Abstract
Platelets, derived from a certain subpopulation of megakaryocytes, are closely related to hemostasis, coagulation, metastasis, inflammation, and cancer progression. Thrombopoiesis is a dynamic process regulated by various signaling pathways in which thrombopoietin (THPO)–MPL is dominant. Thrombopoiesis-stimulating agents could promote platelet production, showing therapeutic effects in different kinds of thrombocytopenia. Some thrombopoiesis-stimulating agents are currently used in clinical practices to treat thrombocytopenia. The others are not in clinical investigations to deal with thrombocytopenia but have potential in thrombopoiesis. Their potential values in thrombocytopenia treatment should be highly regarded. Novel drug screening models and drug repurposing research have found many new agents and yielded promising outcomes in preclinical or clinical studies. This review will briefly introduce thrombopoiesis-stimulating agents currently or potentially valuable in thrombocytopenia treatment and summarize the possible mechanisms and therapeutic effects, which may enrich the pharmacological armamentarium for the medical treatment of thrombocytopenia.
Introduction
Thrombopoiesis, the development of platelets, occurs primarily within bone marrow (BM) and lung. 1 Platelets may be derived from a certain subset of megakaryocytes (MKs) expressing genes associated with proplatelet production and platelet function. 2 Once released by high-ploidy MKs, platelets function as regulators of hemostasis, thrombosis, metastasis, inflammation, and cancer progression. 3 Several factors regulate thrombopoiesis. GATA binding protein 1 (GATA1), friend leukemia virus integration 1 (Fli-1), runt-related transcription factor 1 (RUNX1), and Rap1 GTPases activator C3G play a role in regulating megakaryopoiesis. 4 C-myb (MYB), which adjusts and balances the development of MKs and GATA-1, and Tribbles Pseudokinase 3 gene (TRIB3) are negative modulators of megakaryopoiesis. 5 Thrombopoietin (THPO) is the primary regulating cytokine in thrombopoiesis. 6 Since THPO binds to the myeloproliferative leukemia(MPL) receptor, several downstream signaling pathways consisting of mitogen-activated protein kinase (MAPK)/ extracellular signal-regulated kinases (ERK), PI3K-Akt, and Janus kinase (JAK)/STAT which participate in MK proliferation, differentiation, maturation, and functional platelet division are activated. 7
Thrombocytopenia, characterized by platelet counts less than 150 × 103/μl, is related to decreased platelet production, increased destruction, splenic sequestration, dilution, or clumping. 8 Severe thrombocytopenia may lead to low platelet counts and high bleeding risk, and cause life-threatening bleeding events. Thrombopoiesis-stimulating agents are vital in the drug treatment of thrombocytopenia. In this review, thrombopoiesis-stimulating agents are classified according to their research progress in treating thrombocytopenia. Some drugs are already in clinical trials for the treatment of thrombocytopenia, such as THPO, thrombopoietin receptor agonists (THPO-RAs), all-trans retinoic acid (ATRA), and interleukin-11 (IL-11)-based agents, which have manifested reliable therapeutic effects in different kinds of thrombocytopenia, including immune thrombocytopenia (ITP), chemotherapy-induced thrombocytopenia (CIT), inherited thrombocytopenia (IT), post-hematopoietic stem cell transplantation (HSCT) thrombocytopenia, presurgical thrombocytopenia, and thrombocytopenia related to chronic liver diseases (CLD), aplastic anemia (AA), radiation, and sepsis (Table 1). Another group of agents includes some drugs and natural products which are found to promote thrombopoiesis but are still not in clinical investigations to deal with thrombocytopenia (Table 2). Their various mechanisms of action in promoting thrombopoiesis (Figures 1 and 2) and other indications apart from thrombocytopenia will be briefly introduced to show the multifunction of these agents (Tables 3 and 4). However, their potential in thrombocytopenia treatment should be highly valued.
Thrombopoiesis-stimulating agents for clinical treatment of thrombocytopenia.

AA, aplastic anemia; CIT, chemotherapy-induced thrombocytopenia; CLD, chronic liver diseases; ITP, immune thrombocytopenia; THPO, Thrombopoietin.
Thrombopoiesis-stimulating agents with potential value of clinical use.
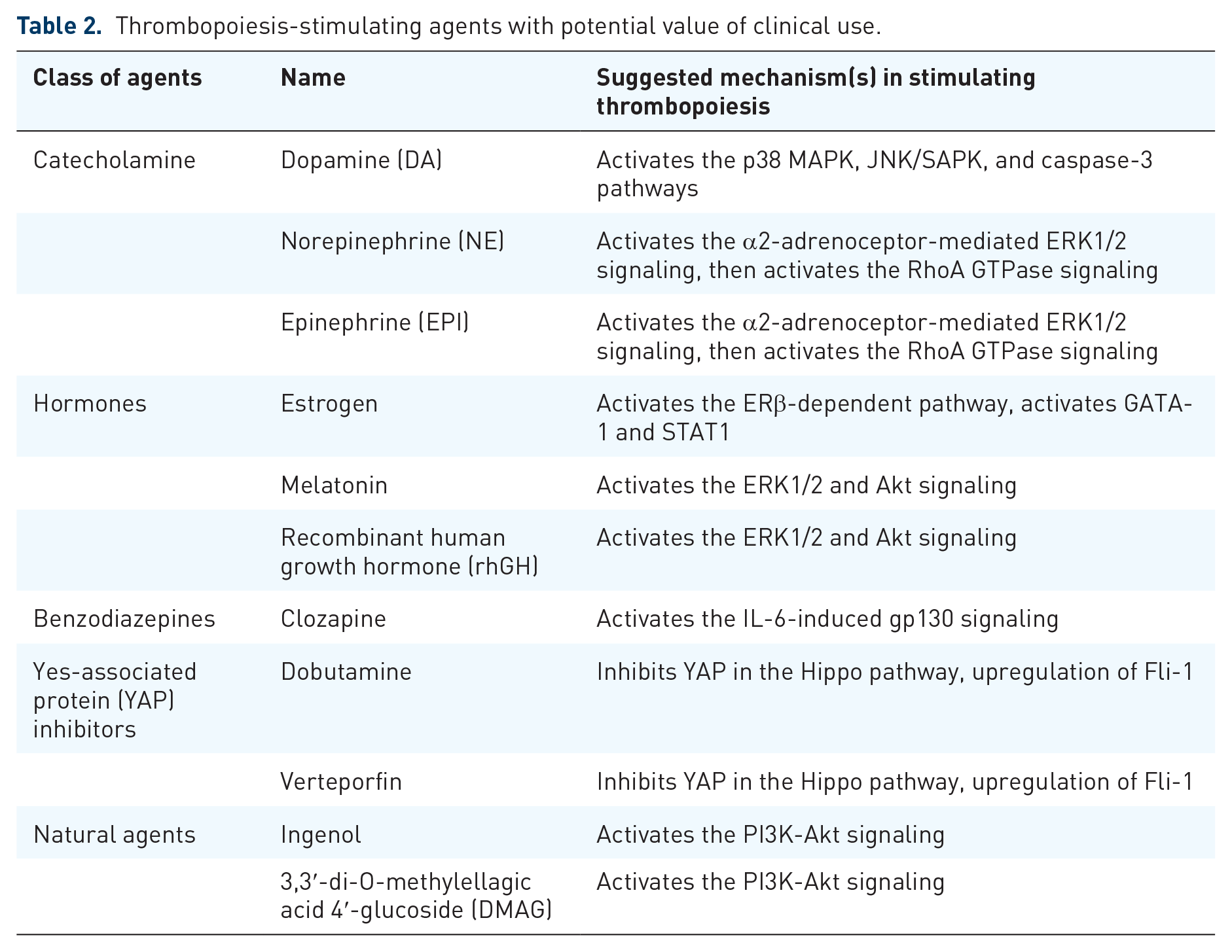

Mechanisms of thrombopoiesis-stimulating agents for clinical treatment of thrombocytopenia.

Possible mechanisms of thrombopoiesis-stimulating agents with potential value of clinical use.
Other indications and mechanisms of action of some thrombopoiesis-stimulating agents for clinical treatment of thrombocytopenia.
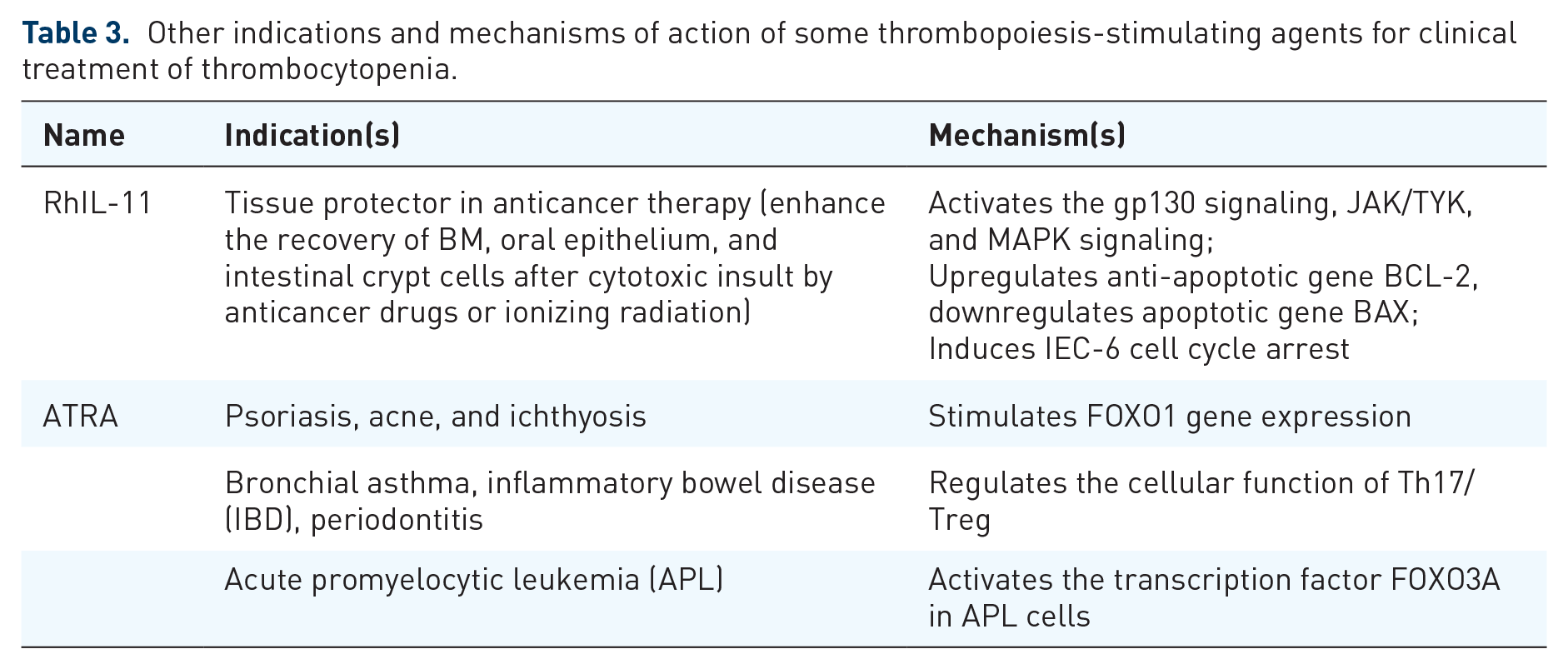
Other indications and mechanisms of action of some thrombopoiesis-stimulating agents with potential value of clinical use.

Thrombopoiesis agents for clinical treatment of thrombocytopenia
THPO and THPO-RAs
THPO
THPO plays an essential part in thrombopoiesis. Through binding to the c-MPL receptor, it can make a difference in the HSC differentiation and survival, MKs differentiation, and maturation. 9 Given the importance of THPO in promoting thrombopoiesis, PEG-recombinant human MK growth and development factor (PEG-rHuMGDF) and recombinant human THPO (rHuTHPO) are developed for thrombocytopenia treatment. Several clinical trials proved the effect of rHuTHPO in preventing and treating CIT.10,11 PEG-rHuMGDF has comparatively longer half-life, which is mainly used for CIT in patients with cancer or hematological malignancies as an alternative for rHuTHPO.12,13 However, the usage of these two recombinant THPO is both limited due to the potential risk of antibody formation to endogenous THPO. 14
THPO-RAs
THPO-RAs are drugs that bind to THPO receptors, promoting megakaryopoiesis and platelet production. 15 The increasing use of THPO-RAs, such as romiplostim, eltrombopag, and avatrombopag, shows promising effects in ITP, AA, CIT, thrombocytopenia in CLD, IT, post-HSCT thrombocytopenia, and perioperative use for reducing platelet infusion.16,17 Novel THPO-RAs with better effect and safety, such as lusutrombopag and hetrombopag, are under clinical investigation for thrombocytopenia treatment.
IL-11-based agents
IL-11 is a cytokine which belongs to interleukin-6 (IL-6) family with hematopoietic proliferative properties. It is crucial for promoting MK colony formation and plays a stimulating effect on thrombopoiesis. 41 Various IL-11-based agents, such as recombinant human IL-11 (RhIL-11), hyper-IL-11 (H11), and PEGylated IL-11, are currently or potentially used to treat thrombocytopenia.
The recombinant form of IL-11, RhIL-11, signals through the gp130 subunit of the receptor. Co-expression of the IL-11 receptor α-chain and gp130 results in high-affinity binding, initiating signal transduction of the JAK/TYK and the MAPK. 42 Due to RhIL-11’s inducing MKs maturation and differentiation, and normalizing Th1/Th2 and T-bet/GATA-3 ratios close to healthy states, 43 it shows therapeutic effect in ITP, CIT, and thrombocytopenia in sepsis patients.44–46 RhIL-11 also acts as a tissue protector in anticancer therapy because it activates the gp130 signaling, JAK/TYK, and MAPK signaling, downregulates apoptotic gene BAX, upregulates anti-apoptotic gene BCL-2, and induces IEC-6 cell cycle arrest,47,48 although the main indication of RhIL-11 is treating thrombocytopenia.
As a fusion protein comprising IL-11, H11 targets the gp130 signal-transducing subunit. 49 Compared with rhIL-11, H11 shows more effectiveness in strengthening early progenitors’ expansion and directing them to MKs, inducing the maturation of MKs and the release of platelet-like particles (PLPs). Therefore, H11 may be more productive for stimulating the MK colony formation and PLPs production than RhIL-11, that may be beneficial for thrombocytopenia treatment and an ex vivo expansion of MKs. 50 Compared with IL-11, PEGylated IL-11 not only has a longer half-life but also displays its ability of inducing longer-lasting increases in hematopoietic cells. 51 H11 and PEGylated IL-11 may have better potent effects on stimulating thrombopoiesis, but further clinical evidence of H11 and PEGylated IL-11 in thrombocytopenia treatment is needed compared with RhIL-11.
ATRA
ATRA is commonly used to treat skin diseases, inflammatory diseases, and acute promyelocytic leukemia (APL). 52 It relieves psoriasis, acne, psoriasis and ichthyosis by stimulating FOX1 gene expression,53,54 regulates the cellular function of Th17/Treg to treat bronchial asthma, inflammatory bowel disease (IBD), periodontitis,55–57 and activates the transcription factor FOXO3A in APL cells. 58 Concentration of 10−12 M ATRA could stimulate thrombopoiesis from hematopoietic progenitors in the presence of THPO or granulocyte-macrophage colony-stimulating factor (GM-CSF) plus IL-3. 59 ATRA could stimulate thrombopoiesis in MEG-01 cell by promoting MK differentiation and generation of functional PLPs. These functional PLPs could be activated by thrombin receptor-activating peptide 6 (TRAP-6) with increased externalization of P-selectin. 60 An increasing level of serum THPO was observed in APL patients with ATRA treatment, resulting in an increase in platelet counts. 61 A phase II clinical trial of 66 patients who were newly diagnosed with primary ITP reported that the proportion of patients with a sustained response (maintaining platelet counts at the level of at least 30 × 109/liter and at least twice higher than the baseline counts, without bleeding events and no rescue medication being required in 6 months) in the ATRA plus high-dose dexamethasone group (68%) was significantly higher than that of high-dose dexamethasone monotherapy group (41%), representing a novel therapeutic regimen for ITP. 62 Compared to low-dose rituximab (LD-RTX) monotherapy, ATRA plus LD-RTX increased the overall and sustained response for corticosteroid-resistant or relapse ITP patients (80% versus 59% of overall response and 61% versus 41% of sustained response). 63 These clinical investigations demonstrated ATRA might stimulate thrombopoiesis through a THPO-dependent manner, which may be beneficial in relieving thrombocytopenia, and drug combination is necessary.
Thrombopoiesis agents with potential value of clinical use
Catecholamine
Catecholamines are a group of tyrosine-derived neurotransmitters, including dopamine (DA), norepinephrine (NE), and epinephrine (EPI). DA has therapeutic effects on cardiovascular diseases (hypertrophy, myocardial ischemia, hypertension, and arrhythmia) and acute renal failure. 64 NE and EPI are commonly used to deal with severe allergic reactions, septic shock, and cardiopulmonary resuscitation.65–67 Animal experiment demonstrated that EPI and NE in response to sympathetic stimulation could promote platelet production by the activation of α2-adrenoceptor-mediated ERK1/2 signaling, which induces MKs adhesion and migration, and the ERK1/2 activation-mediated RhoA GTPase signaling could stimulate proplatelet formation (PPF), platelet release, and platelet activation. 68 However, the insufficient evidence of the effect of exogenous NE and EPI on platelet production requires further investigation to clarify the association between medicinal NE and EPI with thrombopoiesis. As the precursor of NE and EPI, DA cannot accelerate the early stages in thrombopoiesis proceedings but promote production of platelet from MKs in the final stages of thrombopoiesis, relating to oxidative stress-mediated signaling pathways which consist of p38 MAPK, JNK/SAPK, and caspase-3 pathways in human primary MKs. In addition, administration of DA in mice could elevate platelet counts. 69 These findings of catecholamines on stimulating thrombopoiesis provide new insights into drug treatment of thrombocytopenia.
Estrogen
Estrogen, one of the primary sex hormones, plays various effects in relieving perimenopausal syndrome, osteoporosis, ovarian dysfunction, functional uterine bleeding, breast cancer (post-menopause), prostate cancer, and acne.70–72 Estrogen receptors (ER), including ERα and ERβ, which express on MKs, could mediate the promoting effect of MKs differentiation, PPF, and platelet production.73,74 Estrogen-induced platelet production may be regulated by the ERβ-dependent pathway. Through detecting BM-derived MKs and Meg-01 cells of estrogen-treated mouse, it could be found that estrogen acts as a potential activator of GATA-1, then promotes the activation of STAT1 for MKs polyploidization, maturation, and platelet production. That is consistent with the result that platelet counts in estrogen-treated mice had a significant increase. 75 Evidence from clinical cases in postmenopausal women receiving estrogen replacement therapy showed an increase in MK numbers, 76 but the stable effect of estrogen on elevating platelet counts in human, especially thrombocytopenia patients still require further investigation.
Melatonin
Melatonin is a hormone synthesized and secreted mainly from the pineal gland. 77 It is mainly used as a drug to treat sleep disorders, such as insomnia and circadian rhythm disorders. 78 It could be potentially beneficial to the treatment of neurological diseases, cardiovascular diseases, bone diseases, and cancers.79–82 Therapeutic potential of melatonin in thrombocytopenia is under development. Melatonin could promote thrombopoiesis by increasing HSCs and MKs expansion, PPF, and platelet formation. 83 From a molecular perspective, melatonin-induced thrombopoiesis might be mediated by ERK1/2 and Akt signaling. 83 Besides stimulating thrombopoiesis, melatonin could inhibit MKs apoptosis by activating Akt signaling through melatonin receptors. 84 Recent clinical evidence supported that compared with placebo, melatonin treatment could significantly enhance platelet counts (from 175.67 ± 92.84 to 191.10 ± 98.82) in patients who were affected by liver disease, reflecting its potential in thrombocytopenia treatment. 85 Further clinical evidence on patients with different kinds of thrombocytopenia is required, and melatonin is hopeful to become novel choice in the drug treatment of thrombocytopenia.
Recombinant human growth hormone
Recombinant human growth hormone (rhGH) is used for replacing treatment of growth hormone deficiency. 86 It also acts as the thrombopoiesis-stimulating agent which promotes various steps in platelet development, including the late stage of MK differentiation, PPF, and production of platelet. 87 Treatment of rhGH could activate STAT5. In an early phase, activation of ERK1/2 proceeds slowly but in a prolonged period in M07e megakaryoblastic cells and primary MKs treated with rhGH. However, activation of Akt was only observed in mature MKs but not in M07e megakaryoblastic cells, implying that the terminal differentiation of MKs induced by rhGH may be attributed to the Akt pathway. 87 Moreover, through significantly accelerating and promoting platelet production, rhGH was manifested to strengthen the effect of a tandem dimer of thrombopoietin mimetic peptide (dTMP) on thrombopoiesis in M07e cells and mice model of radiation-induced thrombocytopenia (RIT), denoting that rhGH and c-MPL may achieve thrombopoiesis complementarily and synergistically. 87 More clinical evidence is required to confirm the promoting effect of rhGH on relieving thrombocytopenia, especially in combination with THPO and THPO-RAs.
Clozapine
Clozapine is an antipsychotic drug effective for resistant schizophrenia. 88 However, its stimulating effects on the hematopoietic system, such as agranulocytosis, leukocytosis, and thrombocytosis, are recognized as significant side effects.89,90 A follow-up study about clozapine’s stimulating effect on patients’ thrombopoiesis of a 1-year period demonstrated that since the beginning of clozapine treatment in the first week, platelet counts increased synchronically but transiently, and the early spike in platelet counts reflected raised IL-6 levels, which induced thrombopoiesis through the gp130 signaling and interaction with IL-3 and THPO.91,92 This result suggests that IL-6 might be the crucial factor in clozapine-induced thrombocytosis. However, the direct evidence of clozapine’s interactions with IL-6 or other important factors such as THPO to promote thrombopoiesis is required for affirming the relevance of clozapine to the platelet development process.
Yes-associated protein inhibitors
Yes-associated protein (YAP) is an effector molecule in the Hippo pathway, which plays an essential role in MKs proliferation, differentiation, and production of platelet. 93 Dobutamine and verteporfin are two Food and Drug Administration (FDA)-approved YAP inhibitors. Dobutamine is commonly used for septic shock and heart failure,94,95 and verteporfin for cancer, age-related macular degeneration (AMD), central serous chorioretinopathy (CSC), ocular histoplasmosis syndrome, and pathologic myopia.96–98 In vitro experiment revealed that dobutamine and verteporfin could induce megakaryocytic cell maturation and generate thrombopoiesis. 99 Dobutamine phosphorylates YAP, inhibits YAP translocation into the nucleus, and interacts with its target genes. 99 Verteporfin inhibits the interaction between YAP and transcriptional enhanced associate domain (TEAD) and downregulates the expression of their downstream target genes. 99 Treatment with dobutamine or verteporfin on MEG-01 cells results in an increasing level of MKs maturation, differentiation, and production of platelets, and their thrombopoiesis-stimulating effects may be related to upregulation of Fli-1, 99 indicating this group of agents’ potential effect in the treatment of thrombocytopenia.
Natural agents that promote platelet production
Ingenol
Ingenol is a natural product with potential effect in the treatment of warts, skin keratoses, and cancers by activating PKC-α/δ.100,101 Drug screening found that ingenol could promote differentiating MK and producing platelet. Specifically, it could stimulate differentiation of MK in K562 and HEL cells, and accelerated thrombopoiesis in RIT mice model, as an increasing platelet counts and MKs in BM and spleen was seen. 102 RNA-sequencing revealed that the thrombopoiesis-stimulating process of ingenol may foster through PI3K-Akt signaling. 102 Especially, using c-MPL knock-out mice or deleting THPO or c-MPL by their neutralizing antibodies in K562 cells could not suppress the thrombopoietic effect of ingenol, indicating that ingenol may be a THPO-independent thrombopoiesis-stimulating agent, which provides a potential medicine for thrombocytopenia treatment. 102
3,3′-di-O-methylellagic acid 4′-glucoside
3,3′-di-O-methylellagic acid 4′-glucoside (DMAG), a natural ellagic acid derived from Sanguisorba officinalis L. (SOL), has potential antioxidant and hepatoprotective effect. 103 DMAG is found to have thrombopoiesis-stimulating effect as increasing differentiation of MKs was observed in the HEL cells with DMAG treatment. 104 According to Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses, molecular docking simulation, molecular dynamics simulation, and western blot analysis in the human erythroleukemia (HEL) cells, DMAG could bind to TLR2, ITGB3, and ITGA2B and activate the PI3K-Akt signaling pathway. 104 In mice model of RIT, DMAG could increase the number of von Willebrand factor (VWF)-positive MKs and platelets, with no difference in the mean platelet volume (MPV) compared with THPO treatment group. 104 The function of DMAG-induced platelets is assessed by platelet adhesion, platelet aggregation, and tail bleeding time assays, indicating the potential value of DMAG on platelet recovery of thrombocytopenia treatment. 104
Conclusion
Thrombopoiesis is a complex process that offers various targets for different thrombopoiesis-stimulating agents. THPO–MPL is the primary pathway of regulating thrombopoiesis. Targeting directly the THPO–MPL pathway or its downstream pathways, such as JAK/STAT, ERK1/2, and PI3K-Akt signaling, accounts for many thrombopoiesis agents. Meanwhile, other pathways, such as the ERβ-dependent pathway and the Hippo pathway, are significantly effective in thrombopoiesis, and drugs promote thrombopoiesis independent to THPO signaling may reveal novel regulator pathway of platelet development.
Some thrombopoiesis drugs are in clinical treatment for various kinds of thrombocytopenia. THPO-RAs are the most commonly used thrombopoiesis-stimulating agents in the treatment of thrombocytopenia as an upgrade alternative of recombinant THPOs. IL-11-based agents and ATRA might be beneficial to promoting platelet production, but they usually play a role as an assistant medicine in the combined therapy for ITP, with little clinical evidence in the treatment of other kinds of thrombocytopenia. However, the risk of drug-induced thrombocytosis and thrombosis events should be considered when using these drugs in clinical practice.
Further investigations are needed for other thrombopoiesis-stimulating agents which are newly found to promote platelet production and may show potential value in supplementing treating regimens for thrombocytopenia. Some agents discussed above such as neurotransmitters – EPI, NE and DA, hormones – estrogen and rhGH, and melatonin, antipsychotics, such as clozapine, two YAP inhibitors and some natural products, their thrombopoiesis-stimulating effects and potential signaling pathways are investigated. Still, more thrombopoiesis agents are investigated, but the underlying mechanisms are unknown, which worth introducing briefly. Valproic acid (VPA) was found to induce MK polyploidy, maturation, PPF, and PLP formation, 60 and upregulation of FLI-1 was detected in VPA-treated MEG-01 cells. 99 The thrombopoietic effect of VPA may be different from most HDACIs that inhibit platelet production and cause thrombocytopenia,105–107 suggesting the specificity of VPA in stimulating thrombopoiesis. BKT140, a CXCR4 antagonist, could induce the development of MKs and stimulate the production of platelet progenitors, potentially reducing the severity and duration of CIT in mice, however, thrombopoiesis-stimulating effects are not found in other CXCR4 inhibitors. 108 Traditional drug for ITP treatment, such as vinca alkaloids (VAs), may have thrombopoiesis-stimulating effects apart from commonly known immunosuppressive effect, according to animal experiment in the rat.109–111 The development of novel drug screening models has become an effective way for investigating thrombopoiesis-stimulating agents. Using the b1-tubulin reporter line with imMKCL-derived iPSCs, two agents, Wnt-C59 and TCS-359, which promote MK maturation and PLP release, were successfully found. 112 After investigation, figuring out the definite mechanism of their thrombopoietic effect are vital for developing novel medicine in clinical treatment of thrombocytopenia.
The future of thrombocytopenia treatment is encouraging for many thrombopoiesis-stimulating agents which are in clinical trials or early investigations. Thrombopoiesis-stimulating agents could deal with different types of thrombocytopenia, such as ITP, CIT, CLD, and IT, according to clinical evidence. Another large amount of thrombopoiesis-stimulating agents is not yet in clinical use, but research evidence has proved their potential effects in thrombocytopenia treatment to a certain extent. Most agents play their effects through THPO–MPL or its downstream signaling pathways, and some novel pathways regulating platelet development should be highly regarded. The development of thrombopoiesis-stimulating agents may enrich the pharmacological armamentarium for medical treatment of thrombocytopenia.
