Abstract
Pomalidomide is a potent immunomodulatory agent that is currently a standard of care backbone for the treatment of multiple myeloma (MM) patients in the relapsed/refractory setting after exposure to lenalidomide and a proteasome inhibitor. The present review addresses current knowledge regarding the clinical use of pomalidomide in relapsed myeloma patients. Pomalidomide has direct myeloma cell tumoricidal effects by activating proteasomal degradation of Ikaros and Aiolos transcription factors and also indirect effects by modulation of immune responses, interaction with bone marrow stromal cells, and inhibition of angiogenesis. It is approved by regulatory authorities as doublet combination with dexamethasone but four more triplets are also approved for this setting. Many ongoing trials are evaluating the pomalidomide–dexamethasone backbone with newer anti-myeloma class agents or in quadruplet combinations. Pomalidomide–dexamethasone is currently one of the powerful tools available for use in the relapsed/refractory MM setting. Insights into the synergistic immunomodulatory effects of pomalidomide and other anti-myeloma agents and the mechanisms that overcome clonal resistance will potentially allow targeted use of triplet combinations at each relapse.
Keywords
Introduction
In the last several years, the introduction of novel agents has revolutionized the therapeutic landscape for multiple myeloma (MM). 1 Immunomodulatory drugs (IMiDs), proteasome inhibitors (PIs), anti-CD38 monoclonal antibodies, and more recently, anti-B-cell maturation antigen (BCMA) monoclonal antibodies, in triplet or quadruplet combinations in the frontline or relapsed/refractory setting have improved and deepened responses. They have greatly increased overall survival (OS) in these patients. Despite advances in the therapeutic field of MM, nearly, all patients will relapse regardless of the frontline regimen they receive.2,3 With each relapse, clonal genetic heterogenicity and drug resistance develop.4–6 Treatment decisions have become more complex with multiple parameters to be taken into consideration at each step of the decision-making process. Pomalidomide (Pom) is an IMiD, which received approval by the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) in 2013 for MM patients in the relapsed/refractory setting after exposure to lenalidomide and a PI (usually bortezomib). Pomalidomide plus low-dose dexamethasone (Pd) is considered standard of care as the backbone for several triplet novel-agent combinations approved for use in this setting.7,8 The present review covers the scientific and clinical trial data that demonstrate the central role of pomalidomide in the treatment of relapsed/refractory patients with MM.
Mechanism of action
Pomalidomide, an IMiD, is a glutamic acid derivative ((4-amino-2-(2,6-dioxopiperidin-3-yl) isoindoline-1,3-dione). It is a chemical analogue of thalidomide and lenalidomide with a more potent and less toxic profile.9,10 It is administered orally, absorbed rapidly and is metabolized in the liver through CYP3A4 and CYP1A2 11 (Table 1). It has significant anti-myeloma activity; it has direct tumoricidal and immune-stimulating effects.
Main characteristics of pomalidomide.

EMA, European Medicines Agency; FDA, US Food and Drug Administration; MM, multiple myeloma.
Cereblon (CRBN), a component of the E3-ubiquiting ligase complex, is the primary molecular target of all immunomodulatory agents. 12 Binding to CRBN leads to ubiquitination and proteasomal degradation of the transcription factors Ikaros (IKZF1) and Aiolos (IKZF3) which are involved in B and T-cell development. 13 There is downregulation of interferon regulatory factor 4 (IRF4) and MYC, which has direct tumoricidal effects. Several immunomodulatory mechanisms have been described; there is downregulation of proinflammatory cytokines, such as tumor necrosis factor alpha (TNF-α), interleukin 12 (IL-12), macrophage inflammatory protein 1 alpha (MIP1α), and IL-614,15 and enhancement of natural killer (NK) cell cytotoxicity directly and stimulation of T-cells via increased levels of IL-2, interferon gamma (IFN-γ).16,17 There is also inhibition of angiogenesis via a decrease in vascular endothelial growth factor (VEGF) levels, 18 downregulation of the transcription factor PU.1, reduction of bone resorption, 19 and inhibition of the cell cycle and induction of apoptosis.20,21 Pomalidomide acts on the bone marrow stromal cells (BMSC), indirectly affecting MM cells.12,22 The antitumor and immune-stimulating characteristics are distinct to those of lenalidomide; it has more potency against cereblon, different degradation kinetics and a different gene activation profile. In vitro studies have demonstrated activity in lenalidomide-resistant cell lines and animal models. Clinical studies have confirmed its efficacy in patients refractory to lenalidomide.23,24 Immune profile analysis suggests that overcoming lenalidomide resistance is most likely linked to continued immune stimulation.
Initial clinical trial data and drug profile
Pomalidomide–dexamethasone
Pomalidomide was tested initially as a single agent in a continuous (days 1–28) regimen and the maximum tolerated dose (MTD) was found to be 2 mg per day with dose-limiting toxicities being neutropenia (60% of patients had grade 3/4 neutropenia) and deep vein thrombosis (DVT). 25 Its efficacy as single agent is limited in RRMM patients, but the synergistic effects with dexamethasone are significant. Almost two-thirds of patients and 40% of lenalidomide refractory patients achieved a response among 60 patients with RRMM with pomalidomide 2 mg/day plus low-dose dexamethasone.26,27
In 2013, the feasibility of a discontinuous schedule for Pd (d1–21 in a 28-day cycle) with a MTD of 4 mg was demonstrated. 28 This dosing schedule was assessed in a phase II trial, in 221 heavily pretreated patients (median number of prior lines 5) who were randomized to receive Pd (n = 113) versus pomalidomide (Pom) (n = 108) alone; 29 Overall response rate (ORR) was 33% versus 18% and median progression free survival (PFS) 4.2 months versus 2.7 months (HR = 0.68) in the Pd and Pom arms, respectively. Grade 3/4 neutropenia was observed in 41% and DVT in 2% of patients (patients received aspirin as thromboprophylaxis). Refractoriness to lenalidomide (Len) or to both Len and bortezomib did not affect outcomes. Pd had promising activity in patients with t(4;14) or deletion 17p, but both ORR and median PFS were lower than those in the intention-to-treat (ITT) population.
The continuous versus discontinuous regimen in dual refractory patients were compared in a phase II randomized study with similar results. 30 Two sequential phase II trials compared the 2 mg with the 4 mg Pom dose in patients RR to lenalidomide and bortezomib, and no advantage was demonstrated for 4 mg. 31 The 4 mg/day days 1–21 in a 28-day cycle plus dexamethasone has become the standard of care due to its favorable efficacy/safety profile. Preclinical 27 and phase I/II clinical trial data have demonstrated that the Pd combination has significant activity in RRMM patients, regardless of refractoriness to lenalidomide or bortezomib. 31
Following on from phase I/II clinical data, a phase III randomized controlled trial (RCT) (MM-003) evaluated the efficacy of Pd versus high-dose dexamethasone (HDD) at 320 mg per cycle in 455 dual refractory patients (bortezomib and lenalidomide). 7 Patients who progressed in the HDD arm crossed over to the other arm. The median number of prior lines was 5; ORR was 31% in the Pd arm versus 10% in the HDD arm and at 10-month median follow-up median PFS benefit was 2 months and median OS benefit was 5 months for the Pd arm. Importantly, the benefit was retained independent of age (> 75 years), high-risk cytogenetics [17p deletion and t(4;14)] 32 or the type and number of prior treatment lines. 33
Achieving at least minor response (MR) had a significant impact on outcome; patients who achieved at least MR had a median PFS of 7.4 months and a median OS of 17.2 months versus 2.3 and 7.5 months, respectively, for patients with less than MR. Grade III–IV neutropenia was observed in 48% of patients and 30% had grade III–IV infection. However, the rate of DVT was low (2%) for patients who achieved adequate thromboprophylaxis (mostly aspirin). The prospective quality of life (QoL) assessment demonstrated an advantage for patients in the Pd arm.34,35 Given the very favorable results of the phase III RCT MM-003, both the FDA and the EMA approved pomalidomide plus low-dose dexamethasone for patients with MM who are dual refractory in 2013.
MM-010 (STRATUS) was another phase III trial in RRMM patients who had received lenalidomide, an alkylator, and a PI, and were refractory or RR to the last line of therapy. Patients (n = 682, median number of prior lines 5) achieved an ORR of 32.6% and median PFS was 4.6 months and median OS 11.9 months. 36 Results from a more recent phase II trial (MM-014) designed to evaluate Pd versus daratumumab–Pd confirmed an ORR of 32.1% in 56 patients who entered cohort A and received Pd after two prior lines of treatment and immediately following refractoriness to lenalidomide. 37
Patients with renal impairment
A population pharmacokinetics model of pomalidomide in relapsed/refractory multiple myeloma (RRMM) patients with various degrees of renal impairment (RI) has demonstrated that moderate to severe renal dysfunction does not affect clearance or plasma exposure. In patients on dialysis, pomalidomide should be administered after hemodialysis sessions. 38 No dose adjustment is therefore necessary with RI. A sub-analysis of the MM-003 trial demonstrated similar efficacy/safety in patients with moderate RI. 34 The MM-013 trial evaluated Pd combination in relapsed MM patients with estimated glomerular filtration rate (eGFR) <45 ml/min/1.73 m2, including patients on dialysis. The trial included 81 patients in three cohorts; cohort A with moderate RI (eGFR 30– <45 ml/min/1.73 m2); cohort B with severe RI (eGFR <30 ml/min/1.73 m2); and cohort C with severe RI that requires hemodialysis. ORRs were 39.4%, 32.4%, and 14.3% with median duration of response of 14.7 months, 4.6 months, and not estimable, respectively. With a median follow-up of 8.6 months, median OS was 16.4, 11.8, and 5.2 months, respectively. The rate of grades III–IV hematologic adverse events and treatment discontinuation was more frequent in cohort C, but the safety profile was acceptable among all three groups concluding that Pd is effective and safe in RRMM patients independent of renal function. 39 However, responses and survival rates are lower compared with patients not receiving dialysis.
High-risk patients
Patients with extramedullary disease (EMD) are considered high-risk MM patients with poorer prognosis and outcomes. Pomalidomide has significant activity in these patients with an ORR demonstrated in RRMM with EMD who received Pd. 40 IMiDs also have activity in the central nervous system (CNS) as they cross the blood–brain barrier and should be included in the treatment plan of patients with RRMM who have CNS involvement.41–43 Data from the MM-003 trial demonstrated that the benefit of Pd remains in the presence of high-risk cytogenetics [17p deletion and t(4;14)]. Fifty patients with relapsed MM and 17p deletion or t(4;14) received Pd (4 mg, d1–21) until progression in a phase II trial by the IFM group dedicated to myeloma patients with adverse cytogenetic features.
Time to progression (TTP) was 7.3 versus 2.8 months, duration of response 8.3 versus 2.4 months and ORR 32% versus 15% in patients with 17p deletion and t(4;14) respectively, demonstrating a particularly favorable impact of this combination in patients with 17p deletion. 44
Toxicity profile
The most common toxicities observed are hematologic (neutropenia grade III–IV in almost 50% of patients, anemia and thrombocytopenia in 30–50%) and two-thirds of patients develop infections.29,44 It should be noted, however, that the population is usually heavily pretreated and infection-prone. DVT and venous thromboembolism (VTE) is a well-recognized complication associated with IMiDs. Thromboprophylaxis is mandatory and when used systematically, the rate of VTE is < 5%. Current National Comprehensive Cancer Network (NCCN) guidelines (2020) recommend the use of aspirin for patients who have none or one risk factor for VTE and prophylactic doses of direct oral anticoagulants or low molecular weight heparin or vitamin-K antagonists for patients with more than one risk factor who receive IMiDs. VTE risk assessment algorithms are available. No dose adjustments are required for patients > 75 years, but the dose of dexamethasone is reduced. Patients with hepatic impairment (bilirubin > 1.5 × upper limit of normal) have been excluded from clinical studies. Based on pharmacokinetic studies, there is a modest effect on the PKs of pomalidomide. 45 Based on the drug’s Summary of Product’s Characteristics, no dose adjustment is required for patients with hepatic impairment as defined by the Child–Pugh criteria.
Triplet combinations with pomalidomide–dexamethasone backbone
Despite the efficacy of Pd, the ORR is low, around 30–35%, with a median PFS of 4 months. Combining Pd with other anti-myeloma agents in triplet drug combinations has improved greatly on ORR, PFS and OS for patients in the RRMM setting. The therapeutic options have greatly expanded and led to multiple FDA/EMA approvals for triplet combinations with a pomalidomide backbone. Pd is currently approved for use in the relapsed/refractory setting in combination with the proteasome inhibitor (PI) bortezomib, the antiCD38 monoclonal antibodies daratumumab and isatuximab and the SLAM7 monoclonal antibody elotuzumab. In addition to these triplet regimens, the Pd backbone is currently being evaluated with numerous other agents with anti-myeloma activity. The key clinical trials with results that have led to regulatory approvals for Pd-based combinations are presented in Table 2. Table 3 shows ongoing clinical trials that assess newer Pd-based combinations.
Key phases II and III clinical trials of pomalidomide-based combinations in relapsed/refractory multiple myeloma patients.
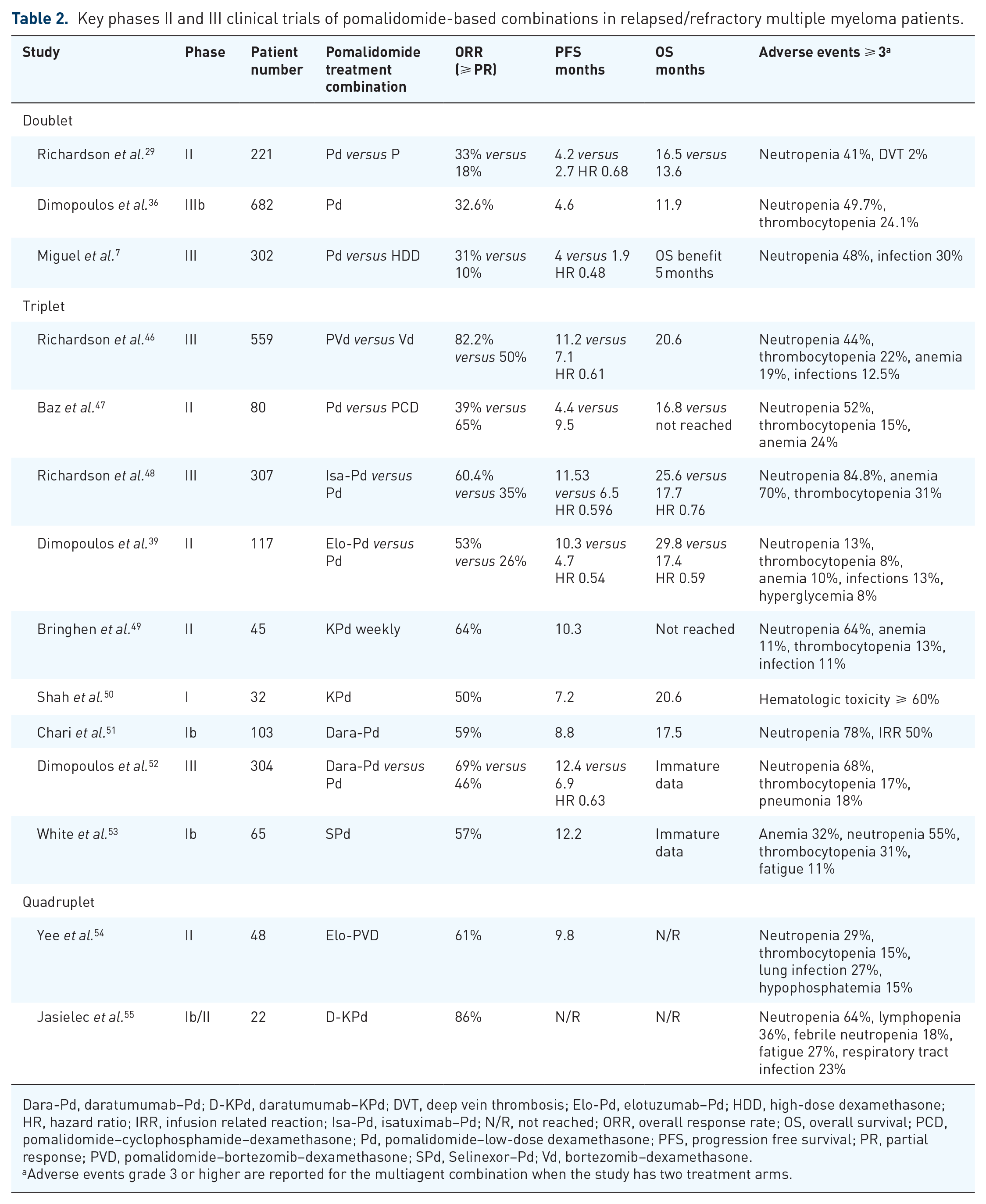
Dara-Pd, daratumumab–Pd; D-KPd, daratumumab–KPd; DVT, deep vein thrombosis; Elo-Pd, elotuzumab–Pd; HDD, high-dose dexamethasone; HR, hazard ratio; IRR, infusion related reaction; Isa-Pd, isatuximab–Pd; N/R, not reached; ORR, overall response rate; OS, overall survival; PCD, pomalidomide–cyclophosphamide–dexamethasone; Pd, pomalidomide–low-dose dexamethasone; PFS, progression free survival; PR, partial response; PVD, pomalidomide–bortezomib–dexamethasone; SPd, Selinexor–Pd; Vd, bortezomib–dexamethasone.
Adverse events grade 3 or higher are reported for the multiagent combination when the study has two treatment arms.
Main ongoing clinical trials of pomalidomide–dexamethasone based combinations.

Bela, belantamab mafodotin; Dara-Cd, daratumumab–cyclophosphamide/dexamethasone; Dara-Pd, daratumumab–Pd; Elo-KPd, elotuzumab-KPd; Elo-PVd, elotuzumab–pomalidomide/bortezomib/ dexamethasone; Ide-Cel, idecabtagene vicleucel; Isa-KPd, isatuximab–KPd; Ixa-Pd, ixazomib–Pd; KPd, carfilzomib–Pd; MM, multiple myeloma; NivoPd, nivolumab–Pd; PCD, pomalidomide/cyclophosphamide/dexamethasone; Pd, pomalidomide/dexamethasone; PVD, pomalidomide–bortezomib–dexamethasone; RRMM, relapsed or refractory multiple myeloma; SDd, selinexor–daratumumab/dexamethasone; SKd, selinexor–carlfizomib/dexamethasone; SKPd, selinexor–KPd; SPd, selinexor–Pd; SPEd, selinexor–elotuzumab–Pd; SPVd, selinexor–PVd; tal, talquetamab; tec, teclistamab.
Prior exposure to lenalidomide and a PI.
Prior exposure to a PI, an IMiD and anti-CD38 monoclonal antibody.
Pomalidomide–dexamethasone and alkylating agents
Pomalidomide–dexamethasone and cyclophosphamide (PCD) is among the first triplet combinations evaluated and is widely used although not being formally approved by regulatory agencies. Larocca et al. 56 2013 in a phase I/II study assessed the combination of pomalidomide–prednisone and cyclophosphamide in patients with RRMM. The MTD was 2.5 mg for pomalidomide (d1–21) in combination with prednisone and cyclophosphamide both at 50 mg every other day for six cycles in total followed by Pom–prednisone maintenance. In 55 patients who received the MTD, ORR was 51% with a median PFS of 10.4 months and toxicity was similar to that observed with Pd alone. The median number of prior treatment lines was 3 and only 46% of the patients were lenalidomide refractory. Dexamethasone is preferred to PCD is the preferred combination. A phase II RCT study compared the standard discontinuous Pd regimen +/– cyclophosphamide 400 mg orally on days 1, 8, and 15 (n = 70). 47 All patients were lenalidomide refractory and the median number of prior lines was 4. The ORR was 64.5% versus 38.9% and the median PFS 9.5 versus 4.4 months in the PCD versus Pd arms, respectively. Hematologic toxicity reported was similar in both arms. Trudel et al. 57 reported real-life single-center data for 49 RRMM patients who received PCD. ORR was 76%, median PFS was 7.3 months and median OS not reached. Nine patients discontinued treatment due to toxicity and grade ⩾ 3 cytopenias were reported in 37% of patients. An ongoing phase III RCT (NCT03143049) is assessing this combination in Asian patients.
Pomalidomide–dexamethasone and PIs
The synergistic action of IMiDs and PIs has been demonstrated in multiple trials. Pomalidomide–bortezomib–dexamethasone (PVD) was EMA/FDA approved in 2019 based on the results of the phase III trial OPTIMISMM. Initial data from a phase I/II trial in patients who were lenalidomide refractory and PI-refractory established that a 21-day cycle at an MTD of 4 mg for pomalidomide (d1–14) and 1.3 mg/m2 for bortezomib (days 1, 4, 8, and 11 for cycles 1–8; days 1 and 8 for cycles ⩾9). 58 The combination has also been evaluated in a 28-day cycle dosing regimen in lenalidomide-refractory RRMM patients. The MTD was established at 4 mg for pomalidomide days 1–21, 1.3 mg/m2 IV/SC for bortezomib at days 1, 8, 15, 22, and dexamethasone weekly 40 mg. 59 Fifty patients entered phase II and 86% achieved a response; 100% of high-risk patients achieved a response and median PFS was 13.7 months.
The phase III registration trial OPTIMISMM enrolled 559 patients who had received 1–3 prior treatment lines, including a lenalidomide-containing regimen. The dosing schedule was based on data from the phase I/II study (21-day cycle). Patients were randomized to bortezomib–dexamethasone +/– pomalidomide. Median PFS was 11.2 versus 7.1 months (HR = 0.61) for PVD and VD, respectively, at a median follow-up of 15.9 months. ORR was 82.2% in the PVD versus 50% in the VD arm; responses were deeper and more durable in the PVD arm. Patients with high-risk cytogenetics had poorer responses but PVd reduced the risk of progression or death, indicating that the triplet combination may partly overcome adverse prognosis in these patients. Grade III/IV neutropenia was 42% versus 9% and grade III/IV infections were seen in 31% versus 18%, respectively. 46
Carfilzomib is an irreversible second-generation PI. A phase I study assessed carfilzomib–Pd (KPd) and established an MTD at 4 mg d1–21 for pomalidomide, carfilzomib 20/27 mg/m2 days 1–2, 8–9, and 15–16, and dexamethasone 40 mg/week. 50 The population (n = 32) was heavily pretreated (median number of prior treatment lines was 6), and 60% were refractory to an IMiD and a PI. The ORR was 50% and responses were seen in patients with 17p deletion. KPd weekly was assessed in another Italian phase I/II trial 49 which enrolled 57 patients, 42 in phase II. The MTD was established at 20/27 mg/m2 d1, 8, 15 for carfilzomib. ORR for patients in phase II was 64% and responses were rapid and were not affected by previous refractoriness to lenalidomide or bortezomib. At median follow-up of 12.8 months, median PFS was 10.3 months, and OS not reached.
A phase I/II trial (NCT02185820) assessed the combination of pomalidomide (4 mg) and dexamethasone (20 mg weekly) plus carfilzomib 20 mg/m2 IV cycle 1, day 1 followed by carfilzomib at different dose levels (27, 36, 45, or 56 mg/m2) based on the different cohorts of the study (d1, 8, 15) for eight induction cycles in total in patients with MM primary refractory or relapsed/refractory to lenalidomide. Patients received the combination until progression or intolerance with carfilzomib at the MTD established in the phase I part of the trial. Recruitment is complete and results are awaited. A phase II study for patients at first or second relapse who are refractory to lenalidomide and the most recent line of treatment assesses carfilzomib at the 20/56 mg/m2 dose on days 1, 8, 15 for cycles 1–12 and d1, 15 for cycles ⩾ 13 dosing schedule in combination with pomalidomide. The study has completed recruitment and results are awaited (NCT04191616).
Ixazomib is an oral PI and the combination of ixazomib–Pd offers an all-oral combination for MM patients at relapse. A phase I/II study, assessed the safety and efficacy of ixazomib–Pd in lenalidomide/PI refractory patients and the results support further investigation of this triplet combination. 60 The MTD used was 4 mg for Pom, 4 mg for ixazomib, and 20/40 mg dose for dexamethasone. The ORR was 51.7%, median PFS was 4.4 months, and median OS was 34.3 months for the 29 patients who entered phase II. No safety signals were raised and the triplet combination had side-effects consistent with the individual drug profiles. In 2021, results were released from a phase II trial comparing ixazomib–dexamethasone (4 mg) versus Pd (4 mg) in patients refractory to lenalidomide/Pi. Median PFS was 7.1 versus 4.8 months in ixa–dex versus Pd (HR = 0.847), but the difference was not statistically significant. Grade ⩾ 3 adverse events were seen in 64% of patients in the ixa–dex versus 81% in the Pd arm. 61 Another dosing schedule of the combination is currently being assessed; Ixazomib twice weekly d1–14, Pomalidomide d1–14 and dexamethasone twice weekly in a 21-day cycle (NCT04094961).
A phase Ib study evaluated the combination of the oral PI oprozomib and Pd in RRMM patients. Patients (n = 31) received oprozomib once daily on days 1–5 and 5–19 at a 150 mg/starting dose (5/14 schedule) or on consecutive days weekly (2/7 schedule, 210 mg starting dose) and pomalidomide at 4 mg d1–21. 62 The ORR was 70.6%, but the MTD for oprozomib was not defined and there were concerns about safety and the oprozomib formulation used. An ongoing phase Ib study is evaluating new formulations in this combination.
Pomalidomide–dexamethasone and monoclonal antibodies
Daratumumab is an anti-CD38 humanized monoclonal IgGκ antibody with very potent anti-myeloma activity. In 2017, the FDA approved the combination of IV daratumumab, pomalidomide and dexamethasone in RRMM patients who have received at least two prior treatment lines including lenalidomide and a PI. The approval was granted based on the phase Ib study (MMY1001, EQUULEUS). 51 A total of 103 patients were enrolled (median number of prior lines 4) and received daratumumab 16 mg/kg in combination with Pd (4 mg d1–28 and 40 mg weekly). The ORR was 59% and, among complete responders, 27% achieved minimal residual disease (MRD) negativity. The median time to response was 1 month and median duration of response 13.6 months. At a median follow-up of 13.1 months, median PFS was 8.8 months and median OS was 17.5 months. Regarding the safety profile, there was a 50% daratumumab-specific infusion-related reaction rate and the incidence of grade III/IV neutropenia was higher than expected for Pd alone, at 78%.
A phase II trial assessed the safety and efficacy of Pd and daratumumab in patients with RRMM after one to two prior treatment lines and exposure to lenalidomide. 63 Daratumumab was administered IV as per the drug’s label at the time of study conduction. ORR was 77.7% and at a median follow-up of 17.2 months, median PFS was not reached. Grade III/IV neutropenia was seen in 62.5% of patients and grade III/IV infections were reported in 31.3%. The same group published data regarding immunomodulation with this triplet combination from the aforementioned trial. 64 A potential mechanism for enhanced immune-mediated cytotoxicity is proposed in which the daratumumab mediated decrease in NK cells is offset partially by the effects of pomalidomide on the remaining NK cell pool of activated and proliferating NK cells. Pomalidomide has tumoricidal activity, enhances T-cells and acts in a complementary manner to daratumumab, which eliminates CD38 + T-cells.
The phase III RCT trial Apollo (EMN14) compared Pd with Pd plus daratumumab in patients with RRMM and released results in 2021. 52 Patients (n = 304) had received at least one prior line of treatment, were exposed to both a PI and lenalidomide, and were refractory to lenalidomide if only one previous line of therapy was received. Daratumumab was administered at 16 mg/kg as an intravenous (IV) infusion (Dara IV) or 1800 mg subcutaneously (Dara SC) at the standard regimen of administration; weekly for 8 weeks, every 2 weeks for 16 weeks, and then every 4 weeks thereafter. Pomalidomide was given at 4 mg d1–21 and dexamethasone at 40 mg weekly (20 mg for patients > 75 years). At a median follow-up of 16.9 months, the median PFS was 12.4 versus 6.9 months (HR = 0.63) for the Dara–Pd and Pd arms, respectively. The ORR was 69% in the Dara-Pd arm versus 46% in the Pd arm. Regarding the most common adverse events, grade III/IV neutropenia was seen in 68% versus 51%, thrombocytopenia in 17% versus 18%, and pneumonia in 15% versus 8% of patients in the Dara-Pd and Pd arms, respectively.
Based on the results of EMN14, EMA extended the marketing authorization of daratumumab in the SC formulation to include the combination with Pd for patients who have received at least one prior treatment line including a PI and lenalidomide and are refractory to lenalidomide or at least two prior treatment lines which included lenalidomide and a PI and progress on or after the last treatment. The SC formulation of daratumumab gained approval for use in combination with Pd (and other doublets) by the FDA in 2021 also based on the results of the phase III EMN14 trial. There is an active phase II trial (NCT03841565) which is evaluating the Dara–Pd combination in RRMM patients who have had previous Dara exposure (have achieved at least PR and have relapsed on or 60 days from the end of treatment). The study aims to assess the efficacy of daratumumab retreatment in combination with Pd.
Resistance to daratumumab may be reversed with the addition of IMiD to daratumumab even when patients are refractory to both drug classes 65 and retaining a daratumumab containing backbone is associated with clinically relevant activity (ORR of 43% and median PFS 5 months). 66
Data from in vitro studies demonstrated that the combination of isatuximab with pomalidomide increased cytotoxicity and CD38-MM cell lysis. The combination of isatuximab, another anti-CD38 monoclonal antibody, and Pd was initially assessed in a phase Ib study. A total of 31 patients were enrolled and received Isa at a dose of 10 mg/kg (weekly for cycle 1 and then every 2 weeks); the ORR was 64.5% and median PFS was 17.6 months with no safety signals raised. 67 The combination was then assessed in the phase III ICARIA-MM study which compared isatuximab in combination with Pd (ISaPd) versus Pd in RRMM patients. 68 Patients had received at least two prior treatment lines which included lenalidomide and a PI. Isatuximab was administered at 10 mg/kg weekly for the first cycle and on days 1, 15, thereafter, plus Pom 4 mg d1–21 and dexamethasone 40 mg per week (or 20 mg for patients > 75 years) versus Pd alone. At a median follow-up of 11.6 months, 307 patients were enrolled and median PFS was 11.5 months versus 6.5 months in the Isa-Pd versus the Pd arms, respectively (HR = 0.596). Infusion reactions, upper respiratory tract infections, and diarrhea were the most common adverse events reported. In the study update in 2021, at a median follow-up of 35.5 months, Isa-Pd was found to significantly improve time-to-next treatment and PFS2 and there was a strong trend in OS benefit in the Isa-Pd arm (24.6 versus 17.7 months, HR = 0.76). 48
In 2020, the EMA and FDA approved the combination of isatuximab-Pd for MM patients who have received at least two previous treatments, including lenalidomide and a PI. In a subgroup analysis, the PFS benefit with Isa-Pd was seen across both the two to three prior treatment line groups (12.3 versus 7.8 months) and the > 3 prior lines of therapy group (9.4 versus 4.3 months). The advantage was also retained independent of refractoriness to a PI or double-refractoriness or refractoriness to lenalidomide in the last line of therapy. ORR was also better in the Isa-Pd arm across all groups. 69 The PFS and ORR benefit in the Isa-Pd is independent of age and remained across all three age groups in another subgroup analysis. 70 In another prespecified subgroup analysis in patients with RI (eGFR < 60 ml/min/1.73 m2), PFS, ORR, and renal response rates were better in the Isa-Pd arm; renal response rates were 71.9% in Isa-Pd versus 38.1% in the Pd arm. In addition, pharmacokinetics were comparable between subgroups and no dose adjustment was recommended for patients with impaired renal function. 71
Elotuzumab is humanized immunoglobulin G1 immunostimulatory monoclonal antibody with anti-myeloma activity, which targets signaling lymphocytic activation molecule F7. A phase II randomized study (ELOQUENT-3) assigned 60 patients to receive elotuzumab–Pd and 57 to receive Pd. 72 Elotuzumab was administered at 10 mg/kg weekly for cycles 1 and 2 and at 20 mg/kg on day 1 for ⩾ cycle 3. Patients had received at least two prior lines of therapy which included lenalidomide and a PI and had relapsed/refractory disease. Median PFS was 10.3 months in the Elo-Pd arm versus 4.7 months in the control group (HR = 0.54) after a minimum follow-up period of 9.1 months. ORR was 53% in the triplet arm versus 26% in the doublet combination arm. Neutropenia grade III/IV was seen in 13% versus 27%, anemia in 10% versus 20% in the EloPd and Pd arms, respectively, and in each cohort, 65% had infections of any grade. This triplet combination has a favorable safety profile and is an option across all age populations. Based on the results of the phase II study, the triplet combination of elotuzumab–Pd received EMA and FDA approval in 2019 for use in patients with RRMM who have received at least two prior treatment lines which has included a PI and lenalidomide.
Other clinical trial data from pomalidomide–dexamethasone-based combinations
The PD-1/PDL-1 axis has a central role in the regulation of immune responses. Plasma cells in MM patients have increased PD-L1 expression and T-cells and NK cells in patients with MM show increased PD-1 expression. Based on these findings, there is a strong rationale for activity of checkpoint inhibitors in MM. IMiDs seem to enhance the effect of checkpoint inhibitors in MM. The combination inhibits the proliferative effects of BMSCs on myeloma cells and allows for reversal of immunoparesis due to the BMSCs. 73 KEYNOTE-183 is a phase III trial that evaluated Pd in combination with pembrolizumab, but the study was halted because at the interim analysis the benefit–risk profile of the triplet combination was unfavorable. CheckMate 602 is a phase III trial with three arms; RRMM patients are randomized to Pd versus Pd–nivolumab versus EloPd–nivolumab. The study is active but not recruiting, 170 patients have enrolled and no results have been released yet (NCT02726581).
Belantamab mafodotin (BLENREP) is a first-in-class antibody drug conjugate that consists of an afucosylated humanized monoclonal IgG1k antibody specific for BCMA and maleimidocaproyl monomethyl auristatin F (mcMMAF). It is FDA and EMA approved as monotherapy for patients with RRMM who have received at least four prior therapies, including a PI, an IMiD, and an anti-CD38 monoclonal antibody. In an attempt to move the drug earlier in the treatment algorithm of RRMM patients, it is currently being evaluated as monotherapy or combination treatment against Pom-based combinations. DREAMM3 is a phase III clinical trial comparing single-agent belantamab mafodotin to Pd in RRMM patients (NCT04162210) who have received a PI and lenalidomide. This trial is designed to assess safety and efficacy of belantamab mafodotin against one of the standard of care options for this patient population. DREAMM8 (NCT04484623) is another ongoing phase III randomized trial comparing the combination of belantamab mafodotin–Pd to bortezomib–Pd for patients that are relapsed/refractory and have been exposed to a PI and lenalidomide. Patients at first relapse are eligible to enter the trial.
Pomalidomide has also been assessed in combination with filanesib, a kinesin spindle protein inhibitor. 74 Median PFS was 7 months and OS was 19 months, 51% of the patients achieved ⩾ partial response (PR), but the toxicity profile was not favorable, as there was a high rate of hematological toxicity.
Pomalidomide–dexamethasone and histone deacetylase inhibitors
Pomalidomide has also been combined with histone deacetylase (HDAC) inhibitors in the relapsed setting. A phase Ib/II trial evaluated ACY-1215 (Ricolinostat), an HDAC6 protein inhibitor in combination with pomalidomide and dexamethasone in patients with RRMM. (NCT01997840) The study started in 2014, but no results have been released yet. Another HDAC inhibitor, AR-42 was combined with pomalidomide and dexamethasone in a phase Ib trial. The study is complete, but no results have been released (NCT02569320).
Pomalidomide–dexamethasone and selinexor
Another agent that has received FDA/EMA approval in the RRMM setting is selinexor, an oral selective inhibitor of nuclear export protein, XPO1. The selinexor–Pd (SPd) combination was assessed in the multi-arm STOPM study (arm 1). 53 Selinexor was evaluated at 60, 80, or 100 mg weekly or 60 or 80 twice weekly in combination with Pd. Results were released in 2021 based on the interim analysis for 65 patients with a median number of three prior lines of therapy (1–10). The combination’s recommended phase II dose was selinexor at 60 mg per week, Pom 4 mg (d1–21) and dexamethasone 40 mg per week. Adverse events ⩾ grade 3 included anemia (32%), neutropenia (55%), thrombocytopenia (31%), and fatigue (11%). Among patients who were naïve or not-refractory to Pom, ORR was 57% and median PFS was 12.2 months (and was better for patients treated with the RP2D). In Pom-refractory patients, ORR was 44%. Based on these results, SPd is safe combination for patients with RRMM with satisfactory efficacy. A phase III is awaited to compare Pd with SPd in patients exposed to lenalidomide, a PI and an anti-CD38 monoclonal antibody 53 (NCT02343042). In the same study, SPd in combination with other anti-myeloma agents are being evaluated: SPd–bortezomib (arm 4), SPd–elotuzumab (arm 9) and SPd–daratumumab (arm 11).
The SCOPE phase I/II trial compares selinexor–Pd–carfilzomib to selinexor–Pd. (NCT04764942). In the phase IIb study (NCT04661137), selinexor in various dosing levels is being assessed in combination with carfilzomib, pomalidomide or daratumumab in patients who are refractory in each of these agents. In the selinexor–Pd arm, selinexor is administered at 60 mg days 1, 8, and 15, and pomalidomide at 4 mg d1–21. Finally, there is a phase III randomized, open-label controlled trial that has not started recruitment yet. It evaluates SPd versus Elo–Pd in RRMM patients (NCT05028348).
Quadruplet combinations with pomalidomide–dexamethasone backbone
The question of whether early and combinatory administration of multiple class agents to allow synergistic activity will improve PFS and OS for patients with MM at first relapse remains unanswered to date. Multiple ongoing trials are currently addressing this question and assessing efficacy and safety of quadruplet regimens in the early relapse setting. The Pd backbone is commonly included in these combinations for patients with exposure to lenalidomide and a PI.
A phase II trial (NCT02718833) with a single group assignment is assessing elotuzumab in combination with pomalidomide, bortezomib and dexamethasone in MM in patients who have received at least one prior line of therapy that includes both lenalidomide and a PI. Prior pomalidomide exposure was permitted. 54 Results were released in 2019; 48 patients were enrolled, median number of prior lines of therapy was 3. At a median follow-up of 18.8 months in 46 evaluable patients, median PFS was 9.8 months, and ORR was 61%; ORR for patients with previous pomalidomide was 43%, previous carfilzomib 46% and previous anti-CD38 46%. ORR was 74% and PFS not reached for patients with 1 prior line of therapy. Grade ⩾ 3 adverse events, included neutropenia (29%), thrombocytopenia (15%), lung infection (27%), and hypophosphatemia (15%).
In another phase Ib/II study, the combination of KPd with daratumumab (D-KPd) has been assessed for safety and efficacy in two sequential cohorts: KPd and D-KPd. Patients after at least one relapse were included (patients at first relapse were lenalidomide refractory). Dosing was as follows: carfilzomib 20/27 mg/m2 cycles 1–8 on d1, 2, 8, 9, 15, and 16, and cycle ⩾ 9 on d1, 2, 15, and 16, pomalidomide 4 mg d1–21 and daratumumab as per standard schedule. The results released in 2020 included 22 evaluable patients with 1 median number of prior lines. After four cycles (in the ITT analysis), 86% achieved ⩾ PR, 46% ⩾ stringent complete response (sCR) and 55% MRD negativity. Interestingly, the presence of high-risk cytogenetics did not affect response. Median PFS was not reached at 20-month follow-up. Adverse events ⩾ grade 3 included neutropenia in 64%, lymphopenia in 36%, febrile neutropenia 18%, fatigue 27%, and respiratory tract infection 23%. 55 The combinations of isatuximab–KPd (NCT04287855), elotuzumab–isatuximab–KPd (NCT04835129) and daratumumab–CPd (NCT03841565) are currently being evaluated in phase Ib/II trials.
Pomalidomide–dexamethasone backbone in combination with bispecific T-cell engagers and chimeric antigen receptor T-cell antigens
Bispecific T-cell engagers (BiTEs) and chimeric antigen receptor (CAR) T-cell antigens are rapidly gaining ground in the treatment of patients with MM, mainly in the relapsed/refractory setting. Ongoing clinical trials are evaluating the safety and efficacy of these agents either compared with or combined with standard anti-myeloma combinations for patients in the relapsed setting. Given the central role of pomalidomide in this setting, pomalidomide-based combinations are included in the comparator arms of multiple ongoing trials.
A phase Ib study (NCT04108195) is currently evaluating two BiTE antibodies in combinations that include daratumumab and pomalidomide. The study randomizes RRMM patients to receive SC daratumumab plus teclistamab (a BiTE that binds BCMA and CD3) or SC daratumumab plus talquetamab (a GPRC5D × CD3 BiTE) or SC daratumumab plus talquetamab plus pomalidomide or daratumumab plus teclistamab plus pomalidomide. Teclistimab is being evaluated in another multi-arm phase Ib study in RRMM. In arm A teclistamab is being assessed for safety and efficacy in combination with Pomalidomide and daratumumab (NCT04722146). Given the high efficacy of daratumumab–pomalidomide–dexamethasone combination in the RRMM safety it is currently one of the comparator arms in a phase III randomized trial evaluating teclistamab in combination with sc daratumumab (MajesTEC-3). The other comparator arm is daratumumab–bortezomib. Patients who have received one to three prior lines of therapy including a PI and lenalidomide are included (NCT05083169).
AMG701 is another anti-BCMA BiTE. T-cell-dependent cellular cytotoxicity of MM cells with or without MM-supporting cells from the bone marrow microenvironment was evaluated in a recent study. 75 The combination of AMG701 with lenalidomide resulted in superior antitumor activity compared to either monotherapy supporting a combination of AMG701 with IMiDs. 76 AMG701 is being evaluated as monotherapy or in combination with pomalidomide plus/minus dexamethasone in a phase I/II open-label study. Patients enrolled have received at least three prior lines of therapy and have exposure to all approved and available therapies for MM (NCT03287908).
The pomalidomide–daratumumab combination is also being evaluated in combination with CAR T-cell antigens in ongoing clinical trials. A phase I/II clinical trial (KarMMa-7) is currently evaluating the safety and preliminary efficacy of the CAR T-cell idecabtagene vicleucel (bb2121, Ide-Cel). Arm C of the study evaluates Ide-Cel with daratumumab + pomalidomide + low-dose dexamethasone versus pomalidomide + bortezomib in patients with RRMM. KarMMa-3 is a phase III trial randomized, Open-label Study comparing Ide-Cel to standard regimens for patients with RRMM. Pomalidomide-based combinations are in two of the comparator arms, one with daratumumab and dexamethasone and the other with elotuzumab–dexamethasone. Patients who had prior exposure to daratumumab, an IMiD, and a PI and had received two to four prior regimens were included in the study (NCT03651128).
Conclusion
Pomalidomide–dexamethasone is considered the standard of care in the relapsed/refractory setting for MM patients either alone or in combination with other anti-myeloma agents. Four different triplet Pd-based combinations are currently FDA/EMA approved for these patients. There is a multitude of ongoing clinical trials with newer agents with anti-myeloma activity and more Pd-based combinations are expected to be added to the treatment armamentarium in the relapsed setting in the future; quadruplet combinations or regimens of Pd combined with belantamab mafodotin, BiTEs, and CAR T-cells. Pomalidomide has manageable and well-understood toxicity, is an oral agent, and does not require renal adjustment. Its efficacy remains high for high-risk disease, EMD and it has some activity in the CNS. It has a unique immunomodulatory profile and further insight into the synergistic mechanisms between pomalidomide and the other anti-myeloma class agents will potentially allow us to identify in the future the optimal drug combinations for particular patient and disease characteristics. We expect pomalidomide to remain one of the central players in the RRMM setting in the foreseeable future. As the therapeutic approach in MM becomes increasingly targeted and tailored to patient and disease characteristics, we expect different Pd-based combinations to emerge as preferable options for different MM patients in the relapsed/refractory setting. Head-to-head comparisons with lenalidomide are lacking and are not expected any time soon. Given the favorable efficacy and safety profile of lenalidomide and the efficacy of pomalidomide in lenalidomide-refractory patients, pomalidomide is not expected to move to the frontline setting.
Footnotes
Author contribution(s)
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.A.D. has received honoraria/personal fees from Amgen, BMS, Celgene, GSK, Janssen, and Takeda. E.T. declares consultancy and honoraria from BMS, Janssen, Celgene, Takeda, Genesis Pharma, and Amgen. M.G. declares consultancy and honoraria from Amgen, Karyopharm, Genesis Pharma, Janssen, and Takeda. D.F. declares no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
