Abstract
Background:
Eltrombopag (EP) is a small molecule that acts directly on hematopoietic stem cells (HSCs) and megakaryocytes to stimulate the hematopoietic process. Mesenchymal stem/stromal cells (MSCs) are key hematopoietic niche regulators.
Objectives:
We aimed to determine whether EP has any effect on MSC function and properties (especially on their hematopoietic-supporting ability) and if so, what changes (e.g. genome-wide transcriptomic alterations) are induced in MSC after EP treatment.
Design/Methods:
MSCs were isolated from 12 healthy donors and treated with 15 µM and 50 µM of EP for 24 h. The toxicity of the drug on MSCs and their differentiation ability were analyzed, as well as the transcriptomic profile, reactive oxygen species (ROS) and DNA damage and the changes induced in the clonogenic capacity of HSCs.
Results:
The results show that EP also modifies MSC functions, decreasing their adipogenic differentiation, increasing the expression of genes involved in hypoxia and other pathways related to oxygen homeostasis, and enhancing their ability to support hematopoiesis in vitro.
Conclusion:
Our findings support the use of EP in cases where hematopoiesis is defective, despite its well-known direct effects on hematopoietic cells. Our findings suggest that further studies on the effects of EP on MSCs from patients with aplastic anemia are warranted.
Introduction
Eltrombopag (EP) is a small molecule that binds to the transmembrane region of the thrombopoietin (TPO) receptor c-MPL, activating downstream signaling in hematopoietic stem cells (HSCs) and megakaryoblasts. These include the JAK/STAT, PI3 K/AKT and Ras/MAPK pathways.1,2 It does not compete directly in its binding to c-MPL with TPO, so TPO and EP signaling effects are both synergic. 3 In addition, it has a potent ability to bind iron ions, stimulating hematopoiesis through molecular reprogramming 4 and expanding the most immature populations during hematopoiesis. 5
Based on these properties, EP is approved for chronic immune thrombocytopenia (ITP) patients who are refractory to other treatments, thrombocytopenia in patients with chronic hepatitis C and in those with acquired severe aplastic anemia.6–8
EP significantly affects HSCs that are intimately related to the hematopoietic microenvironment that acts as a key regulator of hematopoiesis. Mesenchymal stem/stromal cells (MSCs) regulate the self-renewal, survival, migration, and differentiation of HSCs through cytokine release, the production of growth factors, cell contact interactions and the formation of extracellular matrix. 9 Some data suggest that MSCs from patients with bone marrow (BM) disorders may be altered, and, in a previous study by our group, we observed that MSCs from patients with ITP are functionally abnormal, with reduced proliferation and immunomodulation ability. 10 Other changes have also been described in MSCs from these subjects.11–13 In addition, MSCs may be altered in severe aplastic anemia patients, although the results are controversial.14–16 These suggested alterations in MSCs from patients with ITP or AA make it even more worthwhile to evaluate the effects of EP on healthy-donor-derived MSCs.
The effects of EP on HSCs are well documented, but its potential action on the microenvironment, especially on MSCs, has not so far been addressed in depth. A recent report has analyzed the effects of EP on BM-derived MSCs from ITP patients, showing that EP can reduce MSC apoptosis and increase their proliferation, interfering with intracellular iron signaling in cells from these patients, whereas the changes observed in control cells in that particular report were minimal. 17 Nevertheless, neither the effects of EP on the hematopoietic supporting ability of MSC, nor the genomic changes induced by the drug has been assessed. These are therefore the main objectives of the current work.
Methods
Reagents
Eltrombopag was provided by Novartis Pharmaceuticals. The drug was dissolved in sterile water at a concentration of 10 mM. MSCs were cultured with EP in a final concentration of (10–50 μM). Control cells were incubated in the same medium without the drug for the same periods.
Patient samples
MSCs were isolated from bone marrow (BM) from 12 healthy donors (8 female/4 male) with a median age of 43.5 years (range: 18–60 years). All healthy donors were selected following the same criteria and evaluations as used with our allogeneic hematopoietic stem cell transplant donors. These followed international standards (FACT-JACIE) and consisted of a clinical and biological evaluation to confirm the absence of comorbidities. The donors signed a specific informed consent form approved by the Ethics Committee of the University Hospital of Salamanca. Finally, at the time of BM aspiration, normal BM cytomorphology was confirmed by an experienced hematologist.
BM was aspirated from the iliac crest in sterile conditions under propofol sedation. Mononuclear cells (MNCs) were obtained after Ficoll-Paque density-gradient centrifugation (1.077 g/ml; GE Health-care BioSciences, AB, Uppsala, Sweden), and seeded at 1 × 106 MNC/cm2 on culture flasks with Dulbecco’s Modified Eagle Medium (DMEM) + 10% SBF + 1% penicillin/streptomycin according to previously described methods standardized in our laboratory. 18 MSCs were expanded and sub-cultured until the third passage (MSC-BMp3), and then used for the experimental procedures described below.
Cell viability assays
Cell viability of MSCs was evaluated in triplicate by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) method. 19 Briefly, MSCs were seeded into 96-well culture dishes (8000 cells/well) in the absence (control) or presence of EP at different concentrations (ranging from 10 to 50 µM) for 24 h. Cells were then incubated for 4 h with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Bio-Techne R&D Systems), following the manufacturer’s instructions. Results are expressed as percentages of live cells.
All the subsequent experiments were performed at the selected doses of 15 µM and 50 µM for 24 h, using untreated MSCs as controls.
MSC differentiation ability
Standard multilineage differentiation assays in adipocytic and osteoblastic lineages were performed after culturing MSCs with EP, following the established protocols described in detail 20 and summarized below.
Adipocytic differentiation
In all, 2 × 105 MSCs were seeded and treated with or without EP. After 24 h, the drug was removed, the culture was washed twice with phosphate buffered saline (PBS) and the adipocyte induction medium (MesenCultTM MSC Basal Medium, StemcellTM Technologies) was added for 21 days, changing the medium twice a week. Oil Red staining was performed to assess the extent of adipocytic differentiation.
The number of adipocytes per field formed in each condition was scored weekly (10 fields per sample). The mean of each sample was calculated, the results being summarized as medians.
Osteogenic differentiation
In the same way, 3 × 104 MSCs were stimulated with osteogenic differentiation medium (StemMACS Osteodiff Media, Millenyi Biotec) supplemented with 1% penicillin/streptomycin 1000 U/ml, Gibco) for 10 days. The medium was changed every 3 days. As controls, drug-free MSCs were cultured with differentiation medium, and EP-treated or untreated MSCs were cultured without specific induction medium.
Osteoblasts were stained with alkaline phosphatase, for which purpose NBT (4-nitro blue tetrazolium chloride) and BCIT (5-bromo-4-chloro-3-indolyl phosphate) (Roche) were mixed. Subsequently, the cells were counterstained with 1 ml of hematoxylin (Sigma-Aldrich) for 1 min at RT. Images of adipocytes and osteoblasts were captured under an Olympus BX41 inverted microscope connected to a Canon EOS 650D digital camera.
Mineralized bone quantification
Alizarin red staining was performed to evaluate calcium deposits in MSCs after culture with EP and osteogenic differentiation. 21 After 21 days, the supernatant was removed, and cells were fixed with formaldehyde and stained with alizarin red (40 mM, pH 4.2). Cells were detached with 10% glacial acetic acid and transferred to an Eppendorf tube. Cells were then heated to 85ºC for 10 min and the supernatant was subjected to a colorimetric assay and absorbance was measured in a spectrophotometer at 405 nm.
Reverse transcription real-time PCR analyses of genes involved in MSC differentiation
Total RNA was obtained from MSCs after treatment with EP and differentiated (n = 4) as indicated before using TriPure isolation reagent (Roche Diagnostics; Mainheim, Germany). Reverse transcription was performed using the High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA), according to the manufacturer’s instructions. The genes were quantified using the Step One Plus Real-Time PCR System and TaqMan® Gene Expression Assays (Applied Biosystems, Foster City, CA, USA). Gene expression was normalized as values of 2-ΔCt, where ΔCt = Ctgene–CtGADPH. The genes analyzed are shown in Table 1.
Genes studied to confirm adipogenic and osteogenic differentiation.

Gene expression array
Total RNA of MSCs from four donors treated with 0 µM (control), 15 µM or 50 µM of EP were isolated and purified using RNeasy Kit (Qiagen, Valencia, CA, USA). RNA integrity was confirmed by Agilent 2100 Bioanalyzer (Agilent, Palo Alto, CA, USA). In all, 100 ng of total RNA were amplified, labeled using the WT Plus reagent kit (Affymetrix) and then hybridized to Clariom S human Array (Affymetrix). Washing and scanning were carried out using the Affymetrix GeneChip System (GeneChip Hybridization Oven 645, GeneChip Fluidics Station 450, and GeneChip Scanner 7G).
Array analysis compared untreated MSCs versus MSCs treated with 15 µM and 50 µM of EP, respectively. Differential expression analysis was performed with Limma, a package in R, available in Bioconductor software.
To explore the functions and pathways in which these genes are involved, a search in GeneCards 22 and a functional enrichment analysis with the bioinformatics tool WebGestalt were performed.
Gene expression analysis by RT-PCR
To confirm the results obtained from the gene expression array, total RNA was extracted from treated and control MSCs (n = 7) using Trizol. Gene expression of BNIP-3 (Hs00969291-m1), VEFGa (Hs00900054-m1), HIF1-α (Hs00153153-m1), BDH-2 (Hs00560373-g1), and GADPH (Hs99999905-m1) as a control gene, was analyzed in the same way as previously described.
Functional assays
To evaluate the functional implications of some of the findings observed in the functional enrichment analysis, the following assays were performed.
Intracellular reactive oxygen species (ROS) levels
MSCs treated with EP and control MSCs were trypsinized, washed, and resuspended in 250 µL of DPBS and 5 µL of 2’-7’-dichlorodihydrofluorescein diacetate 10 µM (H2DCF-DA) (Sigma-Aldrich) for 30 min at 37°C, in the dark and under hypoxic conditions. 23 The fluorescence intensity of 2’,7’-dichlorofluorescein (DCF) was measured in a BD FACS CantoIITM flow cytometer. Median DCF fluorescence intensity values, directly related to intracellular levels of ROS, were analyzed with InfinicytTM Software (Cytognos).
DNA damage analysis
MSCs from both experimental groups were incubated with the MuseTM Multi-Color DNA Damage Kit (EDM Millipore Corporation) according to the manufacturer’s protocol. Briefly, 1X assay buffer was added to the cells, which were fixed and permeabilized with fixation buffer and permeabilization buffer, respectively. After washing, they were labeled with two antibodies (20X anti-phospho-histone H2A.X-PECy5, 20X anti-phospho-ATM-PE) which measures the phosphorylation status of the histone pH2A.X and the ATM kinase. Cells were acquired with a BD FACS CantoIITM flow cytometer and results were obtained using the FlowJoTM 10 software (BD Biosciences).
Clonogenic capacity
Mobilized CD34+ progenitor cells were isolated from leukapheresis samples from 5 HD (male/female ratio: 3/2; median age: 34 years, range: 18–54 years) as previously described. 18 CD34+ progenitor cells were labeled using the human CD34 MicroBead Kit (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany) and purified in an AUTOMACs device. The purity and viability of immunomagnetically sorted CD34+ cells were determined by flow cytometry using fluorescein isothiocyanate (FITC)-conjugated CD34 (11-0349-42, eBioscience Inc., San Diego, CA, USA) and 7AAD.
These cells were then co-cultured with MSCs pretreated or not with EP. After 24 h of co-culture, 2 × 103CD34+ were seeded in methylcellulose semisolid MACS medium supplemented with stem cell factor (SCF), granulocyte-macrophage colony-stimulating factor (GM-CSF), granulocyte colony-stimulating factor (G-CSF), IL-3, and IL-6 (Miltenyi Biotec GmbH, Germany) and maintained for 14 days in a humidified atmosphere at 37ºC with 5% CO2. CFU-GM colonies were then scored with an inverted microscope, as previously described. 24
Statistical tests
Statistical analyses were performed with IBM SPSS Statistics v.26 for Windows (IBM Corp., Armonk, NY, USA) using the non-parametric Wilcoxon test to compare the differences between paired results. Graphs were generated with GraphPad Prism 8.4.2 (GraphPad Software Inc., San Diego, CA, USA). Differences were considered statistically significant for values of p < 0.05 (*p < 0.05; **p < 0.01).
Results
Eltrombopag increases the viability of MSCs
The dose–response curves obtained with the MTT assays revealed that EP is not cytotoxic for MSCs obtained from healthy donors. Moreover, EP significantly increased MSC viability at higher concentrations [Figure 1(a)]. The final concentrations selected for all subsequent assays in our study were 15 µM and 50 µM [Figure 1(b)].

Viability of MSCs after exposure to increasing concentrations of Eltrombopag over 24 h. The average absorbance values of control untreated samples were taken as 100%. Data are summarized as medians. (a) In vitro MTT assays of MSCs from HD (n = 7). (b) In vitro MTT assays with selected concentrations for subsequent experiments (n = 17).
Eltrombopag inhibits the adipogenic differentiation of MSCs
After appropriate expansion in the specific differentiation media, all MSC samples from both experimental groups were differentiated into either adipocytes or osteoblasts [Figure 2(a)].

Differentiation ability of MSC treated with Eltrombopag. (a) Adipogenic and osteogenic differentiation, control and differentiated MSCs treated with 15 µM and 50 µM EP, untreated. 10X objective (n = 7). (b) Weekly number of adipocytes per field. Data are summarized as medians (n = 7). (c) RT-PCR genes involved in differentiation. MSCs treated with EP and untreated, differentiated for 21 days with specific media (n = 4).
As shown in Figure 2(b), when MSCs were treated with EP, their adipogenic differentiation capacity significantly decreased, this reduction being significantly greater with EP at 50 µM. There were no significant differences in the expression of the main genes involved in adipogenic and osteogenic differentiation.
Eltrombopag modifies the expression of genes involved in hypoxia, metabolism and other functions
The differential expression analysis of MSCs treated at 15 µM, 50 µM of EP and untreated MSC concentration is shown in Figure 3(a). Most of the differentiated genes did not vary as a function of EP concentration (FDR < 0.05) so the concentration at 15 µM was chosen for this analysis, in which 71 and 25 genes, respectively, were overexpressed and underexpressed relative to the control. Table 2 shows the top 10 most overexpressed genes, which were also found at 50 µM. The GEO accession number for the transcriptome data reported in this paper is GSE202127.
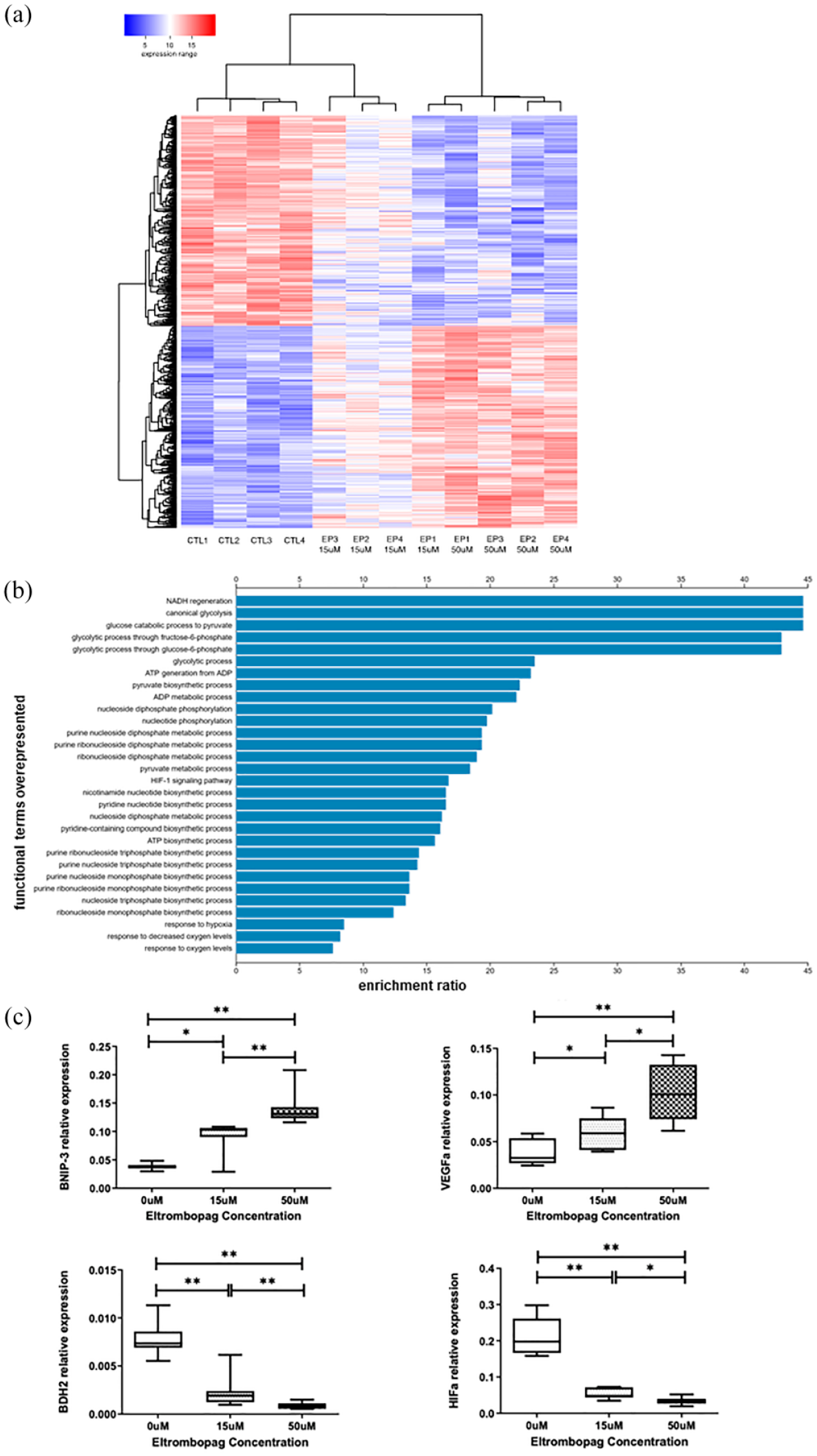
Differential gene expression profiles of MSCs obtained after treatment with Eltrombopag (EP). (a) Heatmap showing the expression profiles of the genes that presented a significant change in MSCs treated with EP 15 µM, EP 50 µM or vehicle control (CTL, 0 µM) (n = 4 donors). The heatmap was constructed after supervised hierarchical clustering of expression profiles of the MSCs and includes 974 genes differentially expressed under the three conditions. A color bar indicates the normalized expression signal (from 1 to 20) over a log2 scale, with blue and red indicating low and high levels of expression, respectively, and white representing values around the median. (b) Functional enrichment analysis was done using the list of the top 200 genes that were upregulated when MSCs were treated with 15 µM of EP. The bar plot includes the main upregulated functions ordered by their enrichment ratio. All these functions correspond to GO-BP terms and KEGG pathways; all of them were significant (FDR < 0.05). (c) Quantitative real-time polymerase chain reaction (qRT-PCR) in independent samples of MSCs isolated from bone marrow: without treatment (0 µM), or treated with 15 µM and 50 µM EP. Results are summarized as medians (7 samples tested).
Top-ten genes overexpressed in Eltrombopag-treated MSC (15 µM).

Some of these genes are involved in the cellular response to hypoxia and negative regulation in apoptosis processes (VEGFA, BNIP3 L, BNIP3), in oxidation and reduction processes (KDM3A), in metabolism and antibiotic biosynthesis pathways (AK4, PGK1), in AMPK signaling pathways (GYS1), as well as in the HIF-1 signaling pathway (PDK1, VEGFA, SLC2A1) and in glycogenesis. Focusing on the top-two underexpressed genes, they were also involved in some of the previous pathways: apoptosis (BDH2) and HIF1α.
Functional enrichment analysis was done using the WebGestalt bioinformatic server (http://www.webgestalt.org/), searching two functional annotation databases (Gene Ontology GO-BP and KEGG pathways) with the list of the top 200 genes that were upregulated when MSCs were treated with EP. The top 30 biological-functional terms obtained in this enrichment were selected (all having an adjusted FDR value p < 0.05) [Figure 3(a)]. The most significant altered functions corresponded to glycolysis, glucose catabolism and the generation of ATP and NADH, suggesting a strong upregulation of cellular energy and respiration metabolism. Moreover, the presence of several biological terms indicating response to oxygen and response to hypoxia confirms that respiration and cellular oxygen homeostasis are affected in these cells by their treatment with EP [Figure 3(b)].
The expression of most of these genes was confirmed by RT-PCR [Figure 3(c)], as was as the underexpression of BDH2 involved in cellular cycle, differentiation, and apoptosis. HIF1α underexpression activates many genes associated with energy metabolism, angiogenesis, apoptosis and other genes involved in increasing oxygen release and facilitating adaptation to hypoxia.
Eltrombopag does not increase intercellular ROS level or affect DNA damage
Analysis showed that the drug did not increase the intracellular levels of ROS, although ROS levels were lower at higher doses [Figure 4(a)]. Likewise, the results of DNA double-strand breaks analyses do not support the presence of cell damage in MSCs treated with the EP [Figure 4(b)].

Functional assays: (a) median fluorescence intensity of the intracytoplasmic ROS levels (n = 7), (b) percentage of cells with DNA damage (n = 5), and (c) absolute number of granulo-monocyte colonies (n = 11).
Eltrombopag increases in vitro hematopoietic supporting ability of MSCs
More granulo-monocytic colonies (CFU-GMs) were induced from healthy CD34+ cells that had been previously co-cultured with MSCs treated with EP than from those that had been co-cultured with MSCs in the absence of the drug [Figure 4(c)].
Discussion
The originality of the present work lies in it addressing, for the first time, the effects of EP on the hematopoietic supporting ability of healthy MSCs, which are key cells in the regulation of hematopoiesis. The role of EP in stimulating hematopoiesis in bone marrow failure has been clinically validated.1,4,5,11,25,26 Nevertheless, besides the well-known direct actions of this drug on hematopoietic progenitor cells, our study helps show how EP exerts some interesting effects on the stromal compartment that can also favor the hematopoietic process. 2
We have shown here for the first time how EP can act on MSCs, increasing their proliferation and osteogenic differentiation capacity while decreasing their adipogenic differentiation ability. In addition, expression arrays revealed the overexpression of genes involved in pathways related to hypoxia, inhibition of apoptosis, energy metabolism and hematopoiesis. These were confirmed by RT-PCR. Functional assays also identified a decrease in oxidative stress in MSCs treated with high doses of the drug. Furthermore, the drug did not induce any significant DNA damage within the cell. Finally, clonogenic assays confirmed that MSCs treated with EP were better able to support hematopoiesis in vitro. Although some of these observations are based on small sample sizes, there were enough cases to enable significant statistical differences to be detected, and to validate the genetic profile study by RT-PCR. An interesting finding of our work is that EP at a concentration of 50 µM increases the viability of MSCs measured by the MTT assay, whereas at 15 µM the viability is similar to that of untreated MSCs. The selection of the concentrations to be used in our experiments requires further comment. Most analyses of the role of EP in megakaryopoiesis and hematopoiesis in vitro use concentrations ranging from 0.1 to 30 µg/ml in samples from healthy donors or patients. 27 In the current study, we initially tested concentrations from 10 to 50 µM. In subsequent studies, we decided to focus primarily on the 15-µM concentration, since it is widely used and is similar to the peak concentration of the drug in plasma of healthy subjects. 28 However, we also wanted to evaluate the effects of EP at 50 µM, since, as we have just mentioned, this concentration significantly increased the viability of MSCs. In a recent study, Di Paola et al. 17 have assessed the effects of 6 µM of EP on MSCs of ITP pediatric patients, showing that the drug is able to increase viability and proliferation, while decreasing the release of IFN-gamma and IL-6 in MSCs from these subjects. The effects of the drug on healthy-donor-derived MSCs (also of pediatric age) were minimal or absent at the evaluated concentration.
The recent description that EP may exert its effects on MSCs from ITP patients by interfering with intracellular iron signaling is evidence of the existence of alternative mechanisms of action of this drug on different cell types. 17 Kurokawa et al. 29 described that EP has the capacity to inhibit the proliferation of non-myeloid tumor cell lines through other mechanisms that do not involve the TPO receptor. Other authors have reported that EP does not increase the growth of bone marrow cells from patients with MDS/AML. 27 This selective finding about proliferation depending on the cell type is very intriguing and warrants further study.
When assessing one of the functional hallmarks of human MSCs, such as proliferation and multilineage differentiation, our studies revealed that EP increased MSC proliferation and differentiation with other bone marrow niche cells. The intimate interaction between HSCs and the BM microenvironment is essential for maintaining and regulating the behavior and properties of HSCs throughout an individual’s lifetime.30–32 Among the minimal criteria for defining ISTC, MSCs should be capable of differentiating in vitro into adipocytes and osteoblasts. 33 In our study, MSCs treated with EP differentiated to osteocytes and adipocytes, although we observed an imbalance in the osteogenic/adipogenic differentiation ability compared with untreated cells. There were significantly fewer adipocytes in the EP-cultured cells. In addition, a tendency toward the stronger expression of genes involved in mineralization and osteogenesis, such as SPARC, SPP1 and RUNX2, as well as a decrease in the level of expression of AEBP1. It has been widely described that the number of adipocytes is inversely related to hematopoietic activity. 34 Autophagy or the liposomal degradation pathway regulates cellular homeostasis. This process has been linked to various age-associated diseases, including cancer. There is evidence that autophagy may also play an important role during cell differentiation and in the maintenance of hematopoietic stem progenitor cells (HSPCs) and in decreasing cellular stress.35,36 Data confirmed overexpression of genes such as BNIP3 that may stimulate autophagy in the stroma and prevent MSCs aging. Its overexpression could be related to the reduction in ROS. Osteogenic differentiation in MSCs from bone marrow is favored by autophagy. 37 In addition, Lee and Joe 38 have demonstrated that hypoxic conditions can enhance the therapeutic efficacy of MSCs by increasing the level of secretion cytokines and autophagy.
Several genes overexpressed in our study, such as BNIP3, PGK1, TMEM45A and VEGFa, are known to be involved in response to hypoxia. Under hypoxic conditions, the maintenance of hematopoietic stem cells becomes stricter, so that the clonogenic capacity, proliferation and differentiation of these cells and MSCs increases. 39 Moreover, under these same conditions there is an increase in the level of expression of the VEGF-a in BM-MSCs. 40 Hematopoiesis and osteogenesis are both increased, and adipogenic differentiation is reduced, under hypoxic conditions.2,41 Our results have shown that EP induces hypoxic conditions in MSCs, and that these facilitate the development of new therapeutic targets.
Chemically, EP has a central metal group that allows it to bind intracellular iron, thereby modifying iron homeostasis. 42 This drug causes molecular changes in HSCs that are capable of restoring intracellular iron concentrations and reducing oxidative stress levels. 4 Although further studies are needed to understand the effects of EP on reactive oxygen species, we have observed a drop in oxidative stress levels in treated MSCs. Iron plays an important role in hematopoiesis. However, its accumulation in certain diseases such as myelodysplastic syndromes or in patients undergoing transplantation increases the levels of intracellular ROS, thereby increasing oxidative stress and decreasing the hematopoietic capacity of the microenvironment and the degree of differentiation of the CD34+ HSCs. 43 Several authors2,44 have studied ROS levels in malignant hematopoietic cells and revealed the depletion of these hematopoietic progenitors in patients. The use of EP in preclinical trials reduces the extent of intracellular ROS accumulation, intracellular free iron levels, and the level of oxidative stress in several cell types.4,10,45 Hernández-Sánchez et al. 46 showed that ITP patients who did not respond to EP treatment presented several genes involved in activating the heme pathway. This could be related to an increase in the levels of intracellular iron, oxidative stress (ROS) and ineffective hematopoiesis. These effects were not present in drug-responsive patients. The latter findings are all consistent with our results, even though the cell type assessed is different.
Previous studies in MSCs have shown that excessive ROS can alter self-renewal, favor adipogenesis, and inhibit osteogenesis, proliferation and immunomodulatory properties.47,48 Our results suggest that EP can reverse all these effects. In the process of differentiating hematopoietic progenitor cells toward megakaryocytic lineage, MSCs are involved and are able to secrete several factors, such as thrombopoietin, which is import for forming megakaryocytes.12,49,50
In our study, EP did not produce DNA damage in MSC. EP inhibited DNA replication in cells from Ewing’s sarcoma. 26 However, several articles conclude that it would be worthwhile to explore further the drug’s ability to stimulate DNA damage repair in abnormal cells.
Finally, the role of MSCs in supporting hematopoiesis is essential for regulating this process.9,51 Our observation that EP-treated MSCs can increase the clonogenic capacity of CD34+ HSCs is probably the most important finding of this study. It provides evidence in favor of using the drug in cases where hematopoiesis is defective, besides their well-known direct effects on hematopoietic cells, and implies that further studies on the effects of EP on MSC from patients with aplastic anemia are warranted.
Footnotes
Acknowledgements
The authors would like to thank Rebeca Ortega of the University Hospital of Salamanca for her technical assistance in the differentiation assays, and Phil Mason for his support with improving the English language of the manuscript.
