Abstract
The treatment landscape of chronic lymphocytic leukemia (CLL) has significantly changed in the past decade. This paradigm shift is due to the introduction of novel agents to the field. The two major classes of drugs that have contributed to this dramatic evolution include the Bruton tyrosine kinase (BTK) inhibitors and BCL2 inhibitors. Ibrutinib was the first-in-class drug which was initially approved by the US Food and Drug Administration (FDA) for the treatment of patients with relapsed/refractory and later for patients with treatment-naïve CLL. Despite encouraging efficacy outcomes, its use has been associated with cardiovascular and gastrointestinal toxicities likely due to off-target inhibition of ITK, TEC and EGFR family kinases. The next generation of BTK inhibitors was developed to be more selective with less off-target inhibition with the prospect to improve tolerability without compromising efficacy. Acalabrutinib, a selective covalent BTK inhibitor, is a second generation BTK inhibitor. The focus of this review is on two major phase III trials that resulted in the FDA approval of acalabrutinib in 2019. The ELEVATE TN trial investigated acalabrutinib with or without obintuzumab
Keywords
Introduction
The treatment landscape of chronic lymphocytic leukemia (CLL) has drastically changed in the past 5 years. Inhibition of the B-cell receptor pathway, particularly of Bruton tyrosine kinase (BTK), is a major therapeutic target in B-cell malignancies, including CLL. The introduction of BTK inhibitors has revolutionized the field of CLL. The first-in-class BTK inhibitor, ibrutinib, was approved for the treatment of patients with relapsed/refractory CLL by the US Food and Drug Administration (FDA) in February 2014. The initial approval was based on the phase III RESONATE trial, which compared single agent ibrutinib to ofatumumab in high-risk patients with relapsed/refractory CLL.
1
Long-term follow-up of the RESONATE trial showed that at a median follow-up of 65.3 months, the median progression-free survival (PFS) of patients in the ibrutinib arm was 44.1 months
Given the promising long-term outcomes of both RESONATE and RESONATE-2 trials, the next rational step was to compare the ibrutinib-based treatment combinations to traditionally used chemoimmunotherapy regimens in the frontline setting. As such, the Alliance group designed a phase III multicenter study randomly assigning 547 patients older than 65 years of age with treatment-naïve CLL to chemoimmunotherapy with bendamustine–rituximab
Despite promising results associated with ibrutinib, its use has been limited by its toxicity profile. In the largest real-world analysis, with a median follow-up of 17 months, an estimated 41% of patients discontinued ibrutinib mainly due to unfavorable side effects. The most common toxicities include cardiovascular (atrial fibrillation, hypertension), diarrhea, skin rash and arthritis. 10 The availability of new generations of the same class, with more selective target inhibition, can improve on the side effect profile of ibrutinib. In this review, we elaborate on the landmark phase III trials that established a place for acalabrutinib in the treatment of patients with CLL and focus on unmet needs and future directions.
Acalabrutinib mechanism of action and pharmacodynamics
BTK is a proximal component of the B-cell receptor signaling pathway and a therapeutic target for a wide range of B-cell malignancies including CLL. Acalabrutinib forms a covalent bond with Cys481 in the ATP-binding pocket which results in irreversible BTK inhibition. Unlike ibrutinib, acalabrutinib is more selective and does not inhibit other kinases, including EGFR, TEC, or ITK.11,12 Inhibition of both on-target and off-target kinases is known to contribute to ibrutinib-related adverse events such as an increased risk of atrial fibrillation, bleeding (through TEK, ITK) and diarrhea (through EGFR).13–15 Toxicity is the most common reason for discontinuation of ibrutinib. 10 For patients who discontinue ibrutinib due to intolerance, treatment with acalabrutinib is commonly well tolerated. 16
In terms of dosing schedule, the phase I study of acalabrutinib in B-cell malignancies demonstrated that all tested doses (100–400 mg daily and 100 mg twice daily) achieved superb BTK inhibition 4 h after dosing, but the 100 mg twice daily dosing schedule showed optimal inhibition 24 h after dosing.
11
However, BTK occupancy measured in the phase I studies of ibrutinib and acalabrutinib was performed in peripheral blood mononuclear cells rather than in tissue compartments.11,17 Given the fact that most B-cell receptor activation and ensuing cell proliferation occur within lymph nodes, it is imperative to study the relationship between different dosing schedules and maximal BTK occupancy in lymph nodes. To pursue this goal, Sun
Acalabrutinib in treatment of patients with treatment-naïve CLL
ELEVATE TN is a phase III, multicenter, international study in patients with treatment-naïve CLL comparing the efficacy of acalabrutinib with or without obinutuzumab against chlorambucil with obinutuzumab. The trial was designed to include older and frail patient populations. The eligibility criteria included patients with untreated CLL aged 65 years or older, or younger than 65 years with creatinine clearance of 30–69 mL/min or a cumulative illness rating scale for geriatrics score greater than 6. Additional criteria included an Eastern Cooperative Oncology Group (ECOG) performance status score of 0–2 and adequate liver, kidney and hematological function. Patients with significant cardiovascular disease were excluded. Of 535 patients, 179 patients were randomly assigned to receive acalabrutinib plus obinutuzumab, 179 were assigned to single agent acalabrutinib, and 177 patients were assigned to obinutuzumab and oral chlorambucil in 28-day cycles. To reduce infusion-related reactions, acalabrutinib was administered for one cycle before obinutuzumab administration. Oral acalabrutinib was administered at 100 mg twice a day until disease progression or unacceptable toxicity. Oral chlorambucil was given (0.5 mg/kg) on days 1 and 15 of each cycle for a total of six cycles. The primary endpoint was PFS. Crossover to acalabrutinib was allowed in patients who progressed on obinutuzumab–chlorambucil. 19
At a median follow-up of 28.3 months, median PFS was longer in acalabrutinib-containing arms compared with obinutuzumab–chlorambucil. Estimated 2-year PFS was 93% with acalabrutinib–obinutuzumab (95% CI 87–96%), 87% with acalabrutinib monotherapy (95% CI 81–92%), and 47% with obinutuzumab–chlorambucil (95% CI 39–55%). The most common grade 3 or higher adverse event was neutropenia among all three arms. Grade 3 or higher infections occurred in 21%, 14% and 8% of patients in acalabrutinib–obinutuzumab, acalabrutinib monotherapy, and obinutuzumab–chlorambucil cohorts, respectively. Importantly, grade 3 or higher atrial fibrillation and hypertension were 0.6% and 2.8% in the acalabrutinib plus obinutuzumab arm, 0 and 2.2% in the acalabrutinib monotherapy arm, and 0 and 3% in the obinutuzumab–chlorambucil arm. In terms of difference between the PFS in acalabrutinib-containing arms, the study was not powered to draw statistically significant conclusions between those two arms.19,20 Based on this study, the FDA granted approval to acalabrutinib – either as a single agent or in combination with obinutuzumab – for patients with untreated CLL.
Acalabrutinib in treatment of patients with relapsed/refractory CLL
Ascend is a phase III, multicenter, international study investigating acalabrutinib in patients with relapsed/refractory CLL. Eligible patients older than 18 years were randomly assigned and stratified by del(17p) status, ECOG performance status score, and number of prior lines of therapy. Patients received acalabrutinib monotherapy or investigator’s choice (idelalisib plus rituximab or bendamustine plus rituximab). The primary endpoint was PFS. Key secondary endpoints included ORR, OS, and safety. Of 310 patients, 155 were randomly assigned to acalabrutinib monotherapy and 155 patients were randomly assigned to investigator’s choice (idelalisib–rituximab,
In addition to the ASCEND trial, acalabrutinib is currently being compared in a head-to-head fashion to ibrutinib in patients with relapsed/refractory CLL (NCT02477696). The accrual target is 533 participants, which started in October 2015 and is anticipated to complete in March 2021. The primary endpoint of this study is PFS, and key secondary endpoints include the incidence of grade 3 and higher treatment-emergent infections, Richter’s syndrome, atrial fibrillation and overall survival between the two arms. A summary of the landmark phase III trials of BTK inhibitors for patients with treatment-naïve and relapsed/refractory CLL is provided in Table 1.
Summary of phase III trials of BTK inhibitors for patients with treatment-naïve and relapsed/refractory CLL.
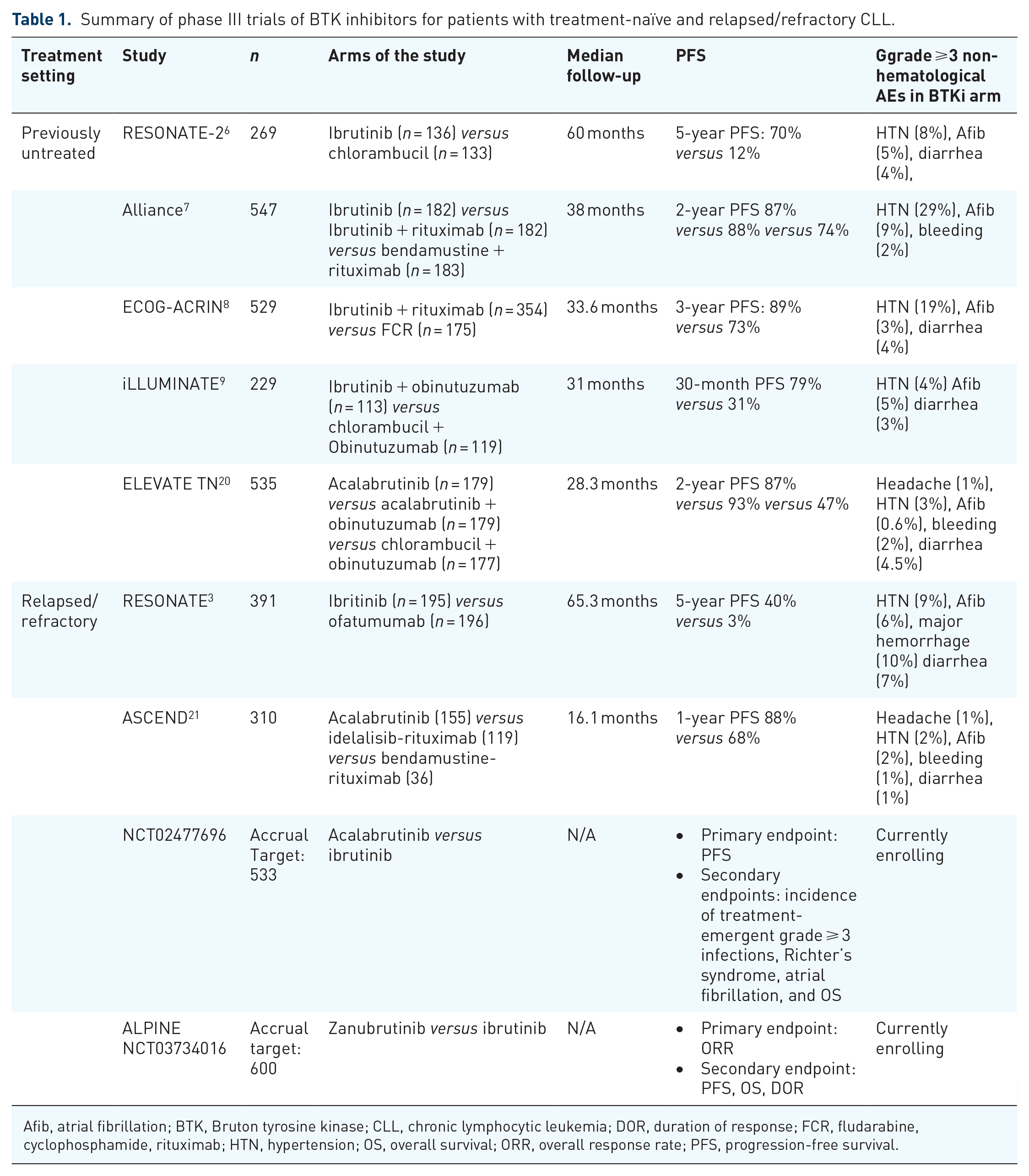
Afib, atrial fibrillation; BTK, Bruton tyrosine kinase; CLL, chronic lymphocytic leukemia; DOR, duration of response; FCR, fludarabine, cyclophosphamide, rituximab; HTN, hypertension; OS, overall survival; ORR, overall response rate; PFS, progression-free survival.
Discussion and future directions
The ELEVATE TN trial in untreated patients with CLL and the ASCEND trial in patients with relapsed/refractory CLL indicate that acalabrutunib is an effective treatment option with an acceptable side effect profile. In the ELEVATE TN trial,
20
after a median follow-up of 28.3 months, the 2-year PFS was 90%
In addition to CLL, both in upfront and relapsed/refractory settings, acalabrutinib has been approved in patients with relapsed/refractory mantle cell lymphoma based on a phase II trial. 24 In this trial, acalabrutinib at the recommended dose of 100 mg twice daily was proved to be well tolerated as evidenced by only 1.6% of patients requiring dose reductions and only 6.5% of patients discontinuing acalabrutinib due to adverse events. Of note, atrial fibrillation was not observed in any patient. The most common side effects were headaches (36%) and diarrhea (38%), both of which were typically grades 1–2 and self-limited.24,25
It is critical to note that despite the FDA approval of acalabrutinib for both upfront and relapsed/refractory settings in patients with CLL, there are currently no data on acalabrutinib in young and fit patients. As well as the currently FDA approved ibrutinib and acalabrutinib in the treatment of patients with CLL, other BTK inhibitors are under investigation in multiple national and international trials. To name a few, zanubrutinib – another covalent BTK inhibitor, has shown high overall response rates in patients with relapsed/refractory CLL. 26 Moreover, LOXO-305 is a non-covalent BTK inhibitor with activity against the most common acquired BTK inhibitor resistance mutations in CLL (Cys485 and phospholipase C gamma-2). 27 The results of phase I of LOXO-305 in patients with relapsed/refractory CLL was presented at the 2020 Annual ASH Meeting, with outstanding response rates in patients with heavily pre-treated CLL. 28
Before addressing the future directions, it is necessary to talk about the German CLL14 trial. In this trial, 432 patients with untreated CLL were randomly assigned to BCL2 inhibitor venetoclax plus obinutuzumab for one year
Considering the growing armamentarium of treatment options available for patients with treatment-naïve CLL, the next reasonable step is to study the combination of BTK inhibitors and BCL2 inhibitors. In doing so, the design of the trials should focus on time-limited approaches with the potential use of minimal residual disease based on next generation sequencing to guide the treatment duration in select populations of patients. The phase II CAPTIVATE trial combined ibrutinib and venetoclax in previously untreated high-risk and older patients with CLL. Patients received ibrutinib monotherapy for three cycles, followed by the addition of venetoclax for a total of 24 cycles. Overall, 92% of the patients had negative prognostic features – unmutated
In summary, the future outlook for patients with CLL has never been brighter. With the development of new targeted agents, the field has entered a new era. Physicians and patients are not choosing between the lesser of two evils anymore. Oncologists have the luxury of tailoring treatment strategies to individual patients’ needs and circumstances. As we celebrate the recent advances, it is critical to bear in mind that opportunity translates into responsibility. Unfortunately, illness is universal but access to care is not. The countries with access to targeted agents have a moral and ethical responsibility to facilitate access to these agents to underserved communities through a variety of national and global health initiatives until every patient with CLL can enjoy the benefits of these new treatments.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics and consent
The authors confirm that ethical committee approval was sought where necessary and is acknowledged within the text of the submitted manuscript.
As the manuscript is a review of literature and no patient information has been included in the text or analysis, the guidelines on patient consent do not apply to this manuscript.
