Abstract
Amyloid light chain (AL) amyloidosis is a protein conformational disease. AL amyloidosis results from aggregation of misfolded proteins that are deposited in tissues as amyloid fibrils. Diagnosis of AL amyloidosis can be challenging due to its low incidence and clinical complexity. Therapy requires a risk-adapted approach involving dose reductions and schedule modifications of chemotherapy regimens along with close monitoring of hematologic and organ responses. We herein describe a patient whose condition was diagnosed as systemic AL amyloidosis and presented with splenic rupture as the initial symptom. Congo red staining of the kidney biopsy was positive. The normal structure of the liver and spleen had been replaced by amyloid deposition. The chemotherapy strategy involved a combination of bortezomib, cyclophosphamide, thalidomide, and dexamethasone.
Keywords
Introduction
Amyloid light chain (AL) amyloidosis, namely primary systemic amyloidosis, is a protein conformational disease. AL amyloidosis is caused by unusual small plasma cell clones that produce misfolded light chains, which are then deposited in various tissues.1–3 AL amyloidosis involves multiple organs, including the heart (75%), kidneys (65%), liver (15%), soft tissues (15%), peripheral and/or autonomic nervous system (10%), and gastrointestinal tract (5%). 4 Presenting symptoms of systemic AL amyloidosis are broad and are often a consequence of advanced irreversible organ damage, mimicking other more common diseases of the older population. Thus, it is frequently misdiagnosed. 5 In approximately 40% of cases, AL amyloidosis is diagnosed more than 1 year after the onset of symptoms, and affected patients die within a few months after diagnosis despite modern treatments.6,7 Therefore, delayed diagnosis remains a major challenge in initiating effective therapy before organ function deteriorates over time, and recognition of the presenting manifestations is thus imperative for improving survival. Therapy requires a risk-adapted approach. Stem cell transplantation (SCT) is preferred, but only 20% of patients are eligible. Non-transplant candidates are offered chemotherapy. Appropriate treatments target the amyloid deposits and interfere with amyloidogenesis and organ damage. Emerging interventions, such as small RNA interference, are currently being developed.6,8–10 The purpose of the present case report is to provide a better understanding of the clinical features, pathogenesis, diagnosis, therapy, and prognosis of AL amyloidosis and thereby promote the management of amyloidosis.
Case report
A 45-year-old man was transferred to the emergency department because of sudden left upper quadrant pain. His condition was diagnosed as spontaneous splenic rupture and hemorrhagic shock as confirmed by computed tomography (CT). Emergency splenectomy was performed.
Frozen section during surgery and postoperative pathological examination were performed to identify any pathomorphological changes of the liver. Pathological examination revealed a nodular hepatic structure, arterial wall thickening in the portal area, extensive accumulation of a homogeneous substance (amyloid) among the hepatic cord cells, and difficult-to-recognize hepatic sinusoids (Figure 1). The ultrastructure of the spleen was impaired, the red pulp and white pulp were diffusely destroyed, and extensive accumulation of a homogeneous substance (amyloid) was present (Figure 2).

Hepatic hematoxylin and eosin staining showed a nodular hepatic structure, arterial wall thickening in the portal area, extensive accumulation of homogeneous substance (amyloid) among the hepatic cord cells, and difficult-to-recognize hepatic sinusoids.
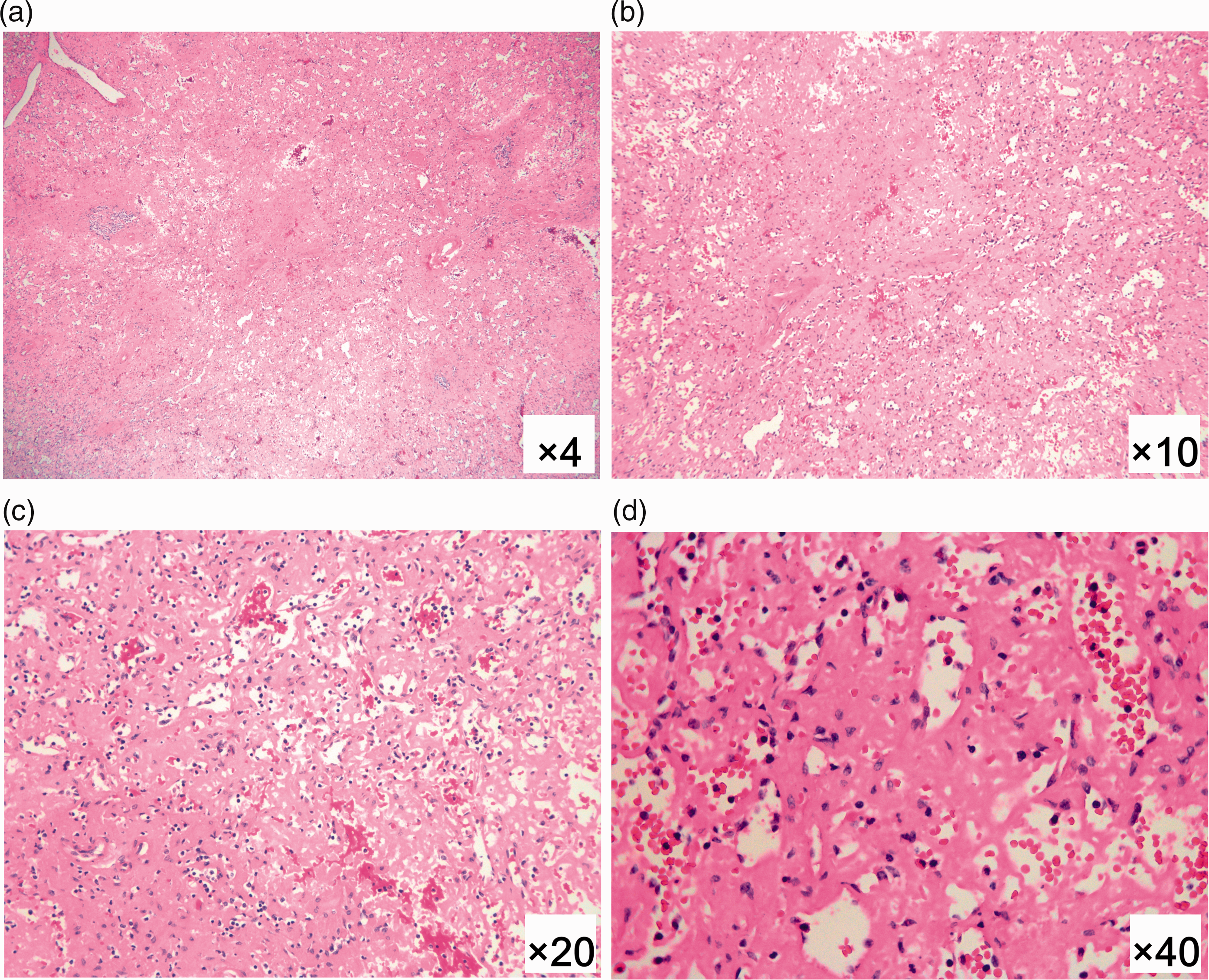
Splenic hematoxylin and eosin staining showed that the ultrastructure of the spleen was impaired and that the red pulp and white pulp were diffusely destroyed with extensive accumulation of homogeneous substance (amyloid).
A biopsy specimen from the kidney was stained with Congo red and periodic acid–Schiff; extensive accumulation of a homogeneous substance (amyloid) was present in the glomerulus, indicating renal amyloidosis (Figure 3(a) and (c)). The oxidized Congo red-stained renal tissue specimens (thickness of approximately 6 µm) were fixed in 8% formaldehyde and embedded in paraffin, dewaxed by dimethylbenzene, and hydrated by alcohol and distilled water. The sections were oxidized by KMnO4 solution (0.5% KMnO4 and 0.3% H2SO4 in a 1:1 ratio) for 5 min to induce degradation of the amyloid deposits according to Wright et al. 11 at 25°C; the sections were then bleached by 5% oxalic acid and distilled water two to three times. Finally, they were redyed by alkaline Congo red solution. Amyloid deposition was detected in the glomerulus, and AL amyloidosis was highly suspected (Figure 3(b)). Therefore, the spontaneous splenic rupture was concluded to be a result of the amyloidosis.

Kidney pathology. (a) Congo red stain showed characteristic amyloid deposition in the glomerulus. (b) Oxidized Congo red stain showed amyloid deposition in the glomerulus, indicating amyloid light chain amyloidosis. (c) Periodic acid–Schiff stain revealed amyloid deposition in the glomerulus.
CT imaging showed an enlarged liver mass in accordance with diffuse liver damage (Figure 4(a)). The patient and his family had no infectious or chronic diseases. Echocardiography showed a left ventricular ejection fraction of 55%, and the images suggested no obvious changes of either cardiac structure or function. Bone marrow biopsy showed no abnormalities (images not obtained). Repeat hepatic CT imaging still showed a diffusely enlarged liver, as before (Figure 4(b)). Laboratory tests (Table 1) results were normal. Immunophenotyping of the cells raised suspicion for plasma cells, accounting for 0.1% of all nucleated cells. A free light chain (FLC) assay revealed the following: serum κ Ig light chain, 462.5 mg/L; λ Ig light chain, 25.7 mg/L; κ/λ, 17.9961; and FLC-diff, 436.8 mg/L. Thus, the patient was classified as stage II according to the Mayo cardiac staging system. The combination of these data led to an ultimate diagnosis of AL amyloidosis; however, this took around 1 year.
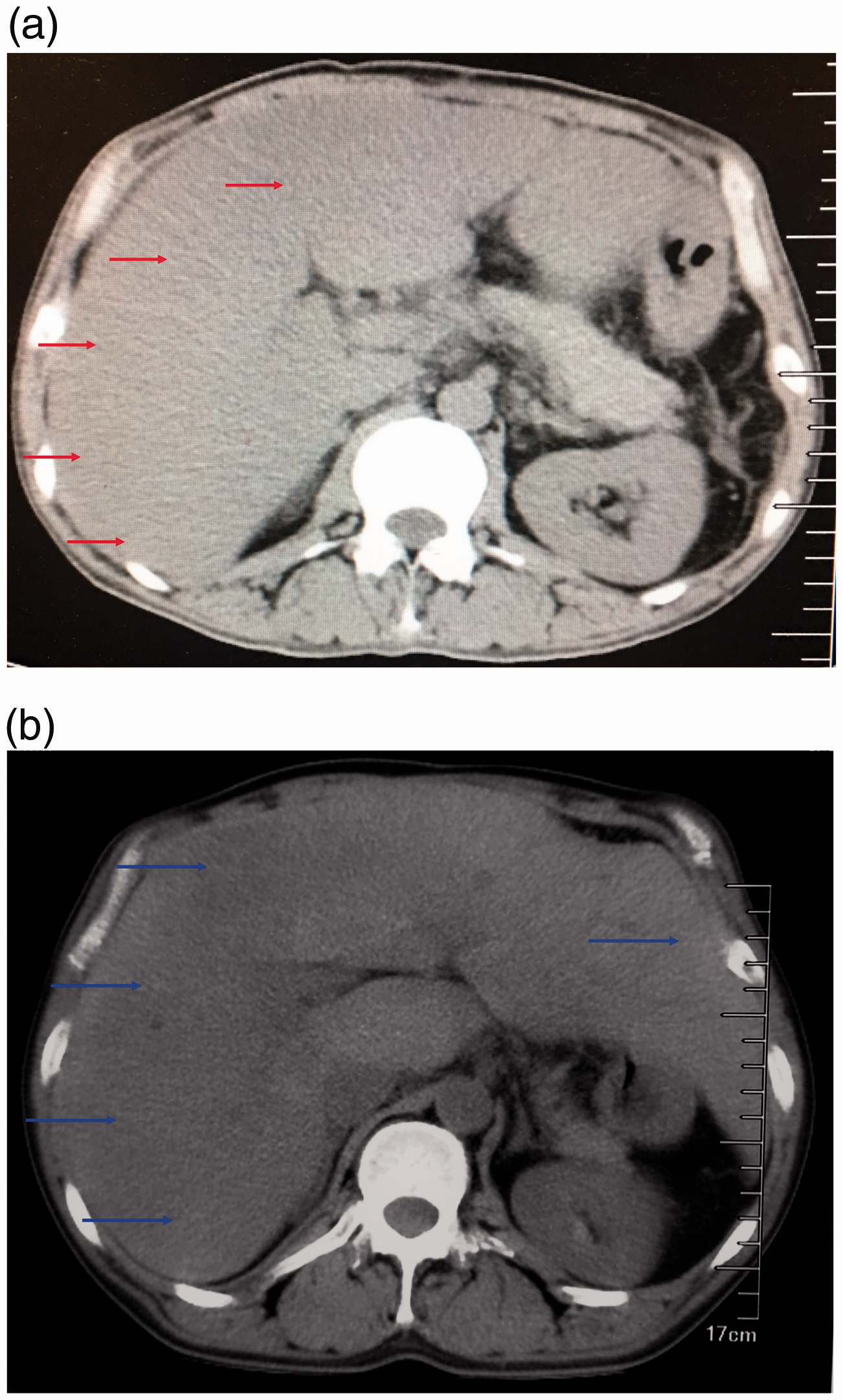
(a) Computed tomography showed an enlarged liver mass (red arrow) in accordance with diffuse liver damage. (b) The most recent follow-up computed tomography image showed a diffusely enlarged liver (blue arrow), as before.
Laboratory values over time.

Because of multiorgan involvement, the patient was treated with the RPCD regimen: bortezomib (1.5 mg/m2) 2.8 mg on days 1, 5, 9, and 12; prednisone 100 mg on days 1–4 and 7–10; cyclophosphamide 1.0 mg on days 1, 3, 5, and 7; and thalidomide 50 mg/d. No obvious adverse effects of chemotherapy were observed with the exception of anesthesia of the hands and feet. After four courses of treatment, the FLC assay showed that the serum κ Ig light chain level, λ Ig light chain, κ/λ, and FLC-diff decreased, suggesting a good partial hematologic response (Table 1). 12 Moreover, α-L-fucosidase, alkaline phosphatase, and γ-glutamyl transpeptidase decreased and prealbumin, total protein, and albumin increased (Table 1). Renal evaluation showed that the urine protein decreased and serum creatinine slightly increased. These data in Table 1 indicated an improvement in liver function and stability of renal function.
The latest follow-up was conducted 1 year after treatment initiation. Laboratory tests showed a stable disease state but gradual development of drug tolerance and an insufficient therapeutic response (Table 1).
This study was performed in compliance with the Declaration of Helsinki and was approved by the Ethics Committee of the First Hospital of Xi’an Jiaotong University. Written informed consent was obtained from the patient.
Discussion
The clinical manifestations, laboratory test results, and biopsy findings of this patient supported the diagnosis of AL amyloidosis (highly suspected Ig-κ type) with liver, kidney, and spleen involvement. The process from the beginning to diagnosis lasted around 12 months, further suggesting the challenge in detecting this disease. All ECG recordings during follow-up show no obvious abnormities, indicating little heart involvement. The patient was classified as stage II according to the Mayo cardiac staging system (troponin T of <0.003 ng/mL, NT-proBNP of 41.01 pg/mL, and FLC-diff of 436.8 mg/L), suggesting a statistical median survival of 40 months.9,13 Palladini et al. 14 proposed a staging system predicted by baseline proteinuria and the eGFR for renal involvement and criteria for early assessment of a renal response and progression in patients with AL amyloidosis. In their case, the patient’s renal function was stage I, indicating a low risk of dialysis at 2 years. The cardiac stage and depth of hematologic remission are major predictors of survival for patients with AL amyloidosis. Renal staging in patients with AL amyloidosis has been studied in the context of renal survival; however, hepatic or splenic involvement has not yet been studied. The lack of a functional spleen in our patient made it difficult to evaluate and compare the influence of splenic involvement in terms of the response to therapy and survival.
Relevant biomarkers are available to detect pre-symptomatic organ damage. NT-proBNP and troponin T have 100% diagnostic sensitivity in cardiac AL amyloidosis. Albuminuria for renal involvement and an abnormal FLC ratio prompt the performance of biomarker-based screening in patients with a high risk of developing AL amyloidosis, aiming to diagnose AL amyloidosis at its early stage and improving the survival rates of patients with AL amyloidosis.10,15–18 Identification of amyloid deposits requires adequate technology and expertise. Patients without definitive findings should be referred to specialized centers.
Some emerging testing approaches have been helpful in the diagnosis of AL amyloidosis. Immunoelectron microscopy can achieve 100% specificity. Diagnostic mass spectrometry can be performed after laser capture microdissection of Congo red-positive areas from slides obtained from paraffin-embedded tissue or on protein extracted from the whole sample, greatly improving diagnostic accuracy. This technique has become the gold standard in many countries. 19 Gene sequencing can help to rule out or confirm possible hereditary amyloidosis. Cardiac scintigraphy with bone tracers can differentiate AL from other types of amyloidosis and can spare cardiac biopsies, particularly in patients of advanced age with senile systemic amyloidosis. Bone marrow biopsies and immunofluorescence in situ hybridization of plasma cells might offer guidance regarding further therapeutic approaches. 20
Treatments based on prognostic stratification and risk assessment are important for patients with AL amyloidosis. 9 Therapeutic regimens include autogenous SCT (ASCT), chemotherapy, targeting of amyloid deposits, and supportive therapy. ASCT is generally preferred, but only 20% of patients are eligible. Requirements for safe ASCT include an NT-proBNP of <5000 ng/mL, troponin T of <0.06 ng/mL, age of <70 years, involvement of fewer than three organs, and serum creatinine of ≤1.7 mg/dL. 21 Despite normal bone marrow findings, our patient had involvement of three organs and refused to undergo ASCT for economic reasons and personal willingness; therefore, SCT was not recommended. Accordingly, chemotherapy was a better option for his management. Chemotherapy of AL amyloidosis is based on regimens developed for multiple myeloma and requires a risk-adapted approach, with dose reductions and schedule modifications of the chemotherapeutic regimen and close monitoring of hematologic and organ responses. 22 Active agents include corticosteroids (dexamethasone, prednisone), alkylating agents (melphalan, cyclophosphamide), immunomodulatory drugs (thalidomide, lenalidomide), and proteasome inhibitors (bortezomib). 23 Combinations of these drugs have been reported, with patients showing a relatively good response and only mild adverse effects. Hematologic and organ responses were seen in 89% and 78% of patients, respectively, but neuropathy was reported in only 44%, 24 similar to the present case; therefore, we used this therapeutic strategy in an effort to improve his survival. Moreover, non-chemotherapy approaches targeting amyloid deposits and interfering with amyloidogenesis have increasingly emerged. Doxycycline, polyphenols, anthracycline 49-iodo-49-deoxydoxorubicin, epigallocatechin gallate, competitive inhibitor of serum amyloid P component, and immunotherapy with a monoclonal anti-light chain antibody (NEOD001) have been reported to improve the clinical status and promote resorption of amyloid deposits in patients with AL amyloidosis.20,25–27
Limitations
Our study had several limitations. The bone marrow examination was performed in another hospital and showed no obvious abnormalities during diagnosis; however, no original images were obtained. The results of the immunofluorescence and immunohistochemistry examinations were not obtained. Polarization microscopy and immunoelectron microscopy were unavailable, preventing further confirmation of the amyloid nature. KMnO4 oxidation alone is not sufficient to differentiate between systemic amyloid A protein (AA) amyloidosis and systemic AL amyloidosis because the resistance or sensitivity of AA deposits and AL deposits may be practically the same, limiting the specificity and sensitivity of this method. 28
Footnotes
Acknowledgments
We would like to thank the patient and his family members for their cooperation in this study. We would also like to thank Dr. Ping Lan from the Department of Nephrology, First Affiliated Hospital of Xi’an Jiaotong University for contributing to the discussion regarding the kidney biopsy.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
