approximately 6% more cDNA was used in ddPCR compared to RT-qPCR. This difference is not expected to have a significant effect on sensitivity. The ddPCR reaction volume was divided into 20,000 droplets, and each droplet became the actual final PCR reaction. Such a nanoscale environment for each droplet will have a relatively higher sensitivity to detect extremely low copy numbers of the target BCR::ABL1 molecule compared to normal qPCR, with a bulky PCR reaction volume. Some previous studies supported the increased sensitivity of ddPCR. Kim et al. 37 also showed that ddPCR is more sensitive than RT-qPCR. Similarly, Bernardi et al. 14 suggested that RT-qPCR could not discriminate patients with a higher risk of MR loss after TKI discontinuation, whereas ddPCR could recognize stable DMR. However, Scott et al. 38 suggested that the results of ddPCR and RT-qPCR were comparable with no statistically significant difference. Therefore, further studies are needed to confirm ddPCR as a more sensitive tool compared to conventional RT-qPCR.
Because we demonstrated a correlation between detectable BCR::ABL1 by ddPCR at the time of stopping TKI and failure of TFR by univariate analysis, but not multivariate analysis due to the small cohort (Table 2), we suggest a potential role for ddPCR in selecting appropriate candidates for TFR. More specifically, patients expressing e13a2 transcript type and detectable transcript on ddPCR (⩾MR5.0) at TKI discontinuation showed a significantly higher probability of molecular relapse than other patients. Therefore, patients who express the e13a2 type and detectable BCR::ABL1 on ddPCR (⩾MR5.0) may be poorer candidates for TKI discontinuation. Similarly, the ISAV and KID trials showed that patients with positive BCR::ABL1 values on ddPCR at the time of imatinib discontinuation were more likely to experience molecular recurrence.13,17 Other groups suggested different cut-off values for BCR::ABL1 on ddPCR that might predict successful TFR. An Italian group proposed a cut-off of <0.468 BCR::ABL1 copies/µL, and a French group proposed <0.0023%IS at TKI discontinuation for predicting significantly lower probability of molecular relapse.14,15 A method to standardize the quantitative measurement of BCR::ABL1 by ddPCR has not been widely established. Therefore, different proposed cut-off values for cohorts may depend on technical differences and pre-analytic steps. 15 Recently, Fava et al. 39 suggested that results obtained by different ddPCR platforms may be aligned. In the future, rigorous standardization will be needed before ddPCR is widely used as a prognostic tool for various hematologic diseases.
Limitations
This study had several limitations. First, the sample size was small despite being a multi-institution study. Second, Sokal and EUTOS risk scores were not analyzed, because of difficulties in obtaining consistent spleen size measurements in a multicenter study. Third, some clinical data was lost or omitted when patients were transferred to a different hospital during treatment. Fourth, we did not measure the BCR::ABL1 level using ddPCR at the time of loss of MMR or DMR. Previous studies found that ddPCR is a more stable method than RT-qPCR to evaluate molecular response.14,40 Therefore, monitoring of BCR::ABL1 by ddPCR may detect more precisely molecular relapse after TKI cessation. Nevertheless, this study contributes significantly to our understanding of prognostic factors for successful TFR, especially in Asian populations.
Conclusion
In conclusion, this study demonstrates that more than half of CML patients could successfully discontinue TKI. E14a2 transcript type and longer DMR duration were predictive indicators for successful TFR, while e13a2 transcript type and detectable BCR::ABL1 on ddPCR are predictors of poor outcome after TKI discontinuation, emphasizing the need for careful consideration when deciding whether to discontinue TKI therapy. Therefore, in the future, ddPCR is expected to become a useful tool for predicting prognosis for CML patients in the era of TKI discontinuation.
Supplemental Material
sj-docx-1-tah-10.1177_20406207231205637 – Supplemental material for Effect of BCR::ABL1 transcript type and droplet digital polymerase chain reaction on successful treatment-free remission in chronic myeloid leukemia patients who discontinued tyrosine kinase inhibitor
Supplemental material, sj-docx-1-tah-10.1177_20406207231205637 for Effect of BCR::ABL1 transcript type and droplet digital polymerase chain reaction on successful treatment-free remission in chronic myeloid leukemia patients who discontinued tyrosine kinase inhibitor by Hyunkyung Park, Hyeong-Joon Kim, Sang-Kyun Sohn, Yoonsuk Baik, Dongho Kim, Sung-Yeoun Lee, Jee Hyun Kong, Hawk Kim, Dong-Yeop Shin, Jae-Sook Ahn, Jinny Park, Seonyang Park and Inho Kim in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
