Abstract
Primary systemic immunoglobulin light chain (AL) amyloidosis is caused by a plasma cell clone of, usually low, malignant potential that expresses CD38 molecules on their surface. Treatment of AL amyloidosis is based on the elimination of the plasma cell clone. The combination of cyclophosphamide–bortezomib–dexamethasone (CyBorD) is the most widely used and is considered a standard of care; however, complete hematologic response rates and organ response rates remain unsatisfactory. Daratumumab, an anti-CD38 monoclonal antibody, has demonstrated encouraging results, with rapid and deep responses, in patients with relapsed or refractory AL amyloidosis as monotherapy with a favorable toxicity profile. The large phase-III, randomized, ANDROMEDA study evaluated the addition of daratumumab to CyBorD in previously untreated patients with AL amyloidosis and demonstrated that addition of daratumumab can substantially improve hematologic complete response rates, survival free from major organ deterioration or hematologic progression, and organ responses. In this review, we discuss the role of daratumumab in the treatment of AL amyloidosis, its mechanism of action, and the results of ANDROMEDA study that led to the first approved therapy for AL amyloidosis.
Introduction
Primary systemic light chain (AL) amyloidosis is a plasma cell dyscrasias characterized by extracellular tissue deposition of insoluble amyloid fibrils, which are generated by misfolded free light chains (FLCs). A small, usually low-grade, lymphoproliferative clone in the bone marrow produces the toxic monoclonal immunoglobulins that can affect almost every organ system, but most frequently the heart, kidneys, liver, nerves, and soft tissues. The incidence of AL amyloidosis ranges approximately from 10 to 15 cases per million persons per year, and the prevalence of the disease has been increased over the last decade from 15 to 40 cases per million people, with an annual rate of 12%, 1 probably due to improved recognition and increased suspicion of the disease. The overall survival (OS) has improved over the past two decades, after the introduction of novel immunotherapeutic anti-plasma cell therapies, with 2-year OS increasing from 42% to 60% and 4-year OS from 31% to 54%. 2 However, the disease is still incurable and the prognosis is highly dependent on the cardiac involvement and the time of diagnosis. 3 Given also that the survival and organ improvement are intrinsically related to hematologic response, 4 the introduction of more efficacious combinations is highly relevant. Improved outcomes are closely related to deep and rapid hematologic responses. Until recently, there was no approved therapies for AL amyloidosis by either US Food and Drug Administration (FDA) or European Medicines Agency (EMA). Based on the results of the ANDROMEDA study, on 15 January 2021, FDA approved the use of subcutaneous (SC) daratumumab in combination with cyclophosphamide–bortezomib–dexamethasone (CyBorD) for the treatment of newly diagnosed AL amyloidosis patients with Mayo stages I to IIIA (https://www.fda.gov/drugs/drug-approvals-and-databases/fda-grants-accelerated-approval-darzalex-faspro-newly-diagnosed-light-chain-amyloidosis). Later, in June 2021, EMA also approved the combination for this indication.
The current review will provide data on the combination of daratumumab with CyBorD as the new standard of care in patients with AL amyloidosis.
Current treatment of AL amyloidosis
The target of therapy in AL amyloidosis is to eradicate, as fast as possible, the lymphoproliferative clone that produces the precursor protein of amyloid fibrils. The achievement of at least very good partial response (VGPR) has been associated with improved OS and improved organ function.5,6 The mainstay of therapy is regimens that have been used in multiple myeloma (MM), adjusted for schedule and dose. The use of alkylating agents has held a central place in the therapy of AL amyloidosis for many decades. Melphalan in combination with prednisone or dexamethasone showed hematologic responses in up to 76% of patients non-eligible for autologous stem cell transplantation (ASCT), with minimal toxicity (mainly myelotoxicity) but with low complete response (CR) rate of 31%. 7 High-dose melphalan (HDM) followed by ASCT is an effective strategy with high rates of CRs, organ responses, and improved OS. 8 However, treatment-related mortality (TRM) is higher among patients with AL amyloidosis; thus, HDM/ASCT is used only in strictly selected subjects and only in experienced centers. 9 Exclusion criteria for ASCT include age, advanced cardiac involvement based on cardiac biomarkers (NT-proBNP and troponin), hypotension, very low serum albumin levels, and multi-organ involvement.
Following advances in MM, proteasome inhibitors (PIs) have been successfully incorporated in the management of AL amyloidosis. Plasma cell clones show intrinsic susceptibility to PIs, probably related to the extensive production of amyloidogenic FLCs, which is also associated with increased proteotoxic stress, altered organelle homeostasis, and reduced autophagy. 10 Bortezomib-based combinations are very effective and well tolerated. Today bortezomib is administered subcutaneously and in a weekly schedule with initial dose of 1.3 mg/m2 (if not contraindicated). Bortezomib with dexamethasone induce deep and rapid hematologic responses 11 and organ responses. 12 Bortezomib has been successfully combined with alkylating agents for the treatment of AL amyloidosis. In a randomized phase 3 trial, melphalan and dexamethasone (MDex) were combined with bortezomib resulting in 79% of patients to achieve hematological responses at 3 months, including 55% of at least VGPR. 13 Importantly, this combination resulted also in a significant improvement of the OS compared with MDex. The addition of cyclophosphamide to bortezomib and dexamethasone (CyBorD) has resulted in the most widely used combination for newly diagnosed AL amyloidosis patients. CyBorD has been evaluated in several small studies and large retrospective cohorts with overall hematological response rate reaching 60%, with 43% achieving at least a VGPR, 17% achieving cardiac response, and 25% renal responses. 14 Despite these promising findings, advanced cardiac disease (Mayo stage IIIB high-risk patients with NT-proBNP levels >8500 pg/ml) shows less favorable results with overall response rate (ORR) of 42% and at least VGPR of 23%. 14 In addition, the major adverse events of bortezomib are neurotoxicity and hypotension, and many AL patients have disease-related neuropathy or orthostatic hypotension. Thus, treatment should always be tailored to individual patients’ characteristics. Moreover, there are data supporting lower efficacy of bortezomib-based regimens for patients harboring translocation (11;14),15,16 which is found in up to 50% of patients, indicating that novel therapeutic combinations are needed.
The introduction of daratumumab
Daratumumab is a human IgGκ1 monoclonal antibody (MoAb) that binds with high affinity to CD38, a surface antigen of both normal and clonal plasma cells. According to studies using flow cytometry or next generation sequencing (NGS), CD38 is ubiquitously expressed on clonal plasma cells in both MM and AL amyloidosis,17,18 representing an attractive target for immunotherapy. CD38 is a 45-kD type II transmembrane molecule that functions as a transmembrane signaling receptor.
It has been shown that daratumumab induces tumor cell death
Daratumumab monotherapy had demonstrated efficacy in pretreated patients with AL amyloidosis in few prospective and several retrospective studies. Kauffman
Daratumumab in combination with CyBorD
The approval of SC daratumumab (1800 mg in 15 ml) with recombinant human hyaluronidase PH20, based on the results from COLUMBA trial in patients with MM, is of particular importance for AL amyloidosis. Especially for patients with cardiac involvement, the concern about volume overload is relevant. In addition, the SC daratumumab is associated with fewer administration-related reactions (ARRs). In the setting of newly diagnosed AL amyloidosis, ANDROMEDA study demonstrated for the first time that the addition of SC daratumumab in CyBorD can substantially improve hematologic and organ response rate. 34
The design of the ANDROMEDA study was straightforward. Briefly, eligible patients had to have confirmed systemic AL amyloidosis (involving at least one organ) and measurable hematologic disease, no prior therapy for plasma cell dyscrasia, no evidence of symptomatic MM, an estimated glomerular filtration rate of at least 20 ml/min/1.73 m2, and were excluded if they had stage IIIB disease per the 2004 Mayo staging system (i.e. if they had stage III disease and an NT-proBNP level >8500 ng/l). Eligible patients were randomized to receive either standard CyBorD for six cycles (weekly dosing, up to 24 doses) or standard CyBorD plus daratumumab for six cycles, and then maintenance therapy with monthly administration of daratumumab for up to 24 cycles for the daratumumab–CyBorD group only.34,35
There was a safety run-in part in which 28 patients were included. 34 The primary goal of this safety run-in was to identify any potential toxicity concerns. The most common treatment emergent adverse events were diarrhea (68%), fatigue (54%), and peripheral edema (50%), which were consistent with the previous experience with both SC daratumumab and CyBorD. Importantly, ARRs occurred in just one patient and was mild (grade 1). There were five deaths, three following autologous transplant and two due to amyloidosis organ progression; none was related to daratumumab–CyBorD therapy.
The ORR with daratumumab–CyBorD in this early small cohort was 96% with VGPR or better in 82% and CR in 36% (in 54% if modified criteria were used that did not require normalization of FLC ratio). The vast majority of responders achieved low difference between involved and uninvolved serum free light chain (dFLC) levels and involved free light chain (iFLC) levels (dFLC < 10 mg/l and iFLC < 20 mg/l), which indicates deep hematologic responses. Median time to first response (at least PR) was just 9 days, median time to VGPR was 19 days, and median time to best response (at least CR) was 85 days. However, daratumumab and other MoAbs are detected by serum immunofixation electrophoresis (IFE) rendering the establishment of CR (which requires negative immunofixation) challenging. A daratumumab-specific IFE reflex assay (DIRA) has been developed to distinguish the monoclonal band derived from therapeutic antibodies from the M-protein. 36 In addition, evaluation of minimal residual disease (MRD) using NGS or next generation flow (NGF) cytometry can help in the establishment of a very deep response (i.e. to the point of undetectable MRD). Detection of MRD is clinically relevant as patients with undetectable MRD have a very high probability of organ response and a very low probability of hematologic progression; 37 however, in ANDROMEDA, the data on MRD are not mature yet. In patients with cardiac involvement, administration of daratumumab is also feasible and well tolerated. This has been shown in patients with relapsed AL but was also seen in the safety run-in of ANDROMEDA. In the safety run-in section of the study, the overall organ response rate was 64% with 54% achieving cardiac response, 83% renal response, and 50% liver response.
The first interim analysis of the ANDROMEDA was presented in 2020 and results were published in 2021. 35 The primary end point of the study was overall hematologic CR rate, as assessed by an Independent Review Committee blinded to treatment. Hematologic CR was slightly different than the International Society of Amyloidosis (ISA) criteria, to account for abnormal FLC ratios due to very low uninvolved immunoglobulin levels. Thus, CR was defined as an iFLC level less than the upper limit of normal with negative serum and urine immunofixation without the requirement of normalized FLC ratio. Secondary end points of ANDROMEDA included major organ deterioration–progression-free survival (MOD-PFS, which was a new composite end point that included end-stage cardiac or renal failure, hematologic progression, or death), organ response rate, OS, 6-month hematologic CR rate, at least hematologic VGPR rate, time to and duration of hematologic CR, time to next treatment, and improvement in fatigue.
A total of 388 patients were enrolled (195 in daratumumab–CyBorD and 193 in the control arm). The characteristics of the patients were well balanced and fairly typical of the population of patients with AL amyloidosis. Although patients with stage IIIB were excluded, ~71% of patients had heart involvement, ~77% stage II or III disease, 36% stage IIIA cardiac disease, and 59% kidney involvement.
After a median follow-up of 11.4 months, hematologic CR rate (the primary end point) was 53.3% in the daratumumab group and 18.1%, corresponding to an odds ratio for CR of 5.1 [95% confidence interval (CI) = 3.2–8.2;
The hematologic CR rate was consistent in prespecified subgroups such as patients with more advanced cardiac disease, different age groups, different body weight, and also in those harboring, t(11;14).35,38 Almost 50% of patients with AL amyloidosis harbor translocation (11;14), and the presence of this abnormality has been associated with inferior response to bortezomib-based therapies (VGPR or better: 52%
Improvement of organ function, as reflected by organ response rates, is also a major goal of any treatment in AL amyloidosis. A strong consensus in AL amyloidosis is that the majority of patients obtaining a hematologic CR will also obtain a cardiac response and this can radically change patients’ prognosis and survival.
In ANDROMEDA study, among those evaluable for cardiac response, the 6-month cardiac response rate was 41.5% in the daratumumab group and 22.2% in the control group (nominal
The kidney is the second most frequently affected organ in AL amyloidosis, and proteinuria is the most relevant clinical biomarker for renal survival. MoAbs are safe for patients with nephrotic syndrome and impaired renal function,
40
while bortezomib in combination with cyclophosphamide and dexamethasone has a protective effect in advanced renal failure due to plasma cell dyscrasias.
41
However, in a series of 168 patients with advanced AL amyloidosis treated with daratumumab in combination with dexamethasone with or without bortezomib, heavy nephrotic-range albuminuria was a significantly adverse factor for hemEFS for both cohorts;
39
such patients often have stage III amyloidotic renal disease.
6
The impaired glomerular barrier in patients with severe non-selective proteinuria leads to excessive urinary losses of MoAbs
42
and might affect the plasma concentration of therapeutic regimen. There were only few patients with stage III renal disease, and in this subgroup the CR rates were lower but with a wide CI, so that no definite conclusion can be made. However, among those evaluable for renal response (daratumumab group,
One of the secondary end points in ANDROMEDA was MOD-PFS. This composite end point includes end-stage organ dysfunction (cardiac or renal failure), hematologic progression, or death, whichever occurs first. The MOD-PFS was significantly improved in the daratumumab–CyBorD arm compared with CyBorD alone (hazard ratio = 0.58, 95% CI = 0.36–0.93,
The median time to next treatment was not reached in the daratumumab group and was 10.4 months in the CyBorD group. In the initial analysis, 19 patients (9.8%) in the daratumumab–CyBorD and 79 patients (42.0%) in the CyBorD arm required and received subsequent therapy, but 48/79 (60.8%) patients in the CyBorD arm received daratumumab-based therapy. Regarding ASCT, 13 patients (6.7%) after daratumumab–CyBorD arm and 20 patients (10.6%) after CyBorD received HDM.
The OS data are not mature yet. In the initial analysis, there was no difference (56 deaths had occurred, 27 in the daratumumab arm and 29 in the control arm); 35 at the most recent update, 31 deaths have occurred in the daratumumab–CyBorD arm and 40 deaths in CyBorD arm. 38 However, significantly longer follow-up will be required to reveal differences in OS between the two arms.
Safety is a major concern for any regimen used for patients with AL amyloidosis, since most are quite frail with multi-organ dysfunction. In ANDROMEDA, the most common grade 3 or 4 adverse events were lymphopenia; pneumonia (7.8% and 4.3%; it was also the most common in both treatment groups), with grade 3 or 4 infections being 16.6%
Early deaths (i.e. <60 days from start of therapy) occurred in 13 patients in each group, and overall death was considered as related adverse events in 23 patients (11.9%) in the daratumumab–CyBorD and in 14 patients (7.4%) in CyBorD group; disease progression as a cause of death was reported in 1.0% with daratumumab–CyBorD
Patients in the daratumumab–CyBorD arm continued with daratumumab monotherapy after the initial six cycles, but the reported treatment-related adverse events which, during the initial six cycles, occurred in 38% were reduced to 21% after cycle 6, 38 while they were 36% in the six cycles of CyBorD alone.
Beyond small administration volume and short administration time, the use of SC daratumumab was also associated with very low rates of systemic ARRs, which occurred in only 14 (7.3%) patients, all of which were mild (grade 1 or 2) and of which 85.7% at the first administration. Nonetheless, all the patients received appropriate pre-medication at least during the initial administrations. No additional systemic ARRs beyond those reported in the primary analysis. Both daratumumab and bortezomib were given subcutaneously, and local injection-site reactions were not uncommon [in 54 patients (28.0%)
The shift of daratumumab in combination with CyBorD in the upfront setting of AL amyloidosis arises new challenges for the treatment of relapsed and/or refractory patients. Clinical trials with newer regimens and individualized options for previously treated AL amyloidosis are needed (Table 1). B-cell maturation antigen (BCMA) is generally overexpressed in the surface of clonal plasma cells, including amyloid clones. 43 In MM, antibodies targeting BCMA have shown efficacy in relapsed/refractory setting. Currently a phase 2 study with belantamab mafodotin, an antibody-drug conjugate, is ongoing in patients with relapsed/refractory AL amyloidosis (NCT04617925). Alkylating agents have been in the armamentarium of AL amyloidosis treatment for years. The introduction of melphalan flufenamide (melflufen), a novel peptide-drug conjugate that is metabolized to melphalan inside clonal plasma cells, is also evaluated in a phase 1/2 study (NCT04115956). The frequent presence of t(11;14) with overexpression of anti-apoptotic factor BCL-2 (B-cell lymphoma 2) in AL amyloidosis implicates susceptibility to the BCL-2 inhibitor, venetoclax.44,45 Pending appropriate prospective trials, this might be a promising option for carefully selected patients. Finally, therapies targeting amyloid deposits represent a novel approach to AL amyloidosis therapy. CAEL-101, a light chain amyloid fibril IgG1 MoAb, demonstrated minimal toxicity and significant organ responses in the analysis of phase 1 and phase 2 studies,46,47 and a randomized phase 3 trial in patients with advanced cardiac involvement is ongoing (NCT04512235 and NCT04504825).
Ongoing clinical trials with daratumumab and novel agents in patients with AL amyloidosis.
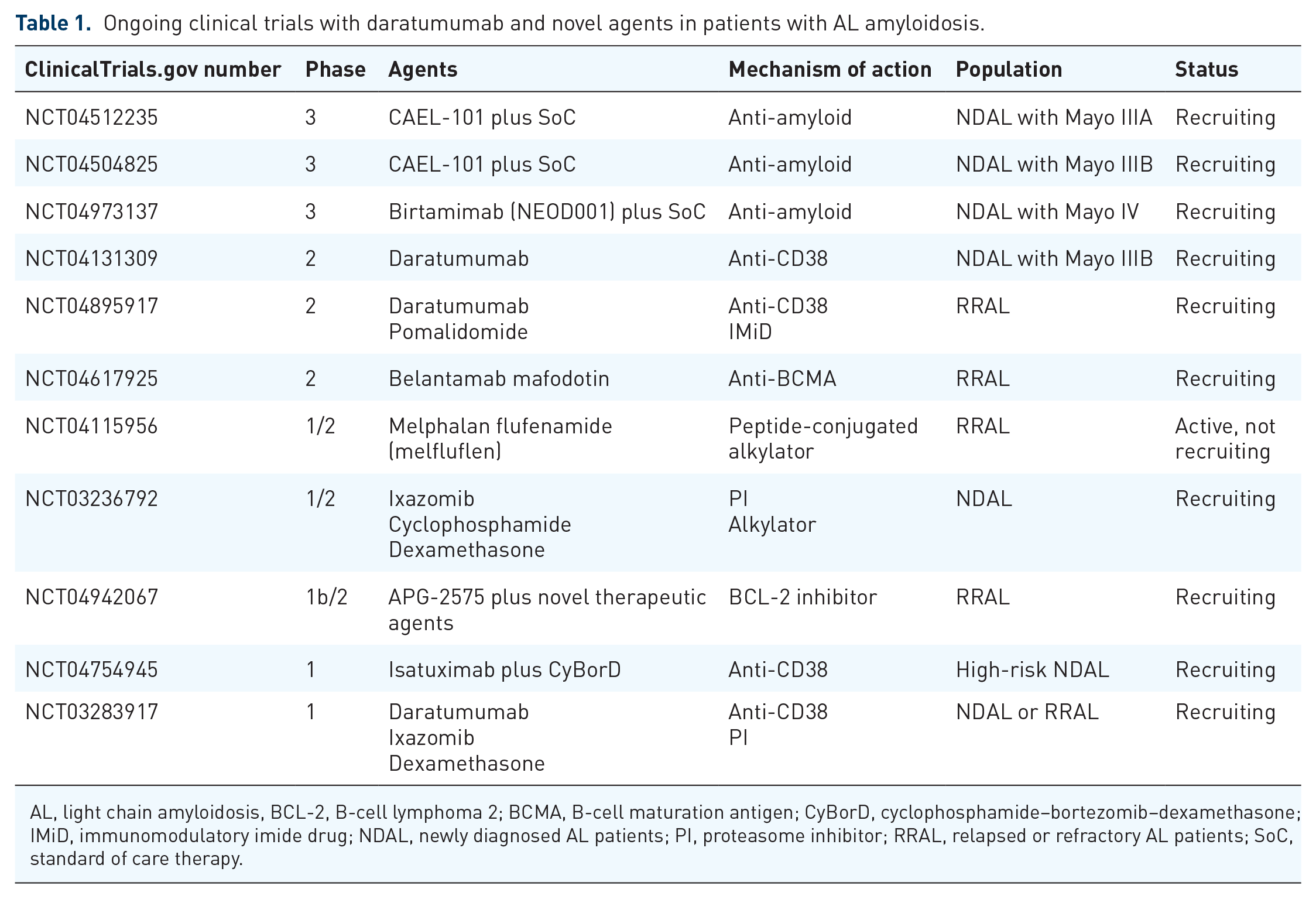
AL, light chain amyloidosis, BCL-2, B-cell lymphoma 2; BCMA, B-cell maturation antigen; CyBorD, cyclophosphamide–bortezomib–dexamethasone; IMiD, immunomodulatory imide drug; NDAL, newly diagnosed AL patients; PI, proteasome inhibitor; RRAL, relapsed or refractory AL patients; SoC, standard of care therapy.
Cardiac amyloidosis
Based on our previous experience and ANDROMEDA results, the administration of daratumumab–CyBorD is overall safe for cardiac amyloidosis patients. The rapid and deep responses observed even after the first administration can improve the outcome of patients with advanced disease. The use of pre-medication for daratumumab includes oral steroids, antihistamine, and montelukast for 2 days prior to the first administration, and dexamethasone, paracetamol, and antihistamine on the day of administration. The approval of SC daratumumab eliminates both volume overload adverse events and allergic reactions. However, the management of ultra-high-risk AL amyloidosis patients (Mayo IIIB, Mayo IV, or patients with severe symptomatic orthostatic hypotension) remains an unmet need.3,48 This population is often ill-tolerated of CyBorD due to deterioration of heart failure and hypotension; thus, doses should be risk-tailored. We recommend the administration of SC daratumumab 1800 mg with bortezomib at 0.7 mg/m2 plus low-dose dexamethasone (20 mg) weekly, and close monitoring for adverse events (AEs). Pre-hydration for patients with severe hypotension may be considered, but it could also exacerbate peripheral edema or dyspnea. Regarding the role of cyclophosphamide, it is questionable today and most probably will be abandoned in the future. The focus will be shifted toward chemotherapy-free combinations, especially for cardiac amyloidosis patients who are extremely frail and prone to chemotherapy toxicities. The combination of novel anti-clonal immunotherapies with anti-amyloid agents, that could efficiently reduce amyloid load from myocardium, holds promise for better survival.
Conclusion
The result of the first phase 3 study in AL amyloidosis has transformed the therapeutic landscape with the introduction of daratumumab in the frontline. The specific characteristics of the amyloid clone make daratumumab an appropriate and promising option in combination with the standard of care CyBorD. The improved tolerability of SC administration is also a novelty of ANDROMEDA trial and it is extremely meaningful for a subgroup of AL amyloidosis patients with concerns about volume overload. Although results from the study regarding daratumumab maintenance and MRD status are still anticipated, the very rapid and deep reduction in toxic FLCs that is observed with daratumumab will improve patients’ prognosis and organ survival.
Footnotes
Author contributions
F.T. and E.K. performed the literature research and drafted the manuscript. M.A.D. and E.K. offered expert opinion and performed revisions.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.K. has received honoraria/personal fees from Amgen, Genesis Pharma, Janssen, Takeda, and Prothena, and research grants from Amgen and Janssen. M.A.D. has received honoraria/personal fees from Amgen, BMS, Celgene, GSK, Janssen, and Takeda. FT declares no conclicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
