Abstract
Bortezomib is a first-in-class, potent, selective and reversible proteasome inhibitor approved for the treatment of multiple myeloma (MM) and relapsed/refractory mantle cell lymphoma. In these diseases, bortezomib targets plasma cells and lymphocytes reducing tumor burden. Recently, preclinical evidence highlighted its efficacy in reducing long-lived plasma cells responsible of autoantibodies production in several models of autoimmune conditions. These findings paved the way to a number of experiences of bortezomib use in patients with various autoimmune conditions, including autoimmune hemolytic anemia (AIHA). The latter is a nice model of autoimmunity in hematology and is caused by the production of autoantibodies against erythrocytes resulting in various degrees of hemolytic anemia. AIHA is classified in warm and cold forms according to the thermal characteristics of the autoantibody, and first-line treatment mainly relies on steroids for warm cases and the anti-CD20 rituximab for cold ones. Relapsed/refractory cases are still an unmet need, and bortezomib has been proposed in this setting with intriguing efficacy. In this review, we collected available literature on bortezomib use in AIHA and in other immune-mediated hematologic and non-hematologic diseases. Overall, most experiences highlight bortezomib efficacy even in multi-relapsed/refractory patients and suggest to consider its use in AIHA after rituximab failure.
Introduction
Bortezomib is a first-in-class, potent, selective and reversible proteasome inhibitor. This drug was initially developed to treat inflammation and cachexia until its antitumor activity emerged in the late 1990s. The drug received US Food and Drug Administration approval for the treatment of patients with multiple myeloma (MM) and relapsed/refractory mantle cell lymphoma. 1 Its use in these diseases is manly based on the proapoptotic effect on plasma cells and lymphocytes. These cells are also involved in the pathogenesis of a plethora of autoimmune diseases, where they directly produce autoantibodies or sustain autoimmunity/inflammation through the release of various mediators. Therefore, the use of bortezomib in autoimmune conditions, including autoimmune hemolytic anemia (AIHA), has recently gained more and more attention and has been addressed in various preclinical and clinical studies. In this review, we collected available literature on bortezomib use in AIHA, in other immune-mediated hematologic and non-hematologic diseases. A brief description of bortezomib pharmacodynamics and pharmacokinetics is also provided, along with evidence of its effects in preclinical studies.
Bortezomib pharmacodynamics and pharmacokinetics
Bortezomib inhibits the ubiquitin-proteasome pathway that plays a prominent role in regulating cell growth, both in normal and neoplastic cells. The proteasome acts via the degradation of most intracellular proteins, required throughout cell cycle progression and mitosis. In addition, the proteasome prevents apoptosis through the degradation of nuclear factor-κB (NF-κB) inhibitory protein, IkB. NF-κB promotes cell viability by increasing the expression of apoptosis inhibitors. Therefore, the degradation of IkB activates NF-κB signaling, making cells less prone to apoptosis. Bortezomib acts by blocking the 26S proteasome subunit, a large complex composed by a 20S proteolytic core and up to two 19S regulatory subunits. The drug specifically binds to the chymotryptic site of the 20S subunit. By inhibiting the proteasome, bortezomib is able not only to induce apoptosis but also to inhibit many other processes related to ubiquitin-proteasome pathway, that is, angiogenesis, cell-cell adhesion and proliferation. 2 Specifically, in plasma cell dyscrasia and in autoantibody-mediated diseases, bortezomib may rapidly lead to protein engulfment and death of the pathogenic cell. Bortezomib is administered intravenously or subcutaneously at a starting dose of 1.3 mg/sqm on 21-day cycles: twice weekly for 2 weeks (days 1, 4, 8 and 11) followed by a 10-day rest period (days 12–21). After eight cycles, a maintenance schedule can be followed, that is, once weekly for 4 weeks (days 1, 8, 15 and 22) followed by a 13-day rest period (days 23–35). Pharmacodynamic parameters are similar with subcutaneous or IV administration. The pharmacokinetic profile bortezomib is characterized by a two-compartment model with a rapid initial distribution (half-life < 10 min), longer elimination phase (half-life > 40 h) and large volume of distribution. The drug does not pass the blood/brain barrier. Molecularly, maximum inhibition of 20S proteasome activity occurred within 1 h after bortezomib administration and recovered to baseline within 72 h. Bortezomib dissociates from the proteasome with a half-life of 20 min, demonstrating a reversible inhibition. The distribution of proteasome in tissue significantly influences bortezomib tissue selectivity and efficacy. In fact, the high abundance of proteasome in erythrocytes limits the distribution outside the blood compartment and maximizes the efficacy in the lympho-hematopoietic system. Bortezomib metabolism occurs mainly in the liver; thus, impaired hepatic function may extend the half-life of the drug. More specifically, the primary metabolic pathway of bortezomib involves multiple cytochrome P (CYP) enzymes, including CYP3A4, CYP2C19 and CYP1A2, and moderate or severe hepatic impairment causes an increase in plasma concentrations of bortezomib. In fact, 20S proteasome inhibition following 1.3 mg/sqm does not apparently change in patients with mild hepatic impairment, while patients with moderate or severe hepatic impairment should be started at reduced doses (i.e. 0.7 and 1.0 mg/sqm). Finally, renal impairment does not influence the intrinsic pharmacokinetics of bortezomib. Among toxicities, peripheral neuropathy is the most frequently registered. It is predominantly sensory, being reversible in more than half of cases after dose adjustment and in about 70% of cases after discontinuation. Other rarer side effects are hypotension, congestive heart failure, acute diffuse infiltrative pulmonary disease and reversible posterior leukoencephalopathy syndrome. Finally, hematological toxicity (thrombocytopenia and neutropenia) may be observed following the typical cyclical pattern of the administration schedule. 1
Preclinical studies about bortezomib in autoimmune diseases
The use of bortezomib in autoimmune disorders is supported by several preclinical studies (Table 1). For instance, in human and animal models of immune thrombocytopenia (ITP), bortezomib eliminated long-lived plasma cells and decreased antiplatelet antibodies, thus alleviating thrombocytopenia. 3 Several studies, involving both animal models and ex vivo human samples, are available for systemic lupus erythematosus (SLE). Bortezomib lowered immunoglobulin levels by 30%, resulting in the reduction of autoantibodies. In particular, in mice models, bortezomib plus anti-CD20 monoclonal antibody (moAb) depleted long-lived and anti-double-strand (ds) DNA-secreting plasma cells in bone marrow and spleen. This resulted in persistent reduction of anti-dsDNA antibodies, delayed nephritis and prolonged survival.4–10 In some experiences, bortezomib ameliorated both the histopathology (glomerular and tubulointerstitial damage, podocyte damage, glomerulosclerosis, immunoglobulin deposition and lymphocyte infiltration) and clinical picture (arterial pressure and albuminuria) of lupus nephritis. In mice models of rheumatoid arthritis, bortezomib exerted a proapoptotic activity in splenocytes and fibroblast-like synoviocytes, reducing their invasiveness.11,12 Ex vivo studies in patients showed that the drug inhibited the release of several NF-κB-inducible cytokines, including tumor necrosis factor–alpha (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6) and interleukin-10 (IL-10); reduced T-cell activation; and induced T-cell apoptosis. In experimental Sjogren syndrome, bortezomib suppressed Th17 differentiation in naïve T cells in culture, ameliorated saliva flow and reduced tissue destruction in the salivary glands. 13
Preclinical evidence of bortezomib effect in autoimmune diseases.

dsDNA, double-strand DNA; IFN, interferon; IgG, immunoglobulin G; IL, interleukin; ITP, immune thrombocytopenia; MG, myasthenia gravis; MS, multiple sclerosis; PKC, protein kinase C; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; SS, Sjogren syndrome; Th17, T helper 17; TNF-α, tumor necrosis factor–alpha.
Bortezomib use in AIHA.

AIHA, autoimmune hemolytic anemia; ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; B-NHL, B-cell non-Hodgkin lymphoma; CAD, cold agglutinin disease; CLL, chronic lymphocytic leukemia; CR, complete response; ES, Evans syndrome; HSCT, hematopoietic stem cell transplant; IgG, immunoglobulin G; IV, intravenous; MDS, myelodysplastic syndrome; MGUS, monoclonal gammopathy of undetermined significance; NR, non-response; PR, partial response; SCID, severe combined immunodeficiency; SLE, systemic lupus erythematosus.
Concerning autoimmune neurological diseases, bortezomib reduced anti-acetyl-choline receptor autoantibody titers, prevented ultrastructural damage of the postsynaptic membrane, improved neuromuscular transmission and decreased myasthenic symptoms in mice models of myasthenia gravis (MG). In multiple sclerosis animal model, the proteasome inhibitor reduced T-cell secreting proinflammatory cytokines, reducing NF-κB activity in central nervous system and lymphoid organs. 16 Furthermore, in cultured thymus cells from MG patients, bortezomib killed long-lived plasma cells and consistently halted the production of autoantibodies.14,15
Bortezomib use in AIHA
AIHA is caused by destruction of red blood cells (RBCs) by autoantibodies. 17 It can be primary (idiopathic) or associated with an underlying condition (secondary), including lymphoproliferative and autoimmune diseases, infections, drugs or neoplasm. Two mechanisms for RBC destruction are recognized: intravascular hemolysis (i.e. directly in the bloodstream) and extravascular hemolysis (i.e. in the spleen, liver or both, where macrophages remove autoantibody-bound RBC). AIHA is usually classified according to the thermal characteristics of the autoantibody. Warm AIHAs (wAIHA) represent about 70–80% of cases and are usually sustained by polyclonal IgG, resulting in a direct antiglobulin test (DAT) positivity for immunoglobulin G (IgG) or IgG plus complement fraction 3d (C3d). Cold AIHAs (cAIHA) are usually caused by a monoclonal IgM that fixes complement at low temperatures and cause complement-mediated RBC lysis. The DAT is typically positive with C3d alone. Finally, mixed forms may also occur when both a warm and a cold autoantibody are present, and the DAT is positive for IgG plus C3d and high-titer cold agglutinins. 18
AIHA therapy
AIHA treatment should be differentiated in warm and cold forms. wAIHA are generally managed with steroids frontline with approximately 80% of responses, sustained in 20–30% of cases only. Second-line treatment comprises the anti-CD20 moAb rituximab or splenectomy in fit young patients. Both treatments are effective in up to 70% of cases and more than half on a long-term basis.17–20 Patients refractory to rituximab can be considered for splenectomy if eligible, but surgical, infectious and thrombotic risk should be remembered and prevented. Classic immunosuppressors, such as azathioprine, cyclosporine, cyclophosphamide and mycophenolate mofetil, are now suggested in third/further lines if a clinical trial is not available.
For cAIHA, steroids are usually effective only at high unacceptable doses, so that rituximab is the preferred first line, inducing about 50–60% of responses, mainly partial. The association of rituximab with fludarabine and bendamustine improved responses although with some side effects, particularly with the former. Splenectomy is not effective and contraindicated, and classic immunosuppressant is poorly effective. Complement inhibitors are new promising therapeutic options. 19
For relapsed/refractory cases, a number of agents targeting various lymphocyte antigens have been suggested as potential therapies, including other anti-CD20 moAbs (obinutuzumab and ofatumumab) and the anti-CD52 moAb alemtuzumab. Moreover, B-cell receptor (BCR) inhibitors ibrutinib and idelalisib that have shown some efficacy in AIHA secondary to chronic lymphocytic leukemia have also been proposed. Specifically for wAIHA, fostamatinib, a spleen tyrosine kinase (Syk) inhibitor and several neonatal Fc receptor inhibitors are under active study. Finally, in both wAIHA and cAIHA, plasma cells depleting agents have also been employed, including bortezomib, and the anti-CD38 moAbs daratumumab and isatuximab that are licensed for MM.19,21
Bortezomib use in AIHA
The use of antiplasma cell therapies in AIHA is supported by their effect on short- and long-lived plasma cells that produce the autoantibodies. 22 In fact, it has been shown that patients with cAIHA harbor long-lived plasma cells within bone marrow and spleen, which have the same heavy and light chain restriction as the B-cells, consistent with a plasmacytoid differentiation of the B-cell clone. These cells are not affected by CD20-directed therapies, thus accounting for rituximab refractoriness, and may be rather targeted by bortezomib.
Bortezomib in cold type AIHA
Table 2 summarizes clinical evidence on bortezomib use in AIHA. Only one clinical trial exists with published results. 31 This is an Italian prospective multicenter, phase 2, open-label study on refractory cAIHA (NCT01696474). Twenty-one patients were enrolled and received a single course of bortezomib – 1.3 mg/sqm IV on days 1, 4, 8 and 11. Among the 19 evaluable patients, 3 achieved a complete response (CR, 15.8%) and 3 a partial response (PR, 15.8%) for an overall response rate (ORR) of 31.6%, including transfusion independence in 4 patients. Among responders, four maintained the response after a median follow-up of 16 months (range: 10–31). One patient relapsed 1 year later but obtained a second remission with a second course of bortezomib. At last follow-up, all patients were alive except one who died during off-treatment period because of a septic shock occurred 10 months after treatment start. Importantly, treatment-related toxicities, including neurotoxicity, were rarely recorded, most probably due to the low cumulative dose of bortezomib administered in this study. Regarding clinical trials, one study on bortezomib plus anti-CD20 moAb in relapsed/refractory AIHA and one in post-transplant AIHA (NCT04083014; NCT01929980) are ongoing. Beyond clinical trials, 14 reports described the use of bortezomib in AIHA, mainly including single patient case reports. In all of them, bortezomib was used in relapsed/refractory patients, mostly pretreated with two or more lines.
Case reports on cAIHA comprise six patients, all adults, that received variable doses of IV or subcutaneous bortezomib (1.3 mg/sqm from 1 to 13 courses), either single agent or in combination with rituximab-dexamethasone (N = 1) or cyclophosphamide-dexamethasone (N = 1).22,23,32,33All patients responded, CR was obtained in four cases (66.6%) and PR in two cases (33.4%). In this setting, bortezomib appeared well tolerated without adverse events potentially attributable to proteasome inhibition. One patient died for a metastatic carcinoma, while AIHA was in PR.
Bortezomib in warm type AIHA
Concerning wAIHA, 16 adult patients who received variable doses of IV or subcutaneous bortezomib (1.3 mg/sqm from one to three cycles) have been reported.22,24,25,29,34,35 Bortezomib was administered with dexamethasone in eight patients, in combination with cyclophosphamide and dexamethasone in two, and in one case with rituximab. On the whole, ORR was 75%, with five CR (37.5%) and seven PR (52.5%). Four patients did not respond: all were multi-treated wAIHA, one post-hematopoietic stem cell transplant (HSCT) and one of them died for massive hemolysis. Bortezomib appeared well tolerated: four patients experienced G1 thrombocytopenia and G3 neutropenia, one patient self-limiting low-grade diarrhea and only two had mild peripheral neuropathy. There was an episode of grade 3 hepatotoxicity in the context of high alcohol intake which necessitated bortezomib cessation.
Finally, two patients have been described with mixed form AIHA and two patients without available DAT.27,30 The two mixed AIHA cases were secondary to SLE and HSCT, an adult and a pediatric patient; received bortezomib single agent 1.3 mg/sqm weekly for 4 doses; and obtained CR without toxicity. Two pediatric patients without available DAT were treated with variable doses of bortezomib from one to six cycles, with success and with only mild toxicities (increased ileal stoma output, G1-2 neutropenia, thrombocytopenia and transient rise in transaminase values).26,36 Finally, four cases of Evans syndrome (ES, association of AIHA and ITP) have been reported, two pediatrics and two adults.26,28,35 They were treated with bortezomib in combination with steroids and, in one patient, with vincristine. All patients responded, but one who died for sepsis with multiorgan failure. Mild peripheral neurotoxicity was experienced in one case.
Overall, bortezomib appears effective and well tolerated in multi-treated AIHA, although only case reports are available in warm cases and a single clinical trial for cold ones. The drug was administered either subcutaneously or intravenously, and generally for less cycles than those used in MM.
Bortezomib use in other hematological autoimmune diseases
Fourteen reports described the use of bortezomib in other autoimmune hematological diseases, mainly including single patient case reports, in relapsed/refractory setting (Table 3).
Bortezomib use in autoimmune hematologic disease other than autoimmune hemolytic anemia.

AIN, autoimmune neutropenia; AH, acquired hemophilia; CR, complete remission; ES, Evans syndrome; HLA, human leukocyte antigen; HSCT, hematopoietic stem cell transplant; ITP, immune thrombocytopenia; IV, intravenous; NR, non-response; PEX, plasma exchange; PR, partial remission; SC, subcutaneous; TTP, thrombotic thrombocytopenic purpura.
Bortezomib in autoimmune cytopenias other than AIHA
Eight cases of ITP have been described, six pediatrics and two adults.22,26,48 They were treated with bortezomib from less than one to two cycles, and five patients responded (four pediatric and one adult). The non-responding adult patient died for uncontrollable bleeding. Notably, two responding cases were ES and required additional treatments (plasma exchange and cyclosporine A). Based on this evidence, a clinical trial of bortezomib in ITP is currently ongoing (NCT03013114). Regarding autoimmune neutropenia, three pediatric cases have been reported. 26 They were treated with bortezomib as single agent, and all patients responded. Notably, one patient experienced febrile neutropenia and Clostridium difficile colitis 29 days after the first dose of bortezomib, and one cellulitis at the injection site, both requiring IV antibiotics.
Bortezomib in autoimmune hemostatic disorders
Eighteen cases with thrombotic thrombocytopenic purpura (TTP) have been described, 17 adults and 1 pediatric.38–47 Two cases were secondary to either checkpoint inhibitors or Sjogren syndrome, and 17 patients (94%) responded. One patient refractory to sc bortezomib responded to IV infusion. Overall, toxicities were mild, except for one patient who discontinued treatment after 13 doses of bortezomib due to worsening interstitial lung disease. Of note, TTP remained in remission after discontinuation. One patient with acquired hemophilia has been described, treated by four cycles of bortezomib obtaining CR. 22 Finally, bortezomib has been effectively used to abrogate anti-human leukocyte antigen (HLA) antibodies in patients subjected to kidney allograft, in combination with IV methylprednisolone, plasmapheresis and rituximab. 37
Overall, early evidences of bortezomib use in hematological autoimmune diseases other than AIHA show promising efficacy with low toxicity and warrant future prospective studies.
Bortezomib use in other autoimmune diseases
Several reports describing bortezomib use in autoimmune conditions are summarized in Tables 4 to 6.
Bortezomib use in systemic autoimmune diseases.

APS, antiphospholipid syndrome; BM, bone marrow; CR, complete remission; Ig, immunoglobulins; IV, intravenous; MM, multiple myeloma; NR, non-response; PC, plasma cell; PR, partial remission; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus.
Bortezomib in systemic connective tissue diseases
Forty-seven adult cases with SLE treated with bortezomib have been described (Table 4).10,49–53,55 In particular, 17 patients suffered from lupus nephritis, and 2 had underlying MM. Bortezomib was administered from one to four cycles, mostly in combination with steroids, and all patients had a clinical improvement. Toxicity was limited, with only mild peripheral neurotoxicity and six infections (one herpes zoster and five G3-G4 fever). Three adult patients with rheumatoid arthritis concomitant to MM received bortezomib in combination with steroids from three to eight cycles with joint disease improvement. Finally, one adult patient with refractory Sjogren syndrome was successfully treated with bortezomib without toxicity. 54 A clinical trial has been designed to evaluate bortezomib use in antibody-mediated autoimmune diseases, including adult patients with treatment-refractory MG, SLE and rheumatoid arthritis. 56
Bortezomib in autoimmune neurologic diseases
Seventeen cases of refractory anti-N-methyl-
Bortezomib use in neurologic autoimmune diseases.
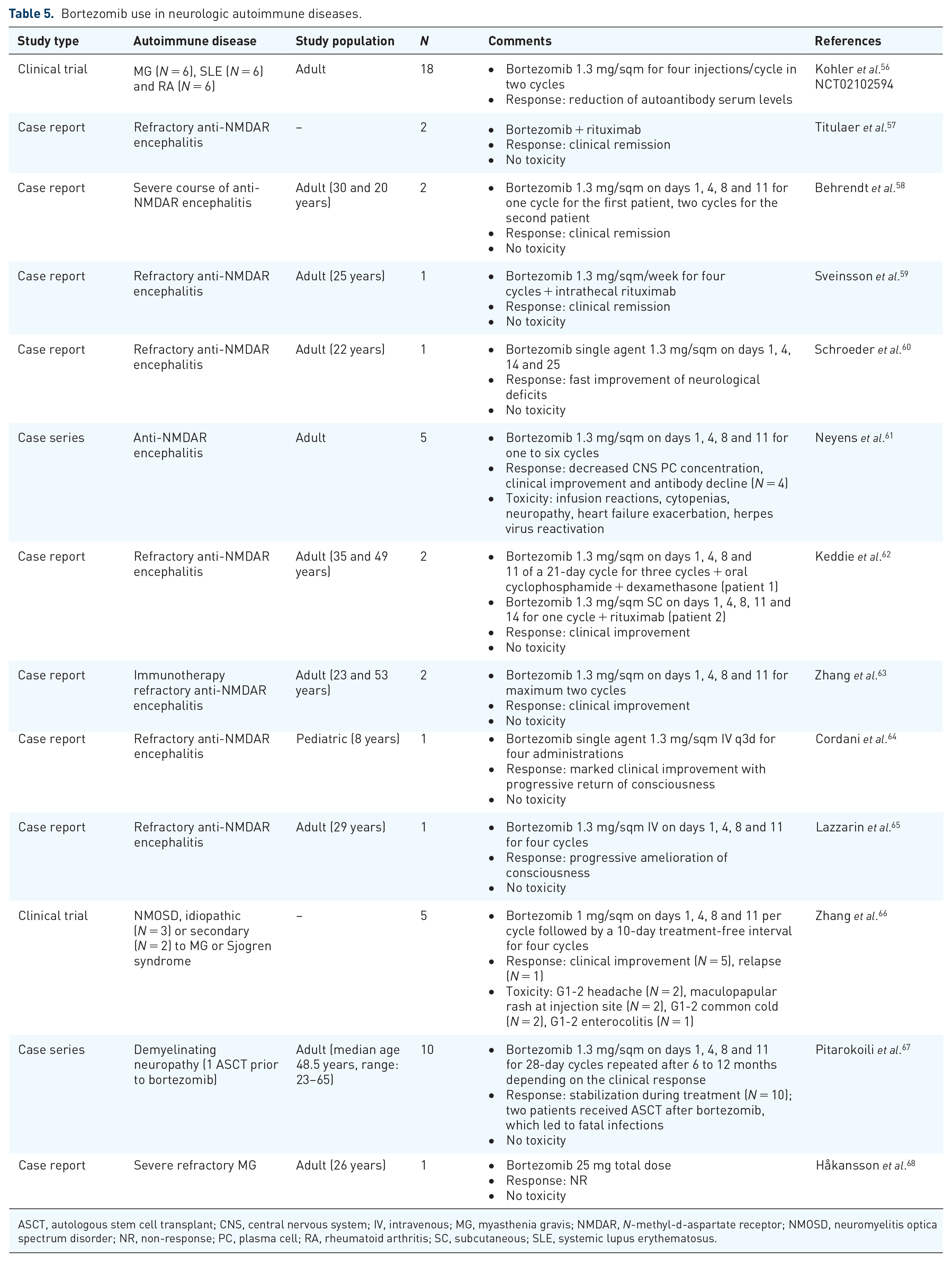
ASCT, autologous stem cell transplant; CNS, central nervous system; IV, intravenous; MG, myasthenia gravis; NMDAR, N-methyl-
Bortezomib in autoimmune renal diseases
There are several case reports of adult patients with immune-mediated nephropathy treated with bortezomib (Table 6).69–73 The latter was administered as single agent or in combination with cyclophosphamide and dexamethasone, and about 60% of patients responded. Two patients suffering from nephrotic syndrome were treated with bortezomib in combination with thalidomide and dexamethasone, or with ramipril and steroids, respectively. The first patient obtained disease stabilization, while the second achieved CR. Another case report described a patient with IgA vasculitis secondary to an IgA smoldering myeloma that obtained CR after two courses of bortezomib plus lenalidomide and dexamethasone, paralleled by the disappearance of the monoclonal protein. 70 Finally, one adult patient with microscopic polyangiitis was successfully treated with bortezomib without toxicity.
Bortezomib use in renal and cutaneous autoimmune diseases.
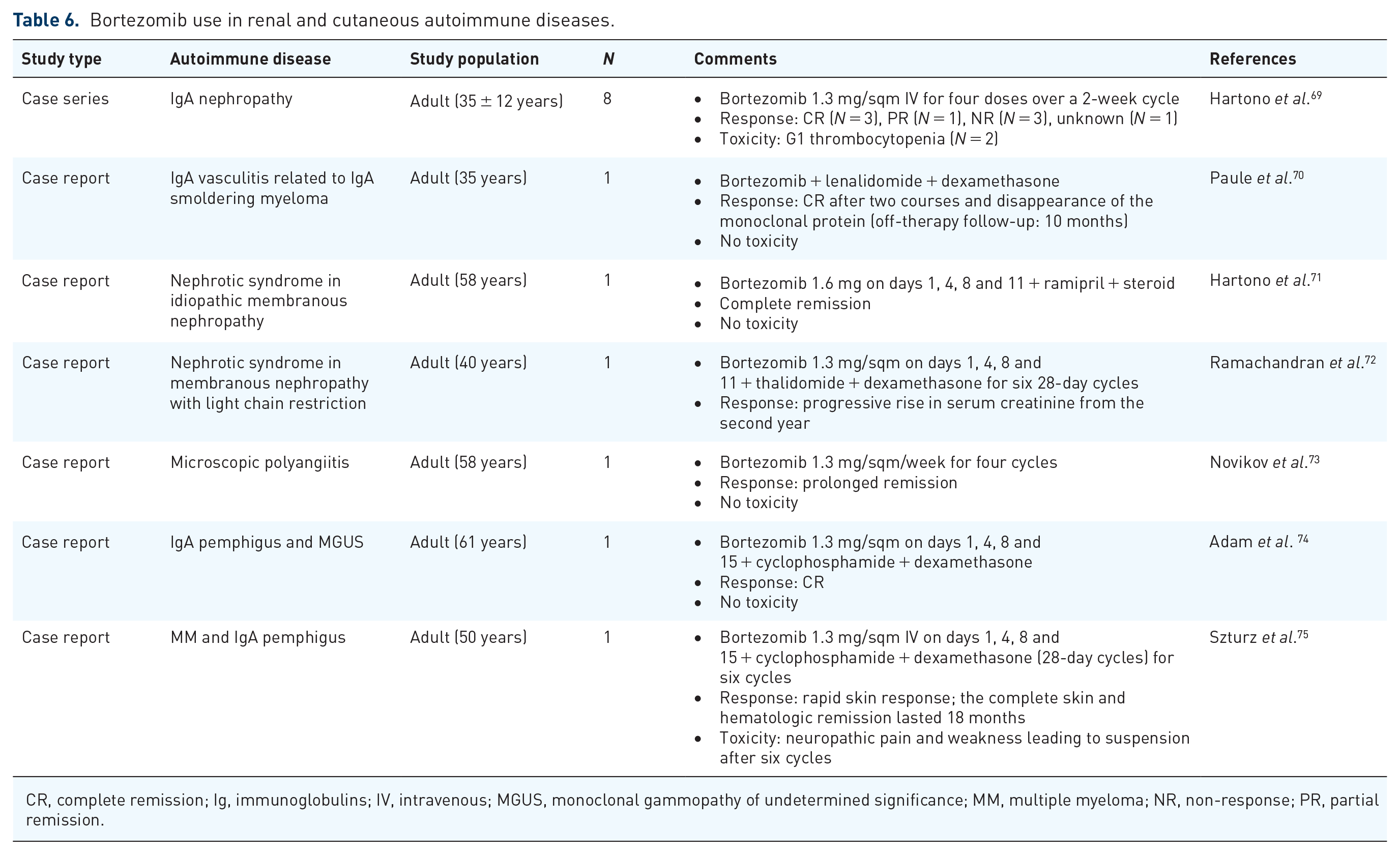
CR, complete remission; Ig, immunoglobulins; IV, intravenous; MGUS, monoclonal gammopathy of undetermined significance; MM, multiple myeloma; NR, non-response; PR, partial remission.
Bortezomib in autoimmune cutaneous disease
Two patients with IgA plasma cell dyscrasia and cutaneous pemphigus were successfully treated with bortezomib in combination with cyclophosphamide and dexamethasone (Table 6). Both patients had cutaneous clinical improvement, and one experienced peripheral neuropathy.74,75 Notably, these two patients likely suffered from monoclonal gammopathy of clinical significance (MGCS), a well-recognized entity that can also occur with cutaneous manifestations. The latter may have therefore improved as the plasma cell dyscrasia improves, as observed with other paraneoplastic autoimmune conditions.
Overall, bortezomib appears effective in organ- and non-organ-specific autoimmune conditions, including cases secondary to plasma cell dyscrasia, both as single agent or in combination with cyclophosphamide and dexamethasone.
Conclusion
Bortezomib is effective not only in myeloma but also in several heterogeneous autoimmune diseases. In AIHA, bortezomib efficacy has been reported in warm, cold and mixed forms, in adult and in pediatric patients. The frequency of responses is difficult to evaluate, since bortezomib has been often used in combination with other treatments, particularly steroids. Moreover, most articles are case reports that usually describe responding patients only, thus limiting response analysis. Beyond these limitations, bortezomib may fill the gap of treatment need in relapsed/refractory wAIHA patients, particularly when rituximab and splenectomy are ineffective or contraindicated. The drug may be useful even in cAIHA after failure of rituximab (alone or in combination) and possibly after upstream complement inhibitors that are, however, not yet commercially available. In fact, the drug exhibited a good toxicity profile in AIHA, with only low grade and mostly transient adverse events, and the number of cycles required appears lower than in myeloma patients. Finally, similarly to what described for rituximab, bortezomib cycles can be repeated with further response.
In addition, many reports show the utility of bortezomib in a plethora of other autoimmune diseases, suggesting that the drug targets a common pathogenic mechanism in these conditions. In fact, preclinical data showed that the drug reduces the number of long-lived plasma cells along with autoantibody production, and also exerts an immunomodulatory activity on several cytokines. Most of the reported experiences highlighted the need of additional therapies, targeting various pathogenic pathways in these complex conditions. Contrary to neoplastic clones that are mono- or oligoclonal, autoimmune diseases are characterized by the derangement of several polyclonal compartments including B-cells, T-cells and innate immunity that may be ‘benign’ but more difficult to immunomodulate.
Footnotes
Author Note
Bruno Fattizzo is now affiliated to Hematology Unit, Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, Milan, Italy.
Author contributions
R.P. and B.F. reviewed the literature and wrote the article. J.A.G. and W.B. revised the manuscript for important intellectual content.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
