Abstract
Background:
Thrombocytopenia is a common hematological manifestation in people living with human immunodeficiency virus (HIV) or acquired immune deficiency syndrome (AIDS; PLWHA). Data on the prognostic relationship and associated factors of thrombocytopenia and HIV infection in China are limited.
Objectives:
We assessed the prevalence of thrombocytopenia, its association with prognosis, and analyzed the associated risk factors among demographic characteristics, comorbidities, hematological and bone marrow indicators.
Design:
We collected patients identified as PLWHA in Zhongnan Hospital. The patients were divided into two groups: the thrombocytopenia group and the non-thrombocytopenia group. We analyzed and compared demographic characteristics, comorbidities, peripheral blood cells, lymphocyte subpopulations, infection indicators, bone marrow cytology, and bone marrow morphology of the two groups. Then we analyzed the risk factors for thrombocytopenia and the effect of platelet (PLT) values on the prognosis of patients.
Methods:
Demographic characteristics and laboratory results were obtained from medical records. In contrast to other studies, we included bone marrow cytology and morphology in this study. Data were analyzed with multivariate logistic regression analysis. The Kaplan–Meier method was used to plot 60-month survival curves for the severe, mild, and non-thrombocytopenia groups. The value p < 0.05 was taken as statistically significant.
Results:
Among 618 identified PLWHA, 510 (82.5%) were male. Overall, thrombocytopenia was found in 37.7% [95% confidence interval (CI): 33.9–41.5%]. Multivariable logistic regression analysis showed that age ⩾40 years [adjusted odds ratio (AOR) 1.869, 95% CI: 1.052–3.320], combined with hepatitis B (AOR 2.004, 95% CI: 1.049–3.826), high procalcitonin (PCT) count (AOR 1.038, 95% CI: 1.000–1.078) were risk factors of thrombocytopenia in PLWHA. An increased percentage of thrombocytogenic megakaryocytes was a protective factor, with an AOR 0.949 (95% CI: 0.930–0.967). Kaplan–Meier survival curve analysis showed that the prognosis was worse in the severe than in the mild (p = 0.002) and non-thrombocytopenia groups (p = 0.008).
Conclusion:
We discovered a general high pervasiveness of thrombocytopenia in PLWHA in China. Age ⩾40 years, combined with hepatitis B virus infection, high PCT, and decreased percentage of thrombocytogenic megakaryocytes indicated a higher risk for developing thrombocytopenia. A PLT count ⩽50 × 109/liter led to a worse prognosis. Therefore, early diagnosis and treatment of thrombocytopenia in these patients are useful.
Background
Human immunodeficiency virus (HIV) infection causes acquired immune deficiency syndrome (AIDS). It has a poor prognosis and high mortality rate. HIV infection not only compromises the immune system, but also disrupts normal hematopoiesis, leading to frequent manifestations of pancytopenia (anemia, thrombocytopenia, and neutropenia).1–3 Thrombocytopenia is a frequent hematological manifestation in people living with HIV or AIDS (PLWHA). The prevalence of thrombocytopenia in PLWHA is 4.1–40%, and increases with disease progression.4–6 A previous cross-sectional study in Canada showed a prevalence of thrombocytopenia of 26% in PLWHA. 7 A long-term study in Denmark found a prevalence of thrombocytopenia of 5.5% in PLWHA. 8 According to a recent report, 64.6% of PLWHA without antiretroviral treatment (ART) demonstrated thrombocytopenia. 9 In a previous large cross-sectional study of newly recognized PLWHA in China, thrombocytopenia was present in 15.6%. 10
Hematological abnormalities are common in patients with PLWHA. HIV-associated hemocytopenia may result from different mechanisms, such as defects in bone marrow production, increased peripheral loss, or destruction of blood cells. 11 Thrombocytopenia is a common blood system disease in PLWHA. The mechanism is estimated to be associated with accelerated peripheral platelet (PLT) destruction and decreased PLT production from impaired megakaryocytes. 2 However, there is no clear research on the specific mechanism of its occurrence.
Objectives
With the increasing number of PLWHA and the increasing level of medical standards, the current prevalence of thrombocytopenia in PLWHA in China has not been evaluated. In addition, previous studies on this topic did not consider bone marrow cytology or bone marrow morphology. Therefore, we analyzed a cohort of PLWHA in China, to assess the prevalence of thrombocytopenia, its association with prognosis, and analyzed the associated risk factors among demographic characteristics, comorbidities, hematological and bone marrow indicators.
Design and methods
A total of 783 identified PLWHA who met the diagnostic criteria of the Chinese Medical Association’s ‘AIDS Diagnosis and Treatment Guidelines (2018 Edition)’, attending in Zhongnan Hospital from January 2016 to October 2020. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 12
Only patients with routine blood information available, who had a bone marrow aspiration, are ⩾18 years old, and met the diagnostic criteria of AIDS were considered for participation in this trial. Exclusion standards included patients who were <18 years old, who lacked routine blood information and outcome information and who had not had a bone marrow aspiration. Finally, 618 identified patients were enrolled. According to PLT count, the patients were divided into two groups: 233 patients in the thrombocytopenia group (PLT <125 × 109/liter, 37.7%) and 385 patients in the non-thrombocytopenia group (PLT ⩾125 × 109, 62.3%). The demographic and clinical data of PLWHA were collected. Besides demographic characteristics, we collected data on comorbidities, peripheral blood cells, lymphocyte subpopulations, infection indicators, bone marrow cytology, and bone marrow morphology. To investigate the effect of PLT values on the prognosis of PLWHA, the Kaplan–Meier method was used to plot survival curves for the severe, mild, and non-thrombocytopenia groups. We collected demographic, clinical, and outcome information on 4 April 2022. The flow chart is shown in Figure 1.

Study flow diagram.
Definition
Conditions were defined based on the following parameters: Anemia: male, hemoglobin (HGB) count <130 g/liter, female, HGB count <115 g/liter. Thrombocytopenia: PLT count <125 × 109/liter. A PLT count between 125 × 109/liter and 50 × 109/liter was considered to reflect mild thrombocytopenia, whereas a PLT count ⩽50 × 109/liter was considered to reflect severe thrombocytopenia. Leukopenia: white blood cell (WBC) count <3.5 × 109/liter. The definition is set according to the Reference Interval of Blood Cell Analysis issued by the National Health Commission of the people’s Republic of China in 2013.
Demographic characteristics and clinical data collection
To determine the demographic, clinical, and laboratory characteristics at the time of thrombocytopenia detection – defined for the purposes of this study as the onset of thrombocytopenia in incident cases or the first clinic visit in prevalent cases – medical records were retrospectively reviewed. Variables included age, sex, comorbidities, peripheral blood cells, lymphocyte subpopulation, infection indicators, bone marrow cytology, and bone marrow morphology.
Bone marrow aspiration and biopsy
Bone marrow aspiration and trephine biopsies were carried out on either the anterior or poster superior iliac spine. Disposable bone marrow trephine biopsy needles were used. Aspiration was performed prior to the trephine biopsy procedure. The samples from a trephine biopsy were placed in formalin fixative immediately. The tissue was decalcified for 3.5 h after at least 4 h of fixation before being processed using standard histology methods. After being stained with hematoxylin–eosin, two level sections were routinely examined. Bone marrow particles from aspiration samples were ‘squashed’ and ‘streaked’ (without particles, and spread out like a peripheral blood smear), and after being stained with Wright–Giemsa stain, they were examined. Bone marrow aspiration and trephine biopsy samples were obtained within the same procedure. Each patient’s blood sample was manually prepared into blood smears, dyed with May–Grünwald–Giemsa dye, and delivered to a facility for analysis. Two hundred cells per blood smear were manually evaluated by two qualified reviewers, independently, blinded to other findings, and the average was taken for analysis.
Statistical analysis
The independent samples’ t-test was used to compare the means of continuous variables with normal distributions. Differences in non-normally distributed variables were assessed using nonparametric tests. The χ2 test was used to compare categorical variables. We utilized multivariate binary logistic regression models to account for relevant confounders. To evaluate the association between each risk factor and thrombocytopenia, odds ratios and 95% confidence intervals (CIs) were computed. We used Kaplan–Meier survival curve analysis to assess the impact of thrombocytopenia on prognosis in PLWHA. We analyzed the data using IBM SPSS version 25.0 (IBM SPSS Inc., Armonk, NY, USA). The value p < 0.05 was taken as statistically significant.
Results
Demographic characteristics and comorbidities of study participants
A total of 618 identified PLWHA were recruited into this study, of which 510 (82.5%) were male. The median age of the participants was 43 years [interquartile range (IQR) 32, 52]. The demographic characteristics and comorbidities of the study participants are shown in Table 1. The sex distribution between the thrombocytopenia and non-thrombocytopenia groups differed significantly (p = 0.004). More patients in the thrombocytopenia were 40 years or older than in the non-thrombocytopenia group (p = 0.002, Figure 2(a)). Leukopenia was almost twice as prevalent in the thrombocytopenia group than in the non-thrombocytopenia group (p < 0.001). Similarly, hepatitis B and hepatitis C virus infections were present in almost double the proportion of patients in the thrombocytopenia group as compared with the non-thrombocytopenia group (both p = 0.002). The incidence of thrombocytopenia among patients with various comorbidities was shown in Figure 2(b). Eventually, 535 of the PLWHA in our study survived, and 83 died.
Demographic characteristics and comorbidities of PLWHA.

CMV, cytomegalovirus, 174 patients missing data; EBV, Epstein–Barr virus, 257 patients missing data; PLWHA, people living with HIV/AIDS.
Values are expressed as number (%) of patients.
39 patients missing data.
18 patients missing data.
28 patients missing data.

The incidence of thrombocytopenia in patients with different (a) ages and (b) comorbidities.
Peripheral blood cells, lymphocyte subpopulation, and infection indicators of study participants
The mean red blood cell (RBC) counts were significantly lower in the thrombocytopenia than in the non-thrombocytopenia group (p < 0.001). The mean HGB concentration was significantly lower in the thrombocytopenia than in the non-thrombocytopenia group (p < 0.001). The median WBC, neutrophil (NEU), lymphocyte (LYM), and CD8+ counts were significantly lower in the thrombocytopenia group than in the non-thrombocytopenia group (all p < 0.001). The median CD4+ /CD8+ ratio was significantly higher in the thrombocytopenia than in the non-thrombocytopenia group (p = 0.016). The median PCT level was significantly higher in the thrombocytopenia than in the non-thrombocytopenia group (p < 0.001; Table 2 and Figure 3).
Comparison of peripheral blood cells, lymphocyte subpopulation, and infection indicators between the two groups.
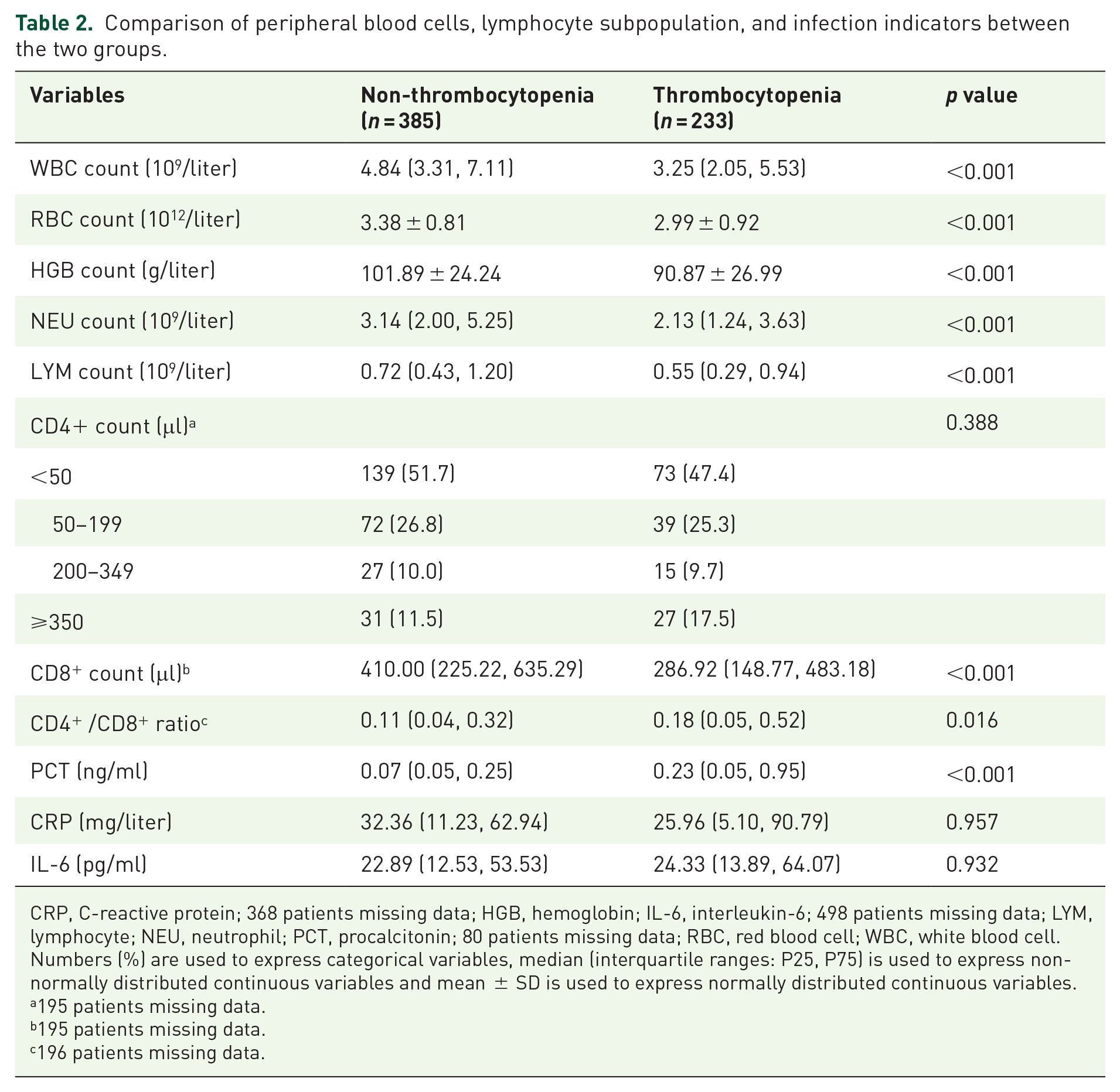
CRP, C-reactive protein; 368 patients missing data; HGB, hemoglobin; IL-6, interleukin-6; 498 patients missing data; LYM, lymphocyte; NEU, neutrophil; PCT, procalcitonin; 80 patients missing data; RBC, red blood cell; WBC, white blood cell.
Numbers (%) are used to express categorical variables, median (interquartile ranges: P25, P75) is used to express non-normally distributed continuous variables and mean ± SD is used to express normally distributed continuous variables.
195 patients missing data.
195 patients missing data.
196 patients missing data.

Comparison of (a) WBC, (b) RBC, (c) HGB, (d) NEU, (e) LYM, (f) CD8+, (g) CD4+/CD8+, (h) PCT, (i) CRP, and (j) IL-6 between the two groups.
Comparison of bone marrow morphology and bone marrow cytology
Next, we compared bone marrow morphology and bone marrow cytology among the two groups (Table 3). In terms of bone marrow morphology, most PLWHA showed increased granularity, thickening, and vacuolar degeneration. Granularity increases and thickening were more prevalent in the non-thrombocytopenia than in the thrombocytopenia group (p = 0.002). Similarly, vacuolar degeneration was significantly more prevalent in the non-thrombocytopenia than in the thrombocytopenia group (p = 0.006). Döhle bodies was found significantly more often in the non-thrombocytopenia group (p = 0.010). On the contrary, basophilic stippling was significantly more common in the thrombocytopenia than in the non-thrombocytopenia group (p < 0.001). Hemophagocytic cells was also more commonly present in the thrombocytopenia group than in the non-thrombocytopenia group (p = 0.034).
Comparison of bone marrow cytology and bone marrow morphology between the two groups.
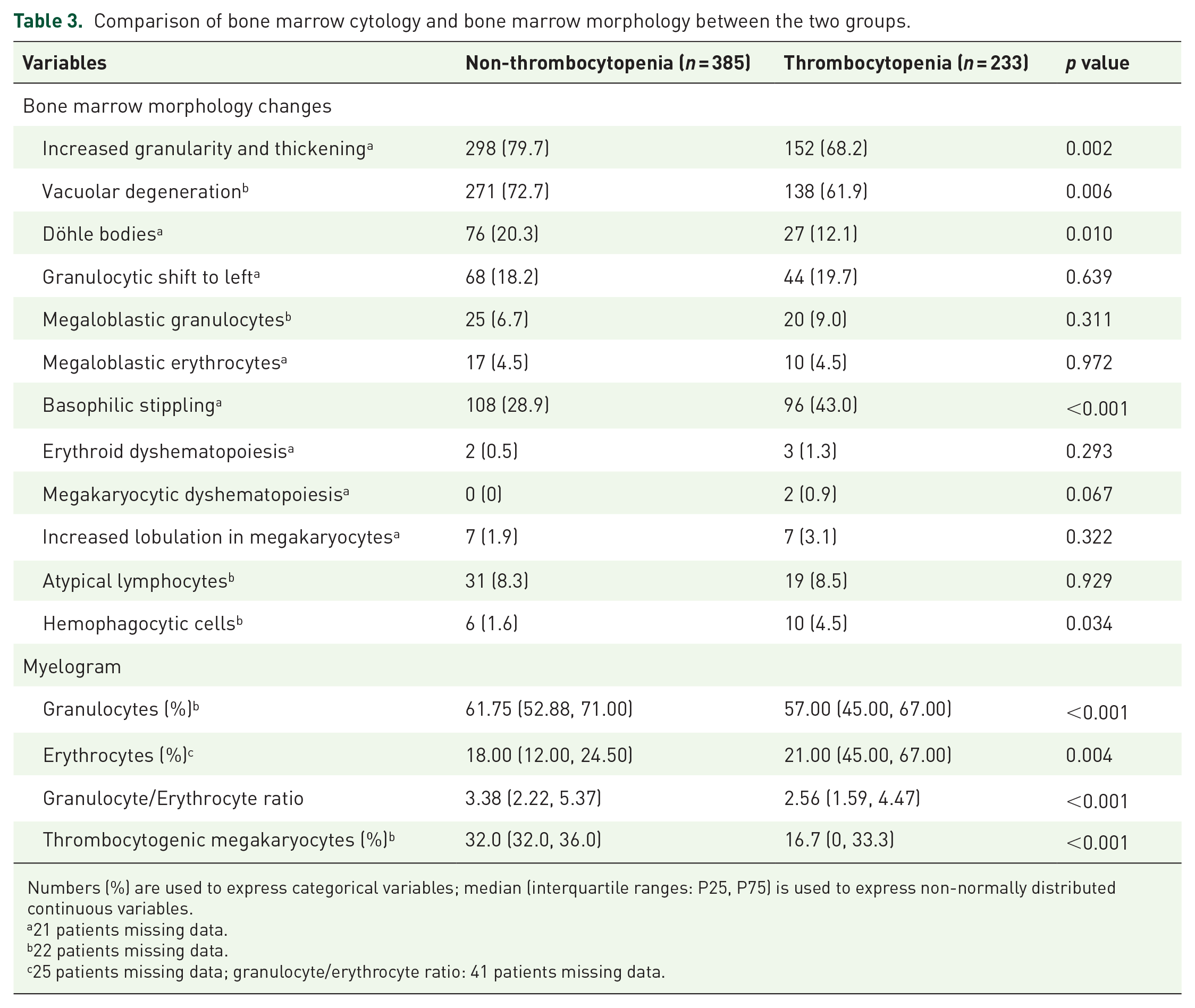
Numbers (%) are used to express categorical variables; median (interquartile ranges: P25, P75) is used to express non-normally distributed continuous variables.
21 patients missing data.
22 patients missing data.
25 patients missing data; granulocyte/erythrocyte ratio: 41 patients missing data.
The prevalence of thrombocytopenia among patients with various bone marrow morphologies is shown in Figure 4. Figure 5 shows typical morphological changes in the bone marrow of PLWHA, including vacuolar degeneration, Döhle bodies, and hemophagocytic cells.

Prevalence of thrombocytopenia in patients with different bone marrow morphology.

Typical morphological changes in bone marrow cells in people living with HIV/AIDS. (a) Vacuolar degeneration, (b) Döhle bodies, and (c) hemophagocytic cells.
In the comparison of bone marrow cytology among the two groups, the median percentage of granulocytes was higher in the non-thrombocytopenia than in the thrombocytopenia group (p < 0.001). Erythrocytes were more common in the thrombocytopenia than in the non-thrombocytopenia group (p = 0.004). The granulocyte/erythrocyte ratio was significantly higher in the non-thrombocytopenia than in the thrombocytopenia group (p < 0.001). The median percentage of thrombocytogenic megakaryocytes in the thrombocytopenia group was almost half that in the non-thrombocytopenia group (p < 0.001; Figure 6).

Comparison of bone marrow morphology between the two groups. (a) Granulocytes, (b) erythrocytes, (c) granulocyte/erythrocyte ratio, and (d) thrombocytogenic megakaryocytes.
Risk factors of thrombocytopenia in PLWHA: multivariate logistic regression analysis
We examined parameters related to thrombocytopenia among PLWHA in a multivariate logistic regression model (Table 4). Because of colinearity, WBC count and leukopenia could not be included in the same model. Replacing leukopenia for WBC count did not change the model qualitatively. Even after accounting for other model factors, there was still a significant relationship between thrombocytopenia status and hepatitis B, age, PCT, and the percentage of thrombocytogenic megakaryocytes. The adjusted odds ratio for hepatitis B was 2.004 (95% CI: 1.049–3.826). PLWHA with hepatitis B are 2.2004 times more likely to develop thrombocytopenia than those without hepatitis B (p = 0.035). The adjusted odds ratio for age was 1.869 (95% CI: 1.052–3.320). PLWHA aged ⩾40 years were 1.869 times more likely to develop thrombocytopenia than those aged <40 years (p = 0.033). The adjusted odds ratio for PCT was 1.038 (95% CI: 1.000–1.078, p = 0.049). The percentage of thrombocytogenic megakaryocytes was a protective factor, with an adjusted odds ratio of 0.949 (95% CI: 0.930–0.967, p < 0.001).
Relationship between associated variables and thrombocytopenia in Chinese PLWHA.

AOR, adjusted odds ratio; HGB, hemoglobin; LYM, lymphocyte; NEU, neutrophil; PCT, procalcitonin; PLWHA, people living with HIV/AIDS; RBC, red blood cell; WBC, white blood cell.
Effects of thrombocytopenia on the prognosis of PLWHA
Finally, we analyzed the 60-month survival rates in different groups of patients (Figure 7). Data were collected on 4 April 2022. The median follow-up time was 50 months (range 11–60). There were 73 survivors (76.0%) in the severe thrombocytopenia group, 126 survivors (92.0%) in the mild thrombocytopenia group, and 336 survivors (87.3%) in the non-thrombocytopenia group. Kaplan–Meier survival curve analysis showed that the prognosis of severe thrombocytopenia group was worse than that of the mild thrombocytopenia group (p = 0.002) and non-thrombocytopenia group (p = 0.008). The prognosis of the mild thrombocytopenia group and non-thrombocytopenia group was not statistically significantly different (p = 0.220). Further analysis was performed to identify confounding factors that might influence patient outcomes (Table 5). The results suggested that age and hepatitis B may influence the outcome of patients.
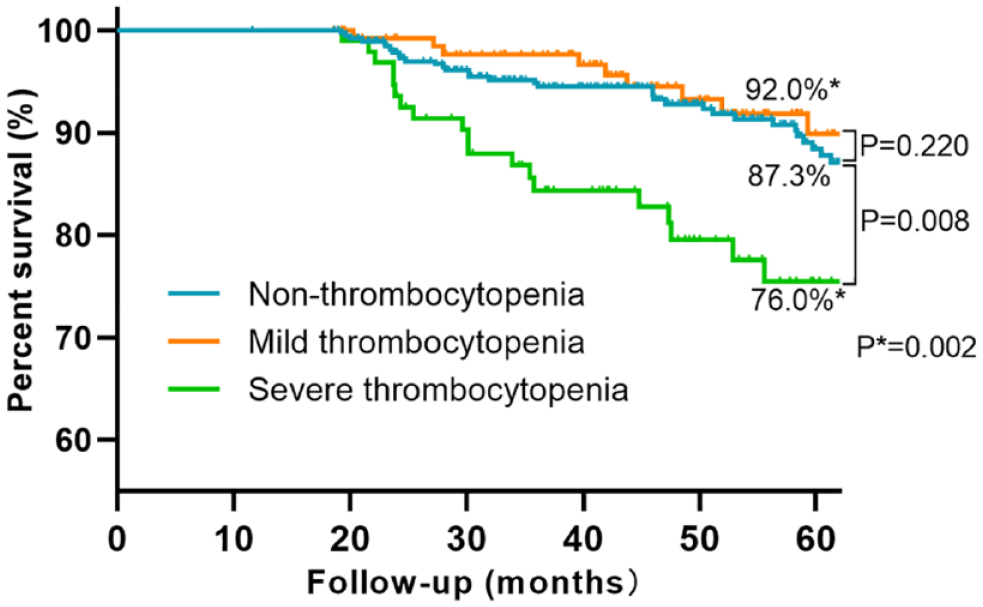
Comparison of 60-month survival of patients in the non-thrombocytopenia group, mild thrombocytopenia group, and severe thrombocytopenia group by Kaplan–Meier survival curves.
Demographic characteristics and comorbidities of outcome.

Discussion
Although PLWHA have a wide range of clinical outcomes, a series of hematological abnormalities, particularly cytopenias, are often identified as the first and most common clinical complication. HIV-related thrombocytopenia is one of these commplications.13–17 According to literature reports, 18 the prevalence of thrombocytopenia in Chinese identified PLWHA was higher than that in the overall Chinese population. In our study, there were 233 patients with thrombocytopenia, accounting for 37.7% of our total cohort. In addition, we identified age ⩾40 years, combined with hepatitis B virus infection, high PCT, and decreased percentage of thrombocytogenic megakaryocytes as risk factors for the development of thrombocytopenia. In addition, a PLT count ⩽50 × 109/liter was associated with a worse prognosis among PLWHA.
The prevalence of thrombocytopenia in different cities was different.2,6,8,18–20 The difference may be due to variation in the definition of thrombocytopenia, region, study design, and study population size, as well as differences in PLT count cutoff values across study areas.4,9,18,20–24 In addition, race and ethnicity may also influence the incidence of thrombocytopenia. 3 For PLWHA, thrombocytopenia may be the main reason for hospital admission. Over time, some patients may experience severe bleeding due to thrombocytopenia. 18 In a Canadian study, 0.6% of identified PLWHA developed severe resistant thrombocytopenic purpura. 25 Therefore, for PLWHA, it is necessary to monitor PLT counts to assess disease progression and prognosis, and to screen for and identify HIV infection in patients presenting with thrombocytopenia or its associated clinical symptoms.
In the multivariate logistic regression analysis, we concluded that hepatitis B virus infection was an independent risk factor for thrombocytopenia in PLWHA, which was consistent with previous findings. 26 A study showed that all patients with severe thrombocytopenia tested positive for antibodies to hepatitis B virus core antigen. 27 This may be because co-infection with hepatitis B virus damages the liver and reduces thrombopoietin production. 28 In addition, age ⩾40 years was a risk factor for thrombocytopenia, which was consistent with previous research results. 29 This may be explained by the higher prevalence of myelodysplasia in older patients.18,30 High PCT values are also a risk factor for thrombocytopenia. PLTs can deliver cytokines, select leukocytes, interact with bacteria and the endothelium, and contribute to microthrombus formation. 31 Thus, when an infection occurs, the PLT count decreases due to depletion. It is worth noting that 95% CI contains 1, indicating that the effect is not strong and further analysis is needed. In addition, the decreased percentage of thrombocytogenic megakaryocytes places individuals at higher risk for developing thrombocytopenia. This may be because HIV infection impairs bone marrow homeostasis and affects cell proliferation and differentiation during hematopoiesis in both direct and indirect ways (Figure 8).32,33 In our study, thrombocytopenia occurred independently of CD4 cell count. This was consistent with the conclusion of another study. 34

Risk factors of thrombocytopenia in people living with HIV/AIDS.
HIV infection increases the likelihood of thrombocytopenia, which could be due to immune destruction of peripheral blood PLTs and decreased PLT production in the bone marrow. On one hand, immune destruction of peripheral blood PLTs leads to a decrease in PLT counts. In a large study conducted in Uganda, 5% of infected individuals carried anti-PLT antibodies. 21 The etiology of thrombocytopenia may be related to the decrease in the number of PLTs caused by immune destruction of PLTs. 35 HIV infection affects the maturation of megakaryocytes. Megakaryocyte maturation is disrupted due to impaired signaling in the colony-forming unit of megakaryocyte growth. 36 It may manifest as an increased proportion of immature megakaryocytes, but may also interfere with degranulation of mature megakaryocytes and PLT release. Furthermore, megakaryocytes in bone marrow express the CD4+ receptor and co-receptor required for HIV infection. Thus, megakaryocytes internalize HIV and express the viral RNA. 37 HIV infection then leads to decreased expression of the thrombopoietin C-MPL receptor by immature megakaryocytes. 25 Megakaryocytes infected with HIV also show morphological alterations and higher apoptosis rates. 38
A PLT count ⩽50 × 109/liter in PLWHA leads to worse prognosis. Thrombocytopenia affects the efficacy of HAART, resulting in higher mortality and impaired quality of life.30,39 Thrombocytopenia is associated with adverse sequelae and is considered to be an independent predictor of mortality in PLWHA because of increased bleeding risk, which can occur in mucosal, skin, soft tissue, and intracranial sites. 21 Determining when these hematological issues manifest requires monitoring of changes in PLT numbers in PLWHA and taking the necessary clinical measures to prevent comorbidities. 40 Several studies have previously reported a higher incidence of thrombocytopenia in patients who did not receive HAART than in those who received HAART.4,21,22 Therefore, starting HAART early seems to be effective in normalizing blood cells in PLWHA. 15 In those with a PLT count ⩽50 × 109/liter, in particular, treatment should be started as soon as possible to improve prognosis.
The study had some limitations. Selection bias could have influenced the results. Nevertheless, our study has important implications for the clinical management of the PLWHA. The large sample size of this study was its primary advantage. We also included bone marrow morphology and bone marrow cytology and identified the common bone marrow morphological changes in PLWHA. At the same time, we also explored differences in the bone marrow between the thrombocytopenia group and the non-thrombocytopenia group. Finally, we identified the prognosis and some risk factors for thrombocytopenia in PLWHA, laying the groundwork for future research into the pathophysiology of HIV-associated thrombocytopenia.
Conclusion
Thrombocytopenia is a common comorbidity in PLWHA. Age ⩾40 years, comorbid hepatitis B virus infection, high PCT, and a decreased percentage of thrombocytogenic megakaryocytes increased the risk for development of thrombocytopenia. Therefore, early diagnosis and treatment of thrombocytopenia in these patients are useful.
