Abstract
Background:
Daratumumab is one of the most widely used treatments for relapsed/refractory multiple myeloma (MM) patients. However, not all patients achieve a lasting therapeutic response with daratumumab.
Objectives:
We hypothesized that a durable response to daratumumab could be predicted by the balance between the MM tumor burden and host immune status.
Design:
We conducted a retrospective study using the real-world data in the Kansai Myeloma Forum (KMF) database.
Methods:
We retrospectively analyzed 324 relapsed/refractory MM patients who were treated with daratumumab in the KMF database.
Results:
In this study, 196 patients were treated with daratumumab, lenalidomide, and dexamethasone (DLd) regimen and 128 patients were treated with daratumumab, bortezomib, and dexamethasone (DBd) regimen. The median age at treatment, number of prior treatment regimens and time-to-next-treatment (TTNT) were 68, 4 and 8.02 months, respectively. A multivariate analysis showed that the TTNT under the DLd regimen was longer with either higher monocyte counts (analysis 1), higher white blood cell (WBC) counts (analysis 2), lower β2 microglobulin (B2MG < 5.5 mg/L) or fewer prior regimens (<4). No parameters were correlated with TTNT under the DBd regimen.
Conclusion:
We propose a simple scoring model to predict a durable effect of the DLd regimen by classifying patients into three categories based on either monocyte counts (0 points for ⩾200/μl; 1 point for <200/μl) or WBC counts (0 points for ⩾3500/μl; 1 point for <3500/μl) plus B2MG (0 points for <5.5 mg/L; 1 point for ⩾5.5 mg/L). Patients with a score of 0 showed significantly longer TTNT and significantly better survival compared to those with a score of 1 or 2 (both p < 0.001). To confirm this concept, our results will need to be validated in other cohorts.
Introduction
The prognosis of multiple myeloma (MM) patients has been dramatically improved by the introduction of immunomodulatory drugs (iMIDs) and proteasome inhibitors (PIs). 1 In addition to iMIDs and PIs, the anti-CD38 antibody daratumumab has shown a high response rate with superior prognosis both for relapsed/refractory and treatment-naïve transplantation-ineligible MM patients when used in combination with iMIDs or PIs.2–4 Regimens containing daratumumab were found to be among the most effective used in relapsed/refractory MM in two network meta-analyses.5,6 However, although more than 90% of MM patients responded to daratumumab treatment in clinical trials,2,3 a discrepancy in the response rate of daratumumab treatment between the clinical trials and real-world data has been reported, especially for relapsed/refractory cases.7,8 Moreover, a substantial number of patients either did not obtain a therapeutic response with daratumumab, or obtained a therapeutic response but could not sustain it. Unfortunately, we do not have appropriate biomarkers to predict the response or the durable efficacy of daratumumab before administration.
To identify, prior to treatment, patients with a potentially durable response to daratumumab, we focused on the immunological aspect of daratumumab. The mechanisms of action of daratumumab are immune-mediated effects, such as complement- or antibody-dependent cell-mediated cytotoxic effects and depletion of CD38-positive regulatory immune cells.9–16 Among the immune cells, natural killer (NK) cells and monocytes play important roles particularly in daratumumab-mediated myeloma cells killing.13–16 Here, we hypothesized that the balance between the tumor burden of myeloma and immune conditions [represented by white blood cell (WBC) counts and other leukocyte counts] might predict the efficacy of daratumumab treatment. As a proof of concept, we conducted a retrospective observational analysis using real-world data from the Kansai Myeloma Forum (KMF) database in Japan.
Methods
Study design and participants
KMF is a study group consisting of 123 physicians at 46 facilities in Japan. The KMF database includes physician-reviewed, real-world clinical data on the diagnosis, treatment, and periodical follow-up of patients with plasma cell dyscrasias. This study was approved by the Data Management Committee of the Kyoto University Graduate School of Medicine Institutional Review Board (approval no. R2887).
In the KMF database, 4133 patients with plasma cell dyscrasias were registered in March 2021. All the patients were diagnosed as having MM or MM-related disorders based on institutional assessment. From the KMF database, we selected patients older than 20 years who were treated for symptomatic MM with active-disease status in relapse setting between November 2017 to March 2021 using a regimen including daratumumab, after its approval for clinical use. A total of 388 patients met the above inclusion criteria for this study (Supplement Figure 1). We conducted secondary research to collect the laboratory data 1 to 7 days before cycle 1 day 1 daratumumab treatment. Sixty-four patients were omitted due to a lack of data, leaving 324 relapsed MM patients whose data were included in the final analysis. These relapsed/refractory MM patients were followed until August 2021.
The serum free light chain κ/λ ratio was measured by latex coagulating nephelometry. The patients’ responses to treatment were assessed based on the criteria of the international uniform response criteria 17 for MM. The patients’ best responses against daratumumab were classified by institutional physicians into five categories: complete response (CR), very good partial response (VGPR), partial response (PR), stable disease (SD), and progressive disease (PD).
For the high-risk cytogenetic abnormalities, we adopted the abnormalities reported in the International Myeloma Working group consensus statement, 18 such as deletion 17p, t(4;14), and t(14;16). Unfavorable cytogenetic abnormalities were categorized by a fluorescence in situ hybridization (FISH) analysis.
Statistical methods
We calculated the time to next treatment (TTNT) for first daratumumab treatment as the time from daratumumab treatment until the date of next treatment, death by any cause, or the date of last contact. We chose the TTNT as the primary endpoint instead of progression-free survival (PFS) for our retrospective analysis,19,20 since the timing of PD was difficult to precisely determine in our cohort. The data were censored for the date of next treatment in cases where the cessation of daratumumab was planned in advance. To analyze the underlying factors affecting the TTNT under daratumumab treatment, we first analyzed the TTNT in relation to the treatment regimen [daratumumab, lenalidomide, and dexamethasone (DLd) or daratumumab, bortezomib, and dexamethasone (DBd)]. Then, we analyzed the data regarding the parameters used to estimate the tumor burden of myeloma,21–23 that is, the κ/λ ratio and β2 microglobulin (B2MG), and the parameters which, based on the mechanism of action of daratumumab, appear to be correlated with the host immune status, that is, the WBC count and other leukocyte fractions. The κ/λ ratio and B2MG are recognized to reflect tumor burden.21–23 We selected the leukocyte fractions (neutrophil, lymphocyte, or monocyte counts) that showed significant correlation with TTNT in the univariate analysis, and applied them to the following analysis.
To determine the cutoff values for the WBC counts, neutrophil counts, lymphocyte counts, monocyte counts, and κ/λ ratio, we first tested the 25th, 50th, and 75th percentile values as potential cut-off values (Supplement Figures 2–6). We determined the cut-off value of monocytes as 200/μl and that of the κ/λ ratio as 0.1/10 according to the difference from the reference arm [Supplement Figure 3(A) and (B)]. We used the median value as a cutoff value for WBCs, neutrophils, and lymphocyte counts (Supplement Figures 4 and 5(A)–(D). The cutoff value of B2MG was determined according to the International Staging system 21 for MM (Supplement Figure 6).
The survival curve according to TTNT and the overall survival (OS) curve were plotted using the Kaplan–Meier method, and the log-rank test was used for comparisons among groups. The Cox proportional hazard model was used to calculate the hazard ratio for each variable along with the 95% confidence interval (CI). Variables considered in the univariate analysis were age, gender, high-risk cytogenic abnormalities, WBC counts, neutrophil counts, lymphocyte counts, monocyte counts, κ/λ ratio, B2MG, number of daratumumab treatments, prior regimen number, and prior use of elotuzumab. A multivariate analysis was conducted for the variables which showed a p value of less than 0.1 in the univariate analysis. To establish a predictive model for the durability treatment of DLd, we first included all the factors which showed p < 0.1 in univariate analysis. Because we found a correlation among three factors related to immune tatus enhancement – namely, monocyte counts, lymphocyte counts, and WBC counts – we divided the cases into three different multivariate analyses, respectively, using one of these three factors. We also adjusted the survival curves by the significant factors in the multivariate analysis. We used the bootstrap method to validate the results of our multivariate analysis.24,25 In each step, 1000 bootstrap samples with replacements were created from the dataset. We used C statistics (C-index) to evaluate the predictive accuracy of prediction models.26,27 C-index is calculated by the area under the receiver operating characteristic (ROC) curve. The c-index of an ideal test became closer to 1.0. All statistical analyses were performed using the EZR (ver. 1.54) software package (Saitama Medical Center/Jichi Medical University, Saitama, Japan) 28 along with a graphical user interface for the R software package (version 4.0.3; The R Foundation for Statistical Computing) or SPSS software version 28 (IBM, USA). P values <0.05 were considered significant in all analyses.
Results
TTNT of daratumumab in relapsed MM
The characteristics of the patients undergoing each regimen are summarized in Table 1. In brief, a total of 324 patients incorporating daratumumab were analyzed. The median age at the time of daratumumab treatment was 68 years old. The numbers of patients treated with the DLd and DBd regimens were 196 (60.5%) and 128 (39.5%), respectively. The median number of prior regimens was 4. In most cases, both iMIDs and PIs were used before daratumumab treatment. Elotuzumab was used before daratumumab treatment in 35 cases (17.9%) treated with a DLd regimen and 9 cases (7.0%) treated with a DBd regimen. Autologous stem cell transplantations (auto-SCT) were performed prior to daratumumab treatment in 61 cases (31.1%) treated with a DLd regimen and 31 cases (24.2%) treated with a DBd regimen. The histogram of laboratory data is shown in Supplemental Figure S1. Patients with a CR, VGPR, or PR were regarded as having a therapeutic response to daratumumab; these included 130 patients (66.3%) treated with the DLd regimen and 79 cases (61.7%) treated with the DBd regimen (Supplement Figure 7).
Characteristics of the patients with multiple myeloma.

Auto-SCT, autologous stem cell transplantation; B2MG, β2 microglobulin; DBd, dexamethasone; DLd, dexamethasone; IMIDs, immunomodulatory drugs; ISS, international staging system; NA, not available; PI, proteosome inhibitor.
The characteristics of multiple myeloma patients who were treated with a daratumumab, lenalidomide, and DLd regimen or daratumumab, bortezomib, and DBd regimen are shown in Table 1. Laboratory data were collected before the daratumumab treatment.
The median TTNT under daratumumab treatment was 8.02 (95% CI: 6.48–9.20) months in this cohort [Figure 1(a)]. When we compared the TTNTs by regimen, the TTNTs under the DLd and DBd regimens were 9.20 (7.85–11.93) and 5.98 (4.30–7.13) months, respectively [Figure 1(b); p = 0.001]. Because the mechanism of action differed between the DLd and DBd regimens, we performed the following analysis according to the type of regimen.

(a) The time-to-next treatment (TTNT) of the multiple myeloma (MM) patients treated with daratumumab. Median TTNT (months) values with the 95% CI (confidence interval) are shown. (b) The TTNT of the multiple myeloma (MM) patients according to the treatment regimen: daratumumab, lenalidomide, and dexamethasone (DLd, black) or daratumumab, bortezomib, and dexamethasone (DBd, red). Median TTNT (months) values with the 95% CI are shown. (c) The TTNT of the MM patients treated with the DLd regimen according to the β2 microglobulin (B2MG) level: less than 5.5 mg/L (black) or 5.5 mg/L or more (red). Median TTNT (months) values with the 95% CI are shown. (d) The TTNT of the MM patients treated with the DLd regimen according to the κ/λ ratio: 0.1–10 (black) and less than 0.1 or 10 or more (red). Median TTNT (months) values with the 95% CI are shown. (e) The TTNT of the MM patients treated with the DLd regimen according to the white blood cell (WBC) counts: less than 3500/μl (black) and 3500/μl or more (red). The median TTNT (months) values with the 95% CI are shown. (f) The TTNT of the MM patients treated with the DLd regimen according to the monocyte counts: less than 200/μl (black) and 200/μl or more (red). The median TTNT (months) values with the 95% CI are shown. The number of patients at risk in each group is shown in the lower panel of each figure.
The underlying factors affecting the TTNT under daratumumab treatment
When we analyzed the impact of tumor burden, B2MG and the κ/λ ratio against TTNT in patients undergoing the DLd regimen, the TTNT under daratumumab treatment was longer in the patients with a lower B2MG (<5.5 mg/L) [p = 0.006; Figure 1(c), Table 2] or a non-deviated κ/λ ratio (κ/λ ratio of 0.1–10) [p = 0.020; Figure 1(d), Table 2]. We next analyzed TTNT according to the immune status, WBC counts and other leukocyte fractions. The patients with higher WBC counts (⩾3500/μl) before daratumumab treatment showed longer TTNT than those with lower WBC counts [p = 0.034; Figure 1(e) and Table 2]. The patients with higher monocyte counts (⩾200/μl) also showed longer TTNT [p = 0.012, Figure 1(f), Table 2]. Higher lymphocyte counts (⩾1000/μl) were associated with slightly longer TTNT, but the neutrophil counts were not correlated with TTNT [Table 2 and Supplement Figure 5(B) and (D)]. Other factors which showed better TTNT in the univariate analysis in patients undergoing the DLd regimen were a prior regimen number < 4 and no prior use of elotuzumab [Table 2, Supplement Figure 8(A)–(D)]. In the analysis of TTNT under the DBd regimen, we could not find any factors which correlated to TTNT (Table 2).
Univariate analysis for TTNT.

Auto-SCT, autologous stem cell transplantation; B2MG, β2 microglobulin; CI, confidence interval; NA, not available; TTNT, time to next treatment.
TNT was calculated from the time of each daratumumab treatment to the time of the next treatment. Univariate analyses against TTNT in MM patients treated with the DLd regimen or DBd regimen were performed for each factor. The log-rank test was used for comparisons among groups. TTNT (months) is shown with the 95% confidence interval (CI) and p value.
Prediction model for daratumumab treatment
We performed a multivariate analysis regarding the TTNT in patients undergoing the DLd regimen by analyzing all the factors that showed p values of less than 0.1 in the univariate analysis. Because the lymphocyte counts and WBC counts were correlated, and the monocyte counts and WBC counts were also correlated [Supplement Figure 9(A)–(B)], we used either monocyte counts (analysis 1), WBC counts (analysis 2) or lymphocyte counts (analysis 3) for the multivariate analysis. In analysis 1, we found that higher monocyte counts (⩾200/μl, p = 0.009), lower B2MG (<5.5 mg/L, p = 0.011), and prior regimen number <4 (p = 0.005) were each independently associated with superior TTNT under the DLd regimen. In analysis 2, higher WBC counts (⩾3500/μl, p = 0.048), lower B2MG (<5.5 mg/L, p = 0.010) and prior regimen number <4 (p = 0.018) were associated with superior TTNT under the DLd regimen (Table 3). In analysis 3, lower B2MG (<5.5 mg/L, p = 0.019) and prior regimen numbers <4 (p = 0.012) were associated with superior TTNT under the DLd regimen. All these multivariate analysis results were confirmed by bootstrap methods (Table 3).
Multivariate analysis for TTNT.

Auto-SCT, autologous stem cell transplantation; B2MG, β2 microglobulin; CI, confidence interval; TTNT, time to next treatment.
Multivariate analyses against TTNT in MM patients treated with the DLd regimen were performed using the factors which showed p < 0.1 in univariate analysis. Because monocyte counts, lymphocyte counts and WBC counts were correlated, we adopted these factors as indexes of immune status as follows: monocyte counts in analysis 1, WBC counts in analysis 2 and lymphocyte counts in analysis 3. The Cox proportional hazard model was used to calculate the hazard ratio for each variable; the 95% CI and p value are shown.
P value indicates the p value after the bootstrapping process (1000 samples).
From these results, we proposed two new models to predict a durable effect (longer TTNT) under the DLd regimen by classifying the patients into three categories based on either (1) monocyte counts and B2MG (model 1) or (2) WBC counts and B2MG (model 2). We assigned 0 points to patients with monocyte counts of 200/μl or more, and 1 point to those with less than 200/μl. We also assigned 0 points to patients with a B2MG less than 5.5 mg/L and 1 point to those with a B2MG of 5.5 mg/L or more. Patients with a total score of 0 showed significantly longer TTNT compared to those with scores of 1 or 2 [p = 0.001, Figure 2(a)]. The c-index for this model was 0.675. We confirmed that this scoring system was significantly correlated with the TTNT under DLd treatment in multivariate analysis with bootstrap methods (Table 4). This model showed the same tendency regardless of the prior regimen numbers [Supplement Figure 10(A)–(B)]. When we analyzed the OS after the DLd treatment, we found that the patients with a total score of 0 showed significantly longer OS than the patients with total scores of 1 or 2 [p < 0.001, Figure 2(b)].

(a) The TTNT of the MM patients treated with the DLd regimen according to the proposed scoring system: 0 points (black), 1 point (red), and 2 points (blue). Total scores were calculated according to the monocyte counts (0 points when ⩾200/μl and 1 point when <200/μl) and B2MG (0 points when <5.5 mg/L and 1 point when ⩾5.5 mg/L) before daratumumab treatment. The median TTNT (months) values with the 95% CI are shown in the figure. The TTNT values were corrected by the number of prior treatment regimens. (b) The overall survival (OS) of the MM patients treated with the DLd regimen according to the proposed scoring system: 0 points (black), 1 point (red), and 2 points (blue). The 1-year OS values of each group with the 95% CI are shown in the figure. The OS values are corrected by the number of prior treatment regimens. (c) The TTNT of the MM patients treated with the DLd regimen according to the proposed scoring system: 0 points (black), 1 point (red), and 2 points (blue). Total scores are calculated according to the WBC counts (0 points when ⩾3500/μl and 1 point when <3500/μl) and B2MG (0 points when <5.5 mg/L and 1 point when ⩾5.5 mg/L) before daratumumab treatment. Median TTNT (months) values with the 95% CI are shown in the figure. The TTNT values are corrected by the number of prior treatment regimens. (d) The overall survival (OS) of the MM patients treated with the DLd regimen according to the proposed scoring system: 0 points (black), 1 point (red), and 2 points (blue). The 1-year OS values of each group with the 95% CI are shown in the figure. The OS values are corrected by the number of prior treatment regimens. The number of patients at risk in each group are shown in the lower panel of each figure.
Multivariate analysis for TTNT.
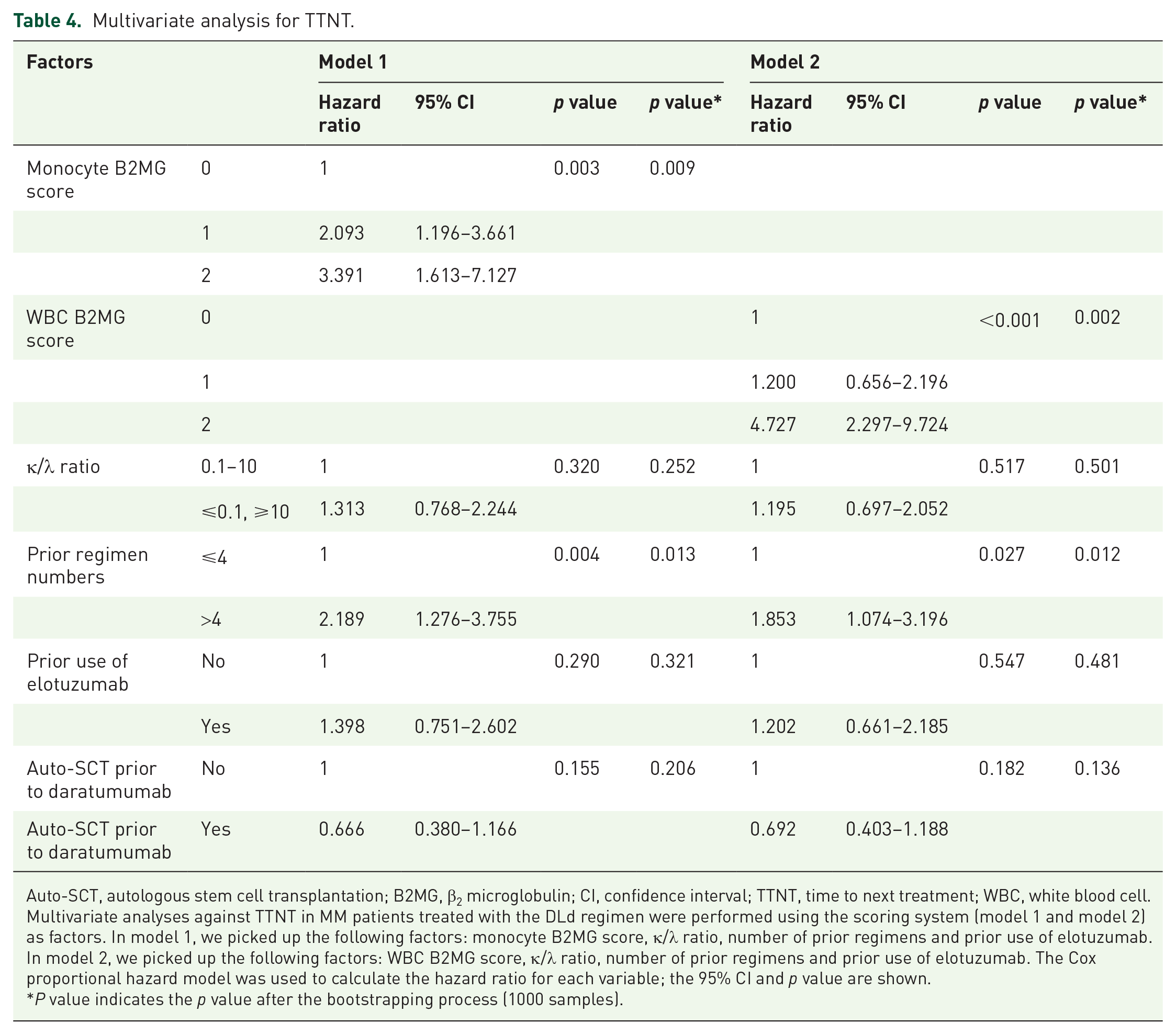
Auto-SCT, autologous stem cell transplantation; B2MG, β2 microglobulin; CI, confidence interval; TTNT, time to next treatment; WBC, white blood cell.
Multivariate analyses against TTNT in MM patients treated with the DLd regimen were performed using the scoring system (model 1 and model 2) as factors. In model 1, we picked up the following factors: monocyte B2MG score, κ/λ ratio, number of prior regimens and prior use of elotuzumab. In model 2, we picked up the following factors: WBC B2MG score, κ/λ ratio, number of prior regimens and prior use of elotuzumab. The Cox proportional hazard model was used to calculate the hazard ratio for each variable; the 95% CI and p value are shown.
P value indicates the p value after the bootstrapping process (1000 samples).
In model 2, we assigned 0 points to the patients with WBC counts of 3500/μl or more and 1 point to those with WBC counts of 3500/μl. We also scored the patients by B2MG in the same manner as in model 1. The patients with total scores of 0 or 1 showed significantly longer TTNT compared to those with a score of 2 [p < 0.001, Figure 2(c)]. The c-index for this model was 0.688. We confirmed that this scoring system was significantly correlated with the TTNT under DLd treatment in multivariate analysis with bootstrap methods (Table 4). This model showed the same tendency regardless of the number of prior regimens [Supplement Figure 11(A) and (B)]. When we analyzed the OS after the DLd treatment, we found that the patients with a total score of 0 showed significantly longer OS than the patients with total scores of 1 or 2 [p < 0.001, Figure 2(d)].
We conclude that by using this simple model, we could predict the patients who could obtain a durable response (longer TTNT) by the DLd regimen (Supplement Figure 12).
Discussion
It has been proposed that the therapeutic effects of novel anti-MM agents involve not only their cytotoxicity against myeloma cells but also their immunomodulatory effects. 29 However, there is a lack of useful biomarkers related to immune status to predict the clinical response before treatment. 30 Recently, several studies have indicated that monocytes might predict the prognosis of MM patients31–33 and also that monocytes are involved in daratumumab-mediated killing of myeloma cells.13–16 We hypothesized that the efficacy of daratumumab could be predicted not only by the tumor burden of myeloma cells but also by the host immune status. As a proof of concept, we chose the κ/λ ratio and B2MG as candidate biomarkers representing the tumor burden. We also selected WBC and several subtypes of WBC which might reflect the host immune status. This study demonstrated that a simple model using B2MG plus either monocyte counts or WBC counts could easily predict the durable efficacy of the DLd regimen in relapsed/refractory MM patients. Patients with a total score of 0—namely, those with a low tumor burden (B2MG < 5.5 mg/L) and preserved host immune cells (monocyte counts ⩾ 200/μl or WBC counts ⩾ 3500/μl)—were determined to be those who could obtain the most benefit from a DLd regimen. In future studies, we plan to use model 1 rather than model 2, because using the monocyte count is a more specific parameter than the WBC count.
Because previous reports have shown that the neutrophil-to-lymphocyte ratio or lymphocyte-to-monocyte ratio is correlated with the prognosis of MM,33,34 we also analyzed neutrophil counts, lymphocyte counts, the neutrophil/lymphocyte ratio, and the monocyte/lymphocyte ratio as possible predictors of host immunity. However, these parameters were not significantly correlated with the TTNT under daratumumab treatment. Our study suggests that among leukocyte fractions, monocytes play the most important role in the DLd regimen, and this result was in keeping with previous reports.16,31,32
Another factor that was associated with a significantly longer TTNT under the DLd regimen in multivariate analysis was a lower number of prior regimens (<4 prior regimens). Because the use of daratumumab in prior regimens has been associated with better prognosis in a clinical study, 2 it is not surprising that the TTNT under the DLd regimen would be shorter in heavily treated patients with treatment-resistant MM. We demonstrated that patients with fewer than 4 prior treatment regimens showed significantly longer TTNT under the DLd regimen, but this difference was not observed in the patients treated with the DBd regimen. Because the median TTNT under the DBd regimen was shorter compared to that in clinical studies,35,36 more treatment-resistant MM patients might have been included in our cohort treated with the DBd regimen.
These findings notwithstanding, we must underscore that daratumumab treatment remains a high-priority treatment option for all MM patients due to its high response rate,2–4 even for the relapsed/refractory MM patients with high tumor burden and suppressive immune status. However, our prediction model provides two important lessons. First, it is important to realize that the efficacy of daratumumab might not be sustained for patients with a high tumor burden and suppressive immune status. Second, it might be necessary for us to prepare for the next treatment after the DLd regimen in these patients.
In our model 1, the TTNT of the patients with a total score of 0 was significantly longer than the TTNT of the patients with total scores of 1 or 2. In model 2, the TTNT of patients with a total score of 0 or 1 was significantly longer than the TTNT of those with a total score of 2. On the contrary, the OS after the DLd treatment was significantly superior in patients with a total score of 0 compared to the OS of the patients with a total score of 1 or 2 in both models. These results could be interpreted as follows. Because there are seldom better treatment options than daratumumab, once patients relapse or become refractory to daratumumab treatment, their prognosis can be quite poor. However, because this study was observational by design, we could not tell whether the prognosis would be changed by choosing another treatment, such as carfilzomib treatment, to debulk the tumor before starting DLd treatment. This might be one of the treatment options, but it needs to be confirmed in a future study. Our results also suggest that the DLd regimen might not be suitable for patients with a score of 2, not only because the TTNT would likely be shorter, but also because the OS after DLd treatment would likely be poor.
Our predicting model works only for the DLd regimen, and this model could not be applied to the patients treated with the DBd regimen. This discrepancy might partly be explained by the different mechanisms of the DLd and DBd regimens. It has been reported that the DLd regimen works through the synergistic actions of its constituent agents, while the DBd regimen works through additive actions. 37 From an immunological point of view, this could be interpreted as meaning that the mechanism of the DLd regimen is more reliant on immune cells, particularly monocytes, compared to the DBd regimen. For this reason, our predictive model using monocyte counts (or WBC counts) as an index of immune status would be most suitable for patients treated with the DLd regimen. We could say that our model is not a prognostic model which fits all MM patients, but rather a predictive model for selecting the DLd regimen for relapsed/refractory MM patients. Our predictive model might also be applied to other regimens, such as a daratumumab, pomalidomide, and dexamethasone regimen, or an isatuximab, pomalidomide, and dexamethasone regimen, both of which preferentially rely on immune cells in the manner of the DLd regimen. We would like to address this issue in a future study.
This study also showed that the effectiveness of daratumumab was attenuated by the prior use of elotuzumab or daratumumab treatment. It has been reported that the expression of CD38 was downregulated and the number of immune cells such as NK cells and monocytes was decreased after daratumumab treatment.13–16,38,39 Because the elimination of myeloma cells by daratumumab depends on a complement- or antibody-dependent cell-mediated cytotoxic effect9–12 and NK cells and monocytes are the key player in DLd regimen,13–16 the prior administration of antibody attenuates the effectiveness of daratumumab. Higher monocyte counts, or higher WBC counts may be a prerequisite along with a higher number of immune cells; this should also be confirmed in future studies.
There were several limitations in this study. First, this was a retrospective observational study in which the choice of treatment was made by the individual physicians. Thus, there may have been some bias for selecting daratumumab treatment that we could not include in the multivariate analysis. We will need to substantiate our results by means of analysis in other cohorts. Second, because the data regarding high-risk cytogenetic abnormalities were limited, we might not have sufficiently assessed the impact of high-risk cytogenic abnormalities. Third, we could not distinguish from the database whether patients were refractory to lenalidomide or bortezomib before daratumumab treatment, which could have affected the response to DLd or DBd treatment.
Despite these limitations, to the best of our knowledge, this is the first study to show that the efficacy of the DLd regimen can be predicted by the balance between tumor burden and host immune status.
In conclusion, we proposed a new scoring system using the combination of B2MG plus either monocyte counts or WBC counts to predict the TTNT in patients under the DLd regimen. These scoring systems would be useful for choosing patients who could obtain a benefit from DLd treatment.
Supplemental Material
sj-docx-13-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-docx-13-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-14-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-docx-14-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-15-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-docx-15-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-1-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-1-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-10-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-10-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-11-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-11-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-12-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-12-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-2-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-2-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-3-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-3-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-4-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-4-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-5-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-5-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-6-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-6-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-7-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-7-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-8-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-8-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-9-tah-10.1177_20406207221142487 – Supplemental material for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide
Supplemental material, sj-tif-9-tah-10.1177_20406207221142487 for Monocyte or white blood cell counts and β2 microglobulin predict the durable efficacy of daratumumab with lenalidomide by Yutaka Shimazu, Junya Kanda, Hitomi Kaneko, Kazunori Imada, Ryosuke Yamamura, Satoru Kosugi, Yuji Shimura, Tomoki Ito, Shin-ichi Fuchida, Hitoji Uchiyama, Kentaro Fukushima, Satoshi Yoshihara, Hitoshi Hanamoto, Hirokazu Tanaka, Nobuhiko Uoshima, Kensuke Ohta, Hideo Yagi, Hirohiko Shibayama, Yoshiyuki Onda, Yasuhiro Tanaka, Yoko Adachi, Mitsuhiro Matsuda, Masato Iida, Takashi Miyoshi, Toshimitsu Matsui, Ryoichi Takahashi, Teruhito Takakuwa, Masayuki Hino, Naoki Hosen, Shosaku Nomura, Chihiro Shimazaki, Itaru Matsumura, Akifumi Takaori-Kondo, Junya Kuroda in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
