Abstract
Ibrutinib, the first in class of the oral covalent Bruton tyrosine kinase (BTK) inhibitors, has profoundly changed the treatment landscape of chronic lymphocytic leukemia (CLL). The phase III RESONATE and RESONATE-2 trials first demonstrated the superiority of ibrutinib over ofatumumab in the relapsed/refractory setting and over chlorambucil in older patients with de novo disease. The phase III ECOG–ACRIN trial extended these results to young, fit patients, demonstrating a significant survival advantage to ibrutinib plus rituximab over fludarabine, cyclophosphamide, and rituximab. Similarly, the Alliance trial demonstrated the superiority of ibrutinib over bendamustine with rituximab as frontline in elderly patients. Challenges with ibrutinib include toxicity, development of resistance, and need for indefinite therapy. The second generation BTK inhibitor acalabrutinib may cause less off-target toxicity. The ELEVATE TN trial demonstrated the superiority of acalabrutinib with or without obinutuzumab over chlorambucil and obinutuzumab as frontline therapy for elderly or comorbid patients. Promising early results from the phase II CAPTIVATE and CLARITY trials, which combined ibrutinib with venetoclax, suggest a future role for minimal residual disease (MRD) testing to determine treatment duration. The ongoing phase III GAIA/CLL13, ECOG EA9161, Alliance A041702, CLL17, and [ClinicalTrials.gov identifier: NCT03836261] trials will assess various combinations of ibrutinib/acalabrutinib, venetoclax, and anti-CD20 antibodies. These trials will answer key questions in the treatment of CLL: should novel agents in CLL be used in combination or sequentially? What is the best frontline agent? Can treatment be safely stopped with BTK inhibitors? Can undetectable MRD be used to determine treatment duration? In this review, we will discuss these and other aspects of the evolving role of BTK inhibition in CLL.
Keywords
Background
Chronic lymphocytic leukemia (CLL) is characterized by clonal proliferation of mature B-cells in the bone marrow, peripheral blood, and lymphoid organs. It is the most common leukemia in the Western world, occurring predominantly in the elderly. 1 The clinical course of patients with CLL is highly variable. Among patients who require therapy, mutated IGHV gene and absence of TP53 aberrancy are typically associated with excellent response rates and deep remissions with chemo-immunotherapy (CIT) regimens.2–4 Unfortunately, many patients have adverse features, or are unable to tolerate CIT due to advanced age or medical comorbidities. Recently, novel targeted agents have dramatically changed the treatment landscape of CLL, particularly high-risk disease. Among the most promising are the Bruton’s tyrosine kinase (BTK) inhibitors.
BTK is a member of the TEC family of non-receptor protein kinases, which is an integral component of the B-cell receptor (BCR) signaling cascade.5,6 Upon BCR crosslinking, src family protein kinases (LYN, FYN, and BLK) interact with the intracellular tyrosine activation motifs located on CD79A/B, leading to activation of spleen tyrosine kinase (SYK). SYK then recruits a plasma membrane-associated signaling complex, which incorporates BTK as well as adaptor molecules [such as B cell linker protein (BLNK)]. The complex then activates Ras and phospholipase C-γ2. Ras propagates the signal down to extracellular regulated kinase (ERK), while phospholipase C-γ2 leads to activation of mitogen-activated protein kinase (MAPK) kinases and transcription factors including Myc and NFκB. 7 Thus, BTK activation triggers multiple signaling pathways involved in cell proliferation and survival, motility, and migration, as well as cytokine production and antigen presentation.8,9
Ibrutinib and acalabrutinib, which are United States (US) Food and Drug Administration (FDA)-approved for the treatment of CLL, both demonstrate excellent clinical activity, as do several other second generation BTK inhibitors in various phases of clinical development.10–15 Current research has focused on combining BTK inhibitors with other targeted agents in time limited regimens,16–22 in some instances using minimal residual disease (MRD) assays to determine treatment duration.16,18,20 BTK inhibitors rarely produce undetectable MRD (uMRD) as single agents, and, although this measure does not correlate with survival in patients treated with ibrutinib, ongoing studies might help determine whether it may be necessary to achieve uMRD before safely stopping therapy.4,22 In contrast, uMRD is strongly associated with progression-free survival (PFS) and overall survival (OS) in patients treated with venetoclax and CIT.23,24 Given the indolent nature of CLL, reliable surrogate endpoints such as MRD are critical to advancing care. The following review covers the successes and challenges with currently available BTK inhibitors; summarizes major trials of BTK inhibitors in combination with CIT, anti-CD20 antibodies, and targeted agents; and highlights key ongoing clinical trials. Moving from disease control to cure in CLL will require the successful marriage of BTK inhibitors with some or all of the agents described below.
How ibrutinib became standard of care in de novo and relapsed CLL
Ibrutinib, an oral covalent inhibitor of BTK, was the first BTK inhibitor approved for patients with previously treated CLL by the FDA in 2014 based on objective response rate (ORR) of 71% in a phase Ib/II trial (see Table 1 for key results of clinical trials of ibrutinib and other BTK inhibitors). 25 These impressive results were replicated in the phase III RESONATE trial, which compared indefinite treatment with ibrutinib with six cycles of ofatumumab – an anti-CD20 monoclonal antibody. Patients in the ibrutinib arm had received a median of three prior lines of therapy. 11 Long-term follow up, published after a median of 44 months, confirmed a PFS and OS advantage of ibrutinib over ofatumumab. 26 At this time point, 46% of patients still remained on ibrutinib therapy. In an open label trial in patients with relapsed/refractory (R/R) CLL with del(17p), ibrutinib demonstrated a PFS rate of 63% at 24 months. 27 In a phase II trial conducted at the National Institute of Health (NIH), the estimated PFS rate at 5 years was 74% in patients with TP53 aberration. 28 Thus, ibrutinib activity in R/R CLL, including patients with high-risk disease, was convincingly demonstrated in multiple trials.
Key clinical trials of BTK inhibitors.
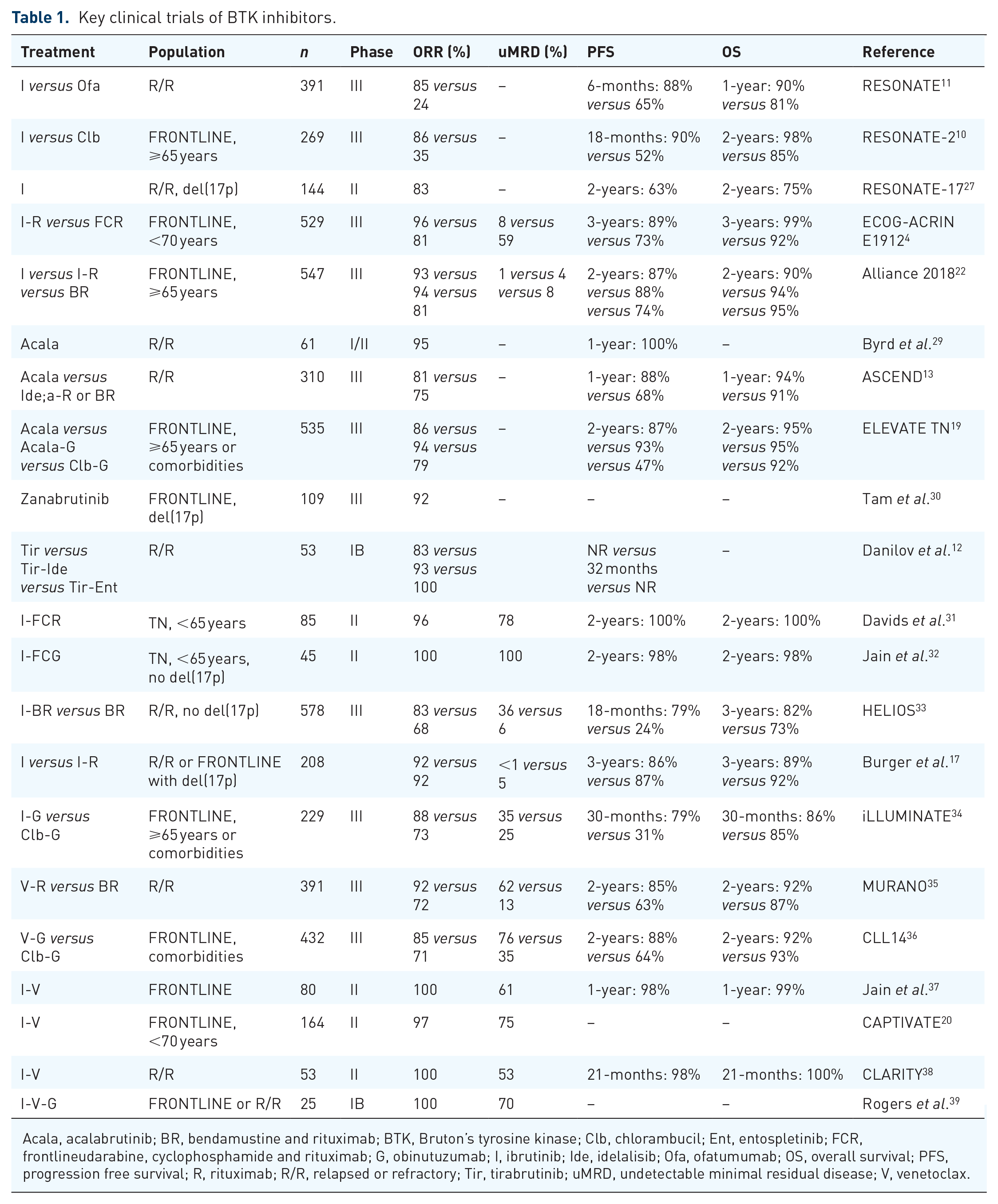
Acala, acalabrutinib; BR, bendamustine and rituximab; BTK, Bruton’s tyrosine kinase; Clb, chlorambucil; Ent, entospletinib; FCR, frontlineudarabine, cyclophosphamide and rituximab; G, obinutuzumab; I, ibrutinib; Ide, idelalisib; Ofa, ofatumumab; OS, overall survival; PFS, progression free survival; R, rituximab; R/R, relapsed or refractory; Tir, tirabrutinib; uMRD, undetectable minimal residual disease; V, venetoclax.
In the frontline setting, the phase III RESONATE-2 trial compared indefinite ibrutinib with chlorambucil in patients over 65 years old without del(17p). Treatment with ibrutinib significantly improved PFS and OS in this study, 10 and this benefit was confirmed in a subsequent analysis after a median follow up of 60 months. 40 Advancing ibrutinib therapy beyond the elderly and R/R disease settings, the phase III ECOG-ACRIN trial investigated indefinite ibrutinib in combination with six cycles of rituximab versus fludarabine, cyclophosphamide, and rituximab (FCR) as frontline therapy for younger (<70 years) fit patients. 4 Treatment with ibrutinib was associated with superior PFS and OS. With the exception of patients with mutated IGHV (M-IGHV), the survival advantage of ibrutinib–rituximab over FCR was seen in all subgroups. Long-term toxicities of the FCR regimen, including immune suppression and secondary malignancies, and difficulty to identify the exact patient population among those with mutated IGHV who derive long-term benefit from FCR, limits use of this regimen in clinical practice. The phase III Alliance trial compared ibrutinib with an alternative standard CIT regimen – bendamustine with rituximab (BR) – in elderly (median age 71 years) patients with previously untreated CLL. 22 Importantly, this trial had both ibrutinib and ibrutinib plus rituximab arms. Both ibrutinib arms demonstrated a significant PFS benefit compared with BR, with no difference in OS. Notably, there was no difference in either PFS or OS with the addition of rituximab to ibrutinib. To date, all phase III trials of ibrutinib have demonstrated a significant PFS advantage. Ibrutinib has improved outcomes in CLL to an extent that many prognostic markers and risk-scores relevant in the CIT era are no longer applicable or need to be revised.41–44
Something new: second generation BTK inhibitors, does selectivity improve outcomes?
Adverse events leading to ibrutinib discontinuation have emerged as an obstacle to indefinite therapy, 45 and it is perceived that the second generation BTK inhibitors may be better tolerated.12,14,29 Acalabrutinib is the most thoroughly studied second generation selective BTK inhibitor in clinical development. A phase I/II study of acalabrutinib in R/R CLL demonstrated an ORR of 95%, with all patients with del(17p) achieving a response. 29 Acalabrutinib does not target epidermal growth factor receptor (EGFR) or ITK signaling, and no cardiac arrhythmias were observed in this study, although hypertension of any grade was observed in 20% of patients, with grade 3–4 hypertension in 7%. Additionally, retrospective and prospective data show acalabrutinib to be well tolerated in ibrutinib-intolerant patients with CLL.46,47 These results suggest that increased selectivity towards BTK could reduce cardiac toxicity. The phase III ASCEND trial compared acalabrutinib therapy versus investigators choice of idelalisib with rituximab or BR in R/R CLL. 13 Acalabrutinib demonstrated superior outcomes with an estimated 1-year PFS rate of 88% with acalabrutinib versus 68% in the investigators choice arm. Acalabrutinib has also been studied in combination with obinutuzumab,19,48 most notably in the phase III ELEVATE TN trial described below. Whether acalabrutinib is a better BTK inhibitor is being addressed in a head-to-head trial of acalabrutinib versus ibrutinib in previously treated, high-risk – either del(17p) or del(11q) – CLL [ClinicalTrials.gov identifier: NCT02477696].
Zanabrutinib is another selective BTK inhibitor in clinical development. In a phase I trial, zanabrutinib produced an ORR of 96% in patients with CLL. 14 No grade 3–4 cardiac toxicities were observed. One patient developed a grade 3 subcutaneous hemorrhage. The ongoing phase III SEQUOIA trial included an additional non-randomized arm of treatment-naïve CLL patients with del(17p) who received indefinite zanabrutinib therapy and in whom preliminary results have been published. 30 The ORR was a promising 92%. Hypertension occurred in 6.4% of patients and major bleeding occurred in 2.8%. Cardiac arrhythmias were not reported. Similar to acalabrutinib, a head-to-head comparison with ibrutinib in R/R CLL is ongoing [ClinicalTrials.gov identifier: NCT03734016].
Yet another selective BTK inhibitor, tirabrutinib (formerly ONO-4059) has also been studied in CLL. 49 In a phase IB study, tirabrutinib was administered as either single agent or in combination with either idelalisib (phosphoinotiside-3 kinase inhibitor) or entospletinib (spleen tyrosine kinase inhibitor). 12 In the single agent tirabrutinib arm, ORR was 83% and the drug was well tolerated.
The results of the ongoing head-to-head trials of selective BTK inhibitors and the long-term follow-up safety data with these agents will be critical in determining the utility of these agents relative to ibrutinib. Although more data is needed, early results appear promising. Patients with comorbid medical conditions have worse outcomes with ibrutinib, and may be a population that would particularly benefit from a second generation BTK inhibitor. 50
Something blue: challenges with BTK inhibition
Despite the significant advance in the treatment of CLL achieved with ibrutinib and other BTK inhibitors, challenges with tolerance and resistance remain. In an analysis of over 600 patients treated at multiple US medical centers, 41% of patients discontinued ibrutinib after a median follow up of 17 months, adverse events were the most common cause of ibrutinib discontinuation occurring in over half of patients, followed by CLL progression. 45 In a multicenter retrospective analysis published by our group, 21% of ibrutinib-treated patients discontinued therapy due to adverse events and high burden of comorbidities, as assessed by the cumulative illness rating scale (CIRS), was associated with increased rates of adverse events leading to treatment cessation. 50 In contrast to these results, discontinuation rates due to adverse events in patients treated on clinical trials ranged recently from 6% to 12%.26,28 Whether the difference in therapy discontinuation rates between research and “real world” patients is due to differences in relative fitness between these populations, or due to provider comfort and familiarity with managing ibrutinib toxicity, is an important unanswered question. Some of the more common adverse events associated with ibrutinib are briefly summarized below and reviewed elsewhere in more detail. 51
New onset atrial fibrillation is a common complication of ibrutinib therapy, which may be due to inhibition of C-terminal src kinase. 52 Long-term follow up of a phase II trial conducted by the NIH found that 21% of patients treated with ibrutinib developed atrial fibrillation, while only 6% discontinued therapy due to adverse events of any kind. 28 A pooled analysis of patients with either CLL or mantle cell lymphoma found a 13% incidence of atrial fibrillation. 53 Rates of atrial fibrillation increased with longer exposure to ibrutinib, indicating that risks persist with continued therapy. Despite the relative frequency of atrial fibrillation in this population, 85% of patients with atrial fibrillation were able to continue ibrutinib therapy. Together, these studies suggest that, while atrial fibrillation is common, it is manageable in most patients. Multiple risk scores have been developed to predict treatment emergent atrial fibrillation, with older age and comorbid heart disease repeatedly identified as predictive factors. 54 Ventricular arrythmias and sudden cardiac death have been observed, albeit rarely, in patients on ibrutinib. 55
Hypertension is another common adverse event. One retrospective study found an incidence of 78% after a median follow up of 30 months. 56 Importantly, development of hypertension in this cohort was associated with increased rates of other cardiovascular events. Minor bleeding is also commonly observed with ibrutinib, but severe hemorrhage has also been described. 57 Use of standard of care antiplatelet medications or anticoagulation for concomitant cardiac disease or atrial fibrillation may increase risk of bleeding, albeit risks of major hemorrhagic events remain low despite concurrent use. 58
Initial clinical trials evaluated ibrutinib in patients who were poor CIT candidates.10,11,22 While treatment with ibrutinib is associated with the emergence of comorbid conditions that contribute to therapy discontinuation and death, increased burden of comorbidities before treatment initiation is also associated with poor outcomes. As mentioned above, research done by our group and others has demonstrated that the presence of comorbidities, measured by CIRS, negatively impacts survival and tolerance of therapy with ibrutinib.50,59 We conducted an analysis to identify which comorbidities are most predictive of outcomes in patients CLL (of whom 58% received ibrutinib). Interestingly, neither cardiac disease nor hypertension were factors strongly associated with shortened survival in these patients. 60 While it may be intuitive that patients with comorbidities have worse outcomes with oncologic drug treatments, we have also shown that comorbidities have minimal impact on idelalisib therapy. 61 This further justifies exploration of the role of comorbidities in patients treated with second-generation BTK inhibitors.
The development of resistance to ibrutinib is another challenge with indefinite therapy as resistance to BTK inhibition increases with time.62,63 One possible solution is that time-limited combination regimens of BTK inhibitors with CIT, venetoclax or anti-CD20 antibodies could limit the development of resistance to these agents and allow their reintroduction either as single agents or in combination at time of relapse. Cost is another concern with these new agents,64,65 and time-limited combination regimens could also reduce financial toxicity, or exacerbate it, depending on duration of remission and other factors. The estimated lifetime cost of CLL therapy in the United States is over $600,000, with out-of-pocket expenses projected to increase by over 500%. 64
It is important to note that the benefit of therapeutic advances in CLL has not been experienced equally by all patients. African American patients with leukemia in general, and CLL in particular, still have shorter survival compared with other groups. 66
Finally, the COVID-19 global pandemic has important implications for the treatment of CLL. Some early experiences in CLL patients infected with the SARS-CoV-2 virus have been reported. 67 How to best manage patients with CLL who become infected with the SARS-CoV-2 virus has not been studied rigorously, but some expert guidance is also available. 68 Whether or not to continue BTK inhibitor is a pertinent question to this review. Most providers would continue ibrutinib in patients with mild-to-moderate COVID-19. Interestingly, preliminary data suggests that BTK inhibitors may be protective against severe pulmonary disease associated with COVID-19. 69 More recent data from a multicenter retrospective study of CLL patients diagnosed with symptomatic COVID-19 infection, whether on treatment or observation, demonstrated a case fatality rate of 33%. 70 Other groups have identified advanced age and recent treatment as risk factors for death in CLL patients who contract COVID-19 infection. 71
Something old: combining chemo-immunotherapy with ibrutinib
Several key trials performed by the German CLL study group and others established combination CIT as the standard of care in CLL. The CLL8 trial demonstrated both a PFS and OS advantage with FCR versus fludarabine and cyclophosphamide, 3 and CLL10 confirmed FCR as a superior regimen in younger patients with previously untreated CLL. 2 Notably, no difference in survival was observed between FCR and BR in patients over 65 years old. Excellent long-term survival was seen predominantly in patients with M-IGHV and no TP53 aberrations. In one study of frontline FCR, the PFS rate at 12.8 years of follow up was 54% versus 9% in patients with M-IGHV and unmutated IGHV (U-IGHV), respectively. 24 In this study, post-treatment uMRD was also found to be predictive of long-term survival, suggesting that a subset of patients with favorable disease could achieve long-term disease control with frontline FCR regimen and that post treatment uMRD could identify these patients early.
Multiple trials have assessed the benefit of adding ibrutinib to CIT regimens. In an open label phase II trial, ibrutinib was combined with FCR (iFCR) in young fit patients with previously untreated CLL. 31 In patients with an objective response after induction, ibrutinib was continued for 2 years, at which time those with uMRD discontinued therapy. A complete response (CR) with uMRD 2 months after completing all treatment was observed in 33% of patients. A similar phase II trial of ibrutinib with fludarabine, cyclophosphamide, and obinutuzumab (iFCG) in previously untreated CLL with M-IGHV and no TP53 aberration also assessed a time-limited treatment strategy based on MRD status. 32 Patients who achieved a CR with uMRD after three cycles continued on ibrutinib for 1 year with three additional cycles of obinutuzumab. All others continued on obinutuzumab and ibrutinib for 1 year. Patients with a CR and uMRD after 1 year stopped all therapy. Impressively, all of the 45 patients included in this trial were able to stop therapy. This is an important population to study as the ECOG–ACRIN trial did not demonstrate a survival advantage with ibrutinib-rituximab versus FCR as frontline therapy for patients with M-IGHV – a CLL subgroup with potential for a functional cure when treated with FCR. Whether the addition of time-limited ibrutinib to FCR or FCG can increase rates of long-term survival in young fit patients with M-IGHV and no TP53 aberration, or help expand the group of patients who will achieve these protracted responses, is an important question that can be answered in longer follow up.
Extending these results into the R/R setting, the phase III HELIOS trial compared BR versus BR plus ibrutinib in previously treated CLL. 33 A significant PFS advantage was observed in the ibrutinib arm. An updated analysis after nearly 3 years follow up demonstrated a significant PFS and OS advantage with the addition of ibrutinib (PFS at 3 years 68%) over BR. 72 The addition of ibrutinib to BR was also associated with higher rates of uMRD (26% versus 6%, respectively). Yet, the PFS rate observed with the combination of ibrutinib-BR is similar to that seen with single agent ibrutinib, 11 thus there is no compelling evidence at this time to pursue a combination of ibrutinib with BR in regular clinical practice.
Something borrowed: does anti-CD20 monoclonal antibody help?
The anti-CD20 monoclonal antibody rituximab has improved outcomes when added to standard of care chemotherapy regimens in nearly all B-cell lymphoma subtypes, including CLL, since it was first introduced over two decades ago.3,73 Based on this experience, the MD Anderson Cancer Center conducted a comparison study of ibrutinib versus ibrutinib plus rituximab. 17 Patients enrolled had either R/R disease or TP53 aberration. As a result, PFS was nearly identical in the two arms, 86% with ibrutinib versus 86.9% with ibrutinib-rituximab at 3 years of follow up. Patients treated with ibrutinib-rituximab had higher rates of uMRD, but this did not translate into superior survival. The results were similar in the Alliance trial, which also demonstrated no difference in outcomes between ibrutinib and ibrutinib-rituximab arms. 22 Other anti-CD20 antibodies have also been investigated in this space. Ofatumumab was combined with ibrutinib after two cycles of bendamustine debulking in a phase II trial. 74 The combination produced a response rate of 92% with 14% of patients achieving uMRD. The phase III iLLUMINATE trial compared ibrutinib with obinutuzumab to chlorambucil and obinutuzumab as frontline therapy for either elderly patients or younger patients with comorbidities. 34 Ibrutinib + obinutuzumab demonstrated superior PFS but no direct comparison with single agent ibrutinib was made and ibrutinib therapy was continued indefinitely leaving these key questions unanswered. An interesting phase II study attempted to use time-limited therapy with frontline ibrutinib-obinutuzumab with the goal of stopping therapy with uMRD and thus avoiding CIT. 75 Patients received 6 months of the regimen followed by 3 months of ibrutinib alone at which time those in a CR with uMRD went on to receive an additional 6 months of ibrutinib while all others received four cycles of iFCG. Unfortunately, only 12% of patients in this study were able to avoid CIT. Thus, the data available so far suggest no benefit of adding an anti-CD20 antibody to ibrutinib in unselected patients with CLL.
Acalabrutinib combinations with obinutuzumab have also been investigated. A phase Ib/II trial of continuous acalabrutinib with six cycles of obinutuzumab produced a PFS rate at 3 years of 94% as frontline therapy and 88% in R/R CLL. 48 The phase III ELEVATE TN trial compared acalabrutinib versus acalabrutinib with obinutuzumab versus chlorambucil with obinutuzumab in elderly or comorbid patients with previously untreated CLL. 19 Both acalabrutinib arms (where the drug was administered continuously) produced superior PFS compared with chlorambucil. The superiority of acalabrutinib-obinutuzumab combination was seen in patients with both mutated and unmutated IGHV. Of note, the estimated PFS at 2 years was 87% with acalabrutinib versus 93% with acalabrutinib-obinutuzumab and while the study was not powered to evaluate the effect of obinutuzumab added to a BTK inhibitor, this difference in PFS is provocative. A phase I trial assessed another anti-CD20 antibody, ublituximab in combination with umbralisib and ibrutinib. 76 A total of 22 patients with CLL were included, all of whom had an objective response.
Novel agents and novel-novel combinations, is three really a charm? A crowd?
In addition to the BTK inhibitors described in this review, several other classes of novel agents have demonstrated activity in CLL. The PI3K inhibitor idelalisib was approved by the FDA in combination with rituximab for R/R CLL. 77
Venetoclax is a BCL-2 inhibitor that was approved by the FDA in 2019 for the treatment of CLL based on the CLL14 trial results. In this phase III trial, fixed duration (12 months) venetoclax + obinutuzumab was compared with chlorambucil + obinutuzumab as frontline therapy in patients with comorbidities. A significant PFS benefit was observed with venetoclax including patients with high-risk disease features. 36 In the phase III MURANO study, patients with R/R CLL received venetoclax combined with rituximab for up to 2 years. This regimen demonstrated a PFS advantage compared with BR in all patients and in those with TP53 aberrations. 35 These pivotal trials demonstrated the potential for time-limited, chemo-free regimens with venetoclax in combination with the anti-CD20 antibody and high efficacy in patients with high-risk disease. The combination of time-limited regimens that are active in difficult-to-treat patients with CLL make venetoclax plus anti-CD20 antibody combinations particularly compelling.
Yet, given the lack of head-to-head trial data comparing novel agents, the choice of frontline therapy is still largely empiric. A retrospective analysis unsurprisingly demonstrated superiority of ibrutinib over idelalisib in de novo CLL; meanwhile, venetoclax and kinase inhibitors were both superior to CIT in relapse. 78 A phase II trial of venetoclax in R/R CLL demonstrated an ORR of 65% and a median PFS of 28 months in patients who had previously progressed on ibrutinib. 79 Similarly, in an analysis of patients from the MURANO study who progressed after venetoclax + rituxumab, ibrutinib demonstrated good clinical activity with an ORR of 87%. 80 Thus, either ibrutinib or venetoclax are reasonable options for frontline therapy, especially in patients with high-risk disease and responses may be expected at the time of progression. Longer follow-up data from well-designed sequencing trials will be required to fully elucidate the best frontline approach.
An as yet unanswered question is whether combining novel agents with distinct mechanisms of action will produce a synergistic effect, especially in regards to OS, in patients with CLL.37,38 BTK inhibition leads to mobilization of CLL cells out of protective niches within the lymph node and into peripheral blood where venetoclax is especially active, highlighting the biological rationale for this combination. 81 Multiple trials that assess this approach are ongoing (Table 2). A phase II trial of ibrutinib-venetoclax as frontline therapy for a fixed 2-year treatment duration reported a CR rate of 88% with 61% of patients achieving uMRD in the bone marrow after 12 months. 37 The phase II CLARITY trial tested this same combination in the R/R setting, but used an MRD-guided strategy to determine treatment duration. After 1 year of treatment, 53% of patients had uMRD in peripheral blood and 36% in the bone marrow. 38 The phase II CAPTIVATE trial assessed frontline ibrutinib-venetoclax with a second randomization at 1 year based on MRD status. 20 After 1 year of combination therapy, patients with uMRD are randomized to ibrutinib or placebo while those who were MRD positive are randomized to ibrutinib or ibrutinib-venetoclax. Full results of the study are currently not available. Another phase II trial investigated the triplet of ibrutinib, venetoclax, and obinutuzumab in both frontline and R/R settings. Early results from this trial indicate a manageable safety profile with uMRD after 1 year of therapy in blood and bone marrow of 67% in previously untreated and 50% in R/R CLL. 39
Key ongoing and future trials of BTK inhibitors in combination with venetoclax and anti-CD20 antibodies.

BR, bendamustine and rituximab; BTK, Bruton’s tyrosine kinase; FCR, frontlineudarabine, cyclophosphamide and rituximab; G, obinutuzumab; I, ibrutinib; R, rituximab; R/R, relapsed or refractory; uMRD, undetectable minimal residual disease; V, venetoclax.
Results from several large ongoing trials should further clarify the use of these agents in combination. The phase III GAIA/CLL13 trial is currently underway and is designed to compare fixed duration venetoclax containing regimens with high-intensity CIT as frontline therapy for fit patients. The four arms in the trial are FCR or BR in patients >65 years, venetoclax-rituximab, venetoclax-obinutuzumab, and venetoclax-ibrutinib-obinutuzumab (Table 2). All venetoclax regimens will be administered for 1-year. 18 An ongoing phase III trial (ECOG EA9161) will compare ibrutinib-obinutuzumab versus ibrutinib-obinutuzumab-venetoclax (for 19 cycles) in younger treatment-naïve patients [ClinicalTrials.gov identifier: NCT03701282]. The phase III Alliance A041702 trial will similarly assess ibrutinib-obinutuzumab versus ibrutinib-obinutuzumab-venetoclax in previously untreated older patients with CLL. After 18 months of therapy, patients in the triplet arm will stop therapy if they achieve uMRD, and will continue ibrutinib indefinitely if they do not. Patients in the doublet arm will continue ibrutinib indefinitely. 21 Unfortunately, neither of the above trials will directly evaluate the need for anti-CD20 directed therapy. Finally, the German CLL study group phase III CLL17 trial will assess ibrutinib versus venetoclax-obinutuzumab versus ibrutinib-venetoclax.
Multiple trials are also underway evaluating acalabrutinib combinations. A phase II trial is assessing acalabrutinib-venetoclax-obinutuzumab in the frontline setting with therapy stopped at 15 or 24 months depending on the achievement of uMRD. Preliminary results, after 8-cycles, demonstrated a manageable safety profile, with uMRD achieved in 65% of patients in peripheral blood and 50% in bone marrow. 82 An ongoing phase III trial will compare acalabrutinib-venetoclax with or without obinutuzumab with investigator’s choice of CIT in previously untreated CLL without TP53 aberrancy. 16 Another phase II trial, in patients with high-risk or R/R CLL, will assess the efficacy of acalabrutinib-venetoclax with obinutuzumab added if patients are not in an early CR with uMRD [ClinicalTrials.gov identifier: NCT04168737].
The above trials will answer some of the most pressing questions regarding optimal frontline therapy using drug combinations and may provide initial insights into sequencing of agents when patients are being followed for OS. Still, CLL remains an incurable disease and, despite the rapid development of new agents in CLL, the majority of patients will still progress and require additional treatments. Time-limited regimens, particularly those directed by MRD status may provide the most judicious use of currently available therapies. Whether ibrutinib or other BTK inhibitors can be safely stopped after the achievement of uMRD is a question that we are beginning to address in the above-mentioned trials (GAIA/CLL13, ECOG EA9161, CAPTIVATE and Alliance A041702) Detailed opinions and discussions on the role of MRD in CLL may be found in recently published reviews.83,84 Increased frequency of toxicities, including infections, and high costs, are additional potential obstacles to triplet therapy.
Cellular therapies still have a role in the treatment of some patients with CLL. Chimeric antigen receptor T-cells (CAR-T) have demonstrated promising activity after CLL becomes resistant to BTK inhibition and other targeted agents.85–87 Interestingly, preliminary studies have suggested that ibrutinib may improve efficacy of CAR-T therapy and reduce toxicity.88,89 CAR-T has also been administered to deepen response in patients not achieving a CR with ibrutinib monotherapy, 87 and ibrutinib has been administered concurrently with CAR-T. In a pilot study combining ibrutinib with CAR-T in patients with high-risk, R/R CLL (median five prior lines of therapy), the response rate was 83%, with 61% achieving uMRD. Cytokine release syndrome was common in this study but of low severity. 87 Thus, the currently available data suggests that CAR-T may be the most promising third line of therapy after BTK inhibitor and venetoclax failures. Furthermore, these data suggest that both prior and concurrent BTK inhibition may facilitate CAR-T cell therapy.
Summary
Targeted therapies have improved survival in patients with CLL. This change has been spearheaded by the development of ibrutinib, which is now considered a standard frontline therapy for patients with CLL, and whose use in this setting has increase dramatically over the past few years. Venetoclax has also demonstrated significant activity, as both a single agent and in partnership with anti-CD20 antibodies, and is a worthy competitor when evaluating frontline therapy options in CLL.
Development of second generation BTK inhibitors (i.e., acalabrutinib, zanubrutinib and others), which so far have demonstrated comparable efficacy, but are expected to have fewer adverse events and better tolerability than ibrutinib, might yet again change the treatment paradigm in CLL. In our practice, use of second-generation BTK inhibitors in place of ibrutinib is expected to increase as they receive their regulatory approvals.
Despite this progress, CLL remains an incurable disease. Current clinical trials are focused on identifying the best combination(s) incorporating BTK inhibitors with the goal of identifying effective time-limited, chemotherapy-free regimens. These trials will shed light onto an important question of whether a shift in the traditional mindset from disease control to a cure in CLL is imminent, and what contribution BTK inhibitors will have in this new era.
Footnotes
Acknowledgements
AVD is a Leukemia and Lymphoma Society Scholar in Clinical Research (#2319-19).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
AVD reports consultancy from Abbvie, Beigene, Celgene, Curis, Janssen, Karyopharm, Nurix, Seattle Genetics, Teva Oncology, and TG Therapeutics; research funding from Aptose Biosciences, Bristol-Myers Squibb, Gilead Sciences, and Takeda Oncology; and consultancy and research funding from AstraZeneca, Bayer Oncology, Genentech, and Verastem Oncology.
