Abstract
Immune thrombocytopenia (ITP) is a bleeding disorder caused by a decrease in platelet count resulting from increased destruction and insufficient production of platelets. Although impaired regulatory T-lymphocyte activity plays a critical role in platelet destruction, many other immunologic abnormalities are also likely to be involved. Importantly, patients with ITP appear to have defects in a thrombopoietin-mediated physiological mechanism that compensates for a decrease in platelet count by increasing platelet production. Thus, simultaneous treatment of multiple pathogenic pathways involved in ITP could potentially result in synergistic efficacy. While conventional treatments for ITP suppress or modulate the immune system to reduce platelet destruction, a unique class of ITP therapy, namely thrombopoietin receptor agonists (TPO-RAs), improves platelet production by activating the thrombopoietin pathway. As hypothesized, preliminary studies show that combinations of eltrombopag, an oral TPO-RA, with conventional treatments improve outcomes in both newly diagnosed and refractory patients. In this review, the clinical experience with eltrombopag-based combinations in patients with ITP is summarized and the implications of the available data are discussed.
Keywords
Introduction
Immune thrombocytopenia (ITP) is a bleeding disorder affecting 3 to 4 per 100,000 adults/year.1,2 Apart from the risk of severe bleeding, patients with ITP may also have poor quality of life, comparable to that of patients with diabetes. 3 ITP is caused by a decrease in platelet count below 100 × 109/l as a result of increased destruction and insufficient production of platelets, caused by an autoimmune reaction.1,4 Multiple immunologic abnormalities are likely to be involved in the immune reaction to platelets, and impaired regulatory T-lymphocyte (Treg) activity is considered to play a significant role.5–8 In most patients with ITP, the immune attack is mediated by antiplatelet antibodies secreted by plasma cells. 8 These autoreactive antibodies cause opsonization of platelets by macrophages and may interfere with platelet production by impairing megakaryocyte function. 8 Importantly, in approximately 40% of patients with ITP, no antiplatelet antibodies are detected and the immune attack may be carried out directly by cytotoxic T-cells. 8
Under normal circumstances, a decrease in platelet count leads to increased plasma thrombopoietin level, which induces a compensatory boost in platelet production in bone marrow. 9 Through this mechanism, platelet production may increase more than 20-fold. 10 However, patients with ITP appear to have defects in this physiological mechanism. The level of thrombopoietin in serum is not elevated in response to thrombocytopenia in such patients and, accordingly, a compensatory boost in platelet production is absent.9,11
Conventionally, the goal of first-line therapy for ITP is to quickly increase platelet count in patients with high risk of bleeding or active bleeding, while durability of response and long-term safety and tolerability are considered less of a priority. 12 Therefore, most patients with ITP relapse and consequently receive multiple lines of monotherapy, including repeated courses of first-line treatments until a treatment providing a durable platelet response is found. During this ‘trial and error’ period, which may last months or even years, these patients may experience significant disease burden associated with the risk of bleeding events and adverse effects of treatment.3,13 In addition, there is a small but significant group of multirefractory patients who do not achieve a stable response with any available monotherapy. 14 Both of these patient groups could benefit from the potential synergistic efficacy of combination treatments that simultaneously address multiple disease mechanisms. Being the only available treatment option for ITP that can address insufficient platelet production, thrombopoietin receptor agonists (TPO-RAs), including eltrombopag, play a central role in combination treatment approaches for ITP (Figure 1). 15

Pathogenesis of immune thrombocytopenia and therapeutic mechanisms of current treatments.
Conventional treatment of immune thrombocytopenia
Treatment-naïve patients
First-line management options for ITP usually include corticosteroids (dexamethasone, prednisone) and immunoglobulins [intravenous immunoglobulin (IVIG), Rho(D) immune globulin (anti-D)], which produce fast but transient responses. 4 Corticosteroids show an initial response in 60–70% of patients within 2–14 days, but the response often lasts <6 months.4,16 IVIG will produce a response in 1–3 days in 90% of patients, but the response typically lasts only 2–4 weeks4,16
The rapid effects of first-line treatments are primarily mediated through inhibition of platelet opsonization, which is the final step of platelet destruction (Figure 1).8,17 However, these reagents have other direct and indirect effects on the immune system, including decreasing autoantibody production and rescuing Treg function, which may play a role in their clinical activity.8,15
Refractory patients
For patients refractory to first-line treatments, common therapeutic options include TPO-RAs, the anti-CD20 antibody rituximab, splenectomy, and immunosuppressive agents.4,12 Rituximab, which is licensed for use in certain hematologic malignancies and autoimmune disorders, depletes the B-cell pool involved in the production of autoreactive antibodies and may also restore Treg activity.5,18 Although this drug is often used in patients with refractory ITP, it has a variable response rate and limited duration of response.19,20 In uncontrolled studies, 63% of patients showed an immediate response to a single course of rituximab, and the 2-year response rate was 40%.19,21 In the only placebo-controlled trial of rituximab, no significant benefits beyond 1.5 years were reported. 20
Splenectomy is the oldest option for long-term management of ITP and is still being used due to its high durable response rate (60–70%).6,22 Nevertheless, this invasive option is associated with both risk of surgical complications and lifelong risks caused by the loss of splenic function, including infection, thromboembolism, and even malignancy.22,23 Therefore, once effective pharmaceutical management options became available, the use of splenectomy has declined; however, it must still be considered according to patients’ comorbidity risk and preference, and it is a very effective therapeutic option. To date, no long-term outcomes have been compared between patients undergoing splenectomy and those treated with medical therapies. 24
Many other agents with immunosuppressive properties, including azathioprine, cyclosporine A (CSA), cyclophosphamide, danazol, dapsone, mycophenolate mofetil, and vincristine, have been used to treat refractory ITP. While most of these agents have limited efficacy as monotherapy, they are sometimes preferred due to their low cost.4,12,25
Eltrombopag for treatment of immune thrombocytopenia
TPO-RAs are the only therapeutic option for ITP that increases platelet production (Figure 1). 15 In addition to promoting platelet production from existing megakaryocytes, TPO-RAs may also enhance proliferation of megakaryocytes in bone marrow, as suggested by in vitro studies and clinical trials in patients with aplastic anemia.26–28 Importantly, TPO-RAs may reduce platelet destruction by restoring Treg and regulatory B-cell activity, thereby attenuating the autoimmune response to platelets.8,29 In patients treated with TPO-RAs for >3 months, Treg activity was significantly improved compared with the pretreatment group (p = 0.001) and was similar to the Treg activity in healthy subjects (p = 0.9).8,29 Moreover, preliminary evidence suggests that autoantibody levels in patients with ITP may progressively decrease with TPO-RA treatment, which may contribute to restoration of immune tolerance to platelets. 30
Eltrombopag is an oral TPO-RA approved for use in more than 80 countries, including the United States and European Union countries. 22 In randomized, controlled trials, eltrombopag has been shown to safely and durably increase platelet count in both adults and children with refractory ITP.31–33 Clinical evidence from patients who received eltrombopag for up to 8.8 years supports that eltrombopag retains its safety and efficacy with long-term continuous use, 34 even though it would be necessary to evaluate further related events associated with eltrombopag for a longer term.
Although many patients require continuous treatment with eltrombopag to maintain a response, in approximately 30% of patients, eltrombopag induces disease remission and can be discontinued.30,35–37 However, it is important to note, that these studies defined remission rate using different criteria, besides, some of these patients could achieve spontaneous remission. On the other hand, some patients achieving remission with eltrombopag had ITP that was highly refractory (seven lines of prior therapy, including splenectomy) and had been diagnosed as long as 40 years before receiving eltrombopag, suggesting that eltrombopag-induced remission may be feasible in any patient with ITP achieving an initial response with eltrombopag. 37 It was proposed that restoration of immune tolerance by reduction of autoantibody levels and increase in Treg function may play a role in TPO-RA-induced remission.30,37
Eltrombopag combinations for newly diagnosed/persistent immune thrombocytopenia
Improving the efficacy and durability of first-line treatments would reduce the number of patients who fail therapy and, therefore, help spare them from the burdensome ‘trial and error’ period. We and others investigated whether the outcomes of first-line treatments could be improved by the addition of eltrombopag in several clinical studies (Table 1).
Studies of eltrombopag combinations for immune thrombocytopenia.
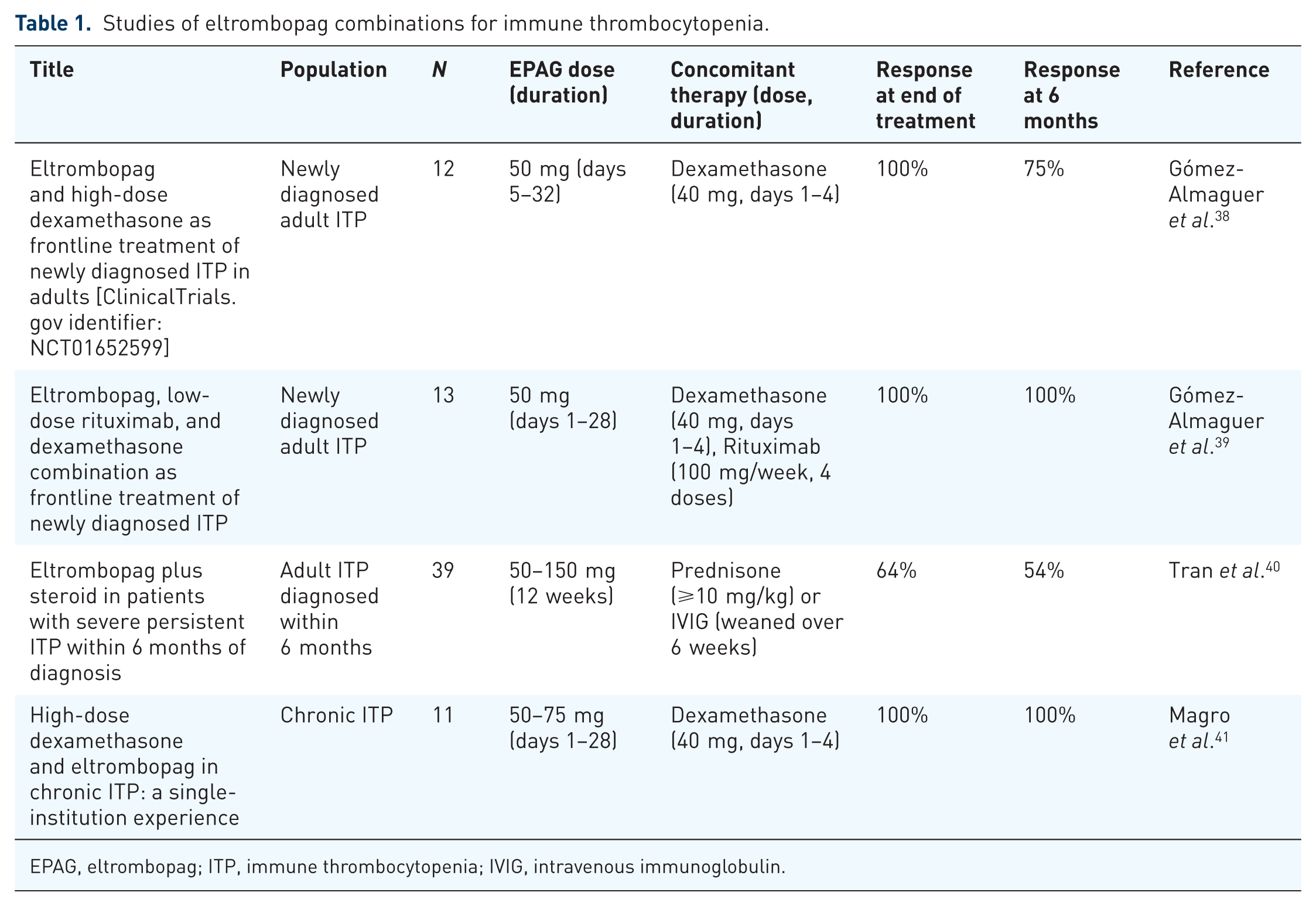
EPAG, eltrombopag; ITP, immune thrombocytopenia; IVIG, intravenous immunoglobulin.
Eltrombopag and high-dose dexamethasone as first-line treatment for newly diagnosed immune thrombocytopenia
In our open-label, single-arm study [ClinicalTrials.gov identifier: NCT01652599], we studied the potential ability of eltrombopag in combination with corticosteroids to increase the response rate in the first-line treatment of ITP. 38 During the study, 12 adult patients with newly diagnosed ITP received dexamethasone (40 mg, days 1–4) in combination with a limited course of eltrombopag (50 mg, days 5–32). 38 All patients had a response (platelet count ⩾ 30 × 109/l) at the end of treatment (day 33), and the proportion of patients with platelet count ⩾ 50 × 109/l at 6 months was 75% with the addition of eltrombopag versus 37% reported with dexamethasone alone in a similar study cited as a historical control. 38 The combination was well tolerated, with no reports of adverse events, myelofibrosis, or thrombosis. 38
Eltrombopag, high-dose dexamethasone, and low-dose rituximab for newly diagnosed immune thrombocytopenia
The combination of rituximab with high-dose dexamethasone was shown to be more effective than either therapy alone and induced durable remission in the majority of adult women with refractory ITP who had been diagnosed for <1 year.42,43 Similarly, we showed that the combination of rituximab plus high-dose dexamethasone may be a more effective first-line therapy than dexamethasone alone. In adult patients who received this first-line combination therapy, the complete sustained response rate at 6 months and relapse rate were 76.2% and 15.8%, respectively, compared with 30% and 62.5% for a historical group who received standard first-line treatment with prednisone. 44 Nevertheless, >20% of these patients and >80% of patients in other populations (e.g. adult men, disease duration > 1 year) will relapse. 43
We hypothesized that the addition of eltrombopag to this combination could address the pathological defects in patients who were refractory to the combination of steroid plus rituximab, thereby resulting in responses in virtually all patients and a reduced relapse rate with triple therapy. We performed an open-label, single-arm study including 13 patients (aged ⩾ 16 years) with newly diagnosed ITP who received eltrombopag (50 mg/day, days 1–28) in combination with low-dose weekly rituximab (100 mg/week, four doses) and high-dose dexamethasone (40 mg/day, days 1–4). 39 All patients had a response at a median of 4 days (range 3–10 days), and all but one achieved a complete response in 9 days (range 7–22 days). Two patients relapsed at 5 and 12 months after diagnosis, respectively, with a probability of relapse-free survival of 79% at 2 years. 39 The treatment combination was generally well tolerated. Adverse events were mild and included thrombocytosis (platelet count > 400 × 109/l; n = 2), insomnia (n = 2), and myalgia (n = 1). 39
Eltrombopag plus steroid in patients with severe persistent immune thrombocytopenia within 6 months of diagnosis
This multicenter, single-arm, open-label study included 39 patients with platelet count <30 × 109/l despite a daily dose of prednisone of 1 mg/kg for >2 weeks from diagnosis or required prednisone ⩾ 10 mg/day or recurrent IVIG to maintain platelet count ⩾ 30 × 109/l within 6 months of diagnosis. 40 In combination with each patient’s ongoing steroid therapy, the investigators administered eltrombopag at 50–150 mg/day based on platelet count. 40 Steroids were weaned over 6 weeks, if weaning was considered clinically possible. Of 39 patients enrolled, 35 (90%) completed 12 weeks of treatment. Discontinuations prior to week 12 occurred in four patients: three required new ITP therapy and one developed thrombocytosis. 40 At week 12, the response rate was 64%, with a complete response seen in 41% of patients and a median platelet count among responders of 168 × 109/l. At week 26, the response rate was 54%, with 28% of patients achieving a complete response. 40 Two patients had serious adverse events (venous thromboembolism), but there were no other adverse events or deaths. 40
Eltrombopag combinations for refractory immune thrombocytopenia
Patients with multirefractory ITP who do not respond to any available monotherapy have a high risk of morbidity and even death, 14 and, therefore, urgently require additional therapeutic options. Simultaneous activation of platelet production and inhibition of autoimmune response to platelets through the use of eltrombopag, in combination with immunosuppressant therapy, may be an effective option in these patients. 14
Eltrombopag and high-dose dexamethasone for chronic immune thrombocytopenia
In an unselected series of 11 patients with chronic ITP treated at a single institution from May 2014 to December 2015, patients received dexamethasone (40 mg/day) for 4 days every 28 days and eltrombopag (50–75 mg/day) daily. All patients achieved a response (platelet count ⩾ 30 × 109/l), with a complete response (platelet count ⩾ 100 × 109/l) seen in 10 of 11 patients. 41 The median time to maximum response was 12 weeks (range 3–39 weeks). Responses were maintained in all patients after a median follow up of 29 weeks (range 8–89 weeks) but complete response was lost in five patients. 41
Thrombopoietin in combination with rituximab for refractory immune thrombocytopenia
While clinical trial data on eltrombopag/rituximab combination therapy in patients with refractory ITP are scarce, several studies reported the results on combinations of rituximab plus recombinant thrombopoietin (rhTPO).
A multicenter, randomized, open-label study of rituximab [ClinicalTrials.gov identifier: NCT01525836] (100 mg/week, 4 weeks) plus rhTPO (300 U/kg/day, 14 days) compared with rituximab alone in 115 patients with corticosteroid-resistant or relapsed ITP 45 demonstrated an increased complete response rate and decreased time to response with rituximab plus rhTPO compared with rituximab alone (45% versus 24%, 7 days versus 28 days, respectively). The rates of long-term responses (6, 12, and 24 months) were numerically greater in patients who received combination therapy, but the difference did not reach statistical significance (p = 0.12)
A single-arm study of 14 patients with refractory ITP who received low-dose rituximab in combination with rhTPO 46 reported a response rate of 93%, with 50% complete response. Relapses were observed in two patients at 6 and 16 months after complete response, respectively. Severe pulmonary complications, potentially related to rituximab, caused death in two elderly patients with concurrent diabetes. Furthermore, it is important to note that a small patient series of romiplostim in combination with rituximab is available with similar documented success. 47
Eltrombopag in combination with immunosuppressive therapy for refractory immune thrombocytopenia
Several immunosuppressive therapies (ISTs; e.g. CSA, cyclophosphamide, as well as the antimetabolites azathioprine, 6-mercaptopurine and mycophenolate mofetil) have been used in patients with ITP with varying success.4,12 However, because combinations of these agents with eltrombopag have not been formally investigated in patients with ITP, direct evidence supporting the safety and efficacy of these combinations is limited.14,48,49
Although eltrombopag plus CSA is commonly studied in patients with aplastic anemia, no clinical study has directly investigated this combination in patients with ITP. Indirect evidence comes from a randomized, controlled study in 36 patients with steroid-resistant ITP, which showed that CSA (1.5–2.0 mg/kg, twice daily for 3 months) plus rhTPO (1 µg/kg, 14 days) significantly decreased the relapse rate at 3 months (29.4% versus 87.5% with rhTPO only; p < 0.01). 48
Eltrombopag combinations for immune thrombocytopenia with other medications
Before the availability of rituximab and TPO-RAs, other agents, including danazol, dapsone, and vinca alkaloids, were used as alternative treatments for ITP. While these therapies are not recommended as monotherapy under current ITP treatment guidelines due to the lack of sufficient clinical trial data supporting their efficacy in ITP, 4 their low cost may allow their inclusion in combination therapies in patients who are refractory to other options.
Discussion
Two major obstacles in the treatment of ITP are the high rate of relapse with first-line treatments and the significant minority of patients who are refractory to multiple lines of treatment. It may be possible to surmount these challenges by modifying the current therapeutic approach, which relies on monotherapies targeting only one aspect of the disease mechanism.
Preliminary clinical evidence suggests that improvement of platelet production with eltrombopag may enhance the outcomes of concomitant ITP therapies. First, eltrombopag added to standard first-line therapy increased the durability of response and decreased the relapse rate. Thus, in patients with newly diagnosed ITP, when used as frontline therapy, eltrombopag may help reduce the need for additional courses of steroid or IVIG. A relatively short duration of eltrombopag treatment early in the disease may increase Treg activity, thereby potentially altering the natural course of the disease without the need for continued administration.
Second, eltrombopag in combination with immune modulation may be effective in patients who are refractory to single-agent ITP treatments. By inhibiting the autoimmune response to platelets and reducing antiplatelet antibodies, which may interfere with megakaryocyte function, 8 steroids may induce an immunologic environment more permissive to a response to eltrombopag. While available clinical data do not suggest a major long-term benefit with TPO pathway activation in combination with rituximab, 45 it is possible that patients who are receiving rituximab therapy may benefit from eltrombopag as a bridge therapy.
Finally, eltrombopag in combination with IST may be a viable option in patients who are refractory to eltrombopag as a single agent, which is becoming more relevant, as it is increasingly used in earlier disease stages, as illustrated by a recent claims database analysis in the United States. 50 Many ISTs were permitted in clinical trials of eltrombopag, and no serious adverse events attributed to concomitant use of these therapies with eltrombopag have been reported.51,52 However, these trials included only a small number of patients receiving each combination. It also should be noted that danazol, azathioprine, and 6-mercaptopurine are associated with liver toxicity 12 and should be used with caution in patients receiving eltrombopag.
Importantly, no dose reduction appears to be required when eltrombopag is used in combination with other ITP therapies. For adult patients with ITP, the recommended starting dose for eltrombopag is 50 mg/day, which can be increased to 75 mg/day if a sufficient platelet count is not reached. 31 Higher doses (75–150 mg/day) are approved in patients with severe aplastic anemia and have been tested in a limited number of patients with ITP.14,31,43,53 Preliminary results suggest increased efficacy in some patients, but more evidence is needed. 53
Because increased platelet destruction in ITP may result from diverse immunologic abnormalities, combinations of ISTs with distinct mechanisms of actions may further increase response rates. A pilot study of eltrombopag, rituximab, and dexamethasone as first-line ITP therapy showed a remarkable 100% initial response, with a 2-year relapse-free survival rate of 79%. 39
Conclusions
With improved understanding of the pathogenesis of ITP, the therapeutic potential of combination treatments that address multiple disease mechanisms has become clear. As the only drug class that may improve platelet production in ITP, TPO-RAs, including eltrombopag, are essential components of combination therapies for ITP. Albeit limited, current evidence is consistent with expectations and suggests that combination therapy with eltrombopag may help patients with newly diagnosed or highly refractory ITP attain their treatment goals. Future studies are needed to confirm the preliminary evidence, develop potential diagnostic methods to identify treatment approaches based on pathogenic mechanisms, and elucidate optimal ISTs for eltrombopag combinations.
Footnotes
Funding
ClinicalThinking, Inc., NJ, USA provided medical writing and editorial support for the preparation of this manuscript, which was funded by Novartis Pharmaceuticals Corporation, East Hanover, NJ, USA. Novartis did not influence the content of the manuscript nor did the author receive financial compensation for authoring the manuscript.
Conflict of interest statement
The author declares no conflicts of interest in preparing this article. The author has received honoraria for speaker roles by Novartis, Amgen, Celgene, Bristol, Janssen and Tecnofarma, none related to products discussed in this publication.
