Abstract
Background:
Patients with immune thrombocytopenia (ITP) are at risk of bleeding and, paradoxically, thromboembolic events (TEEs), irrespective of thrombocytopenia. The risk of thrombosis is increased by advanced age, obesity, and prothrombotic comorbidities: cancer, hyperlipidemia, diabetes, hypertension, coronary artery disease, and chronic kidney disease, among others. Certain ITP treatments further increase the risk of TEE, especially splenectomy and thrombopoietin receptor agonists. Spleen tyrosine kinase (SYK) is a key signaling molecule common to thromboembolic and hemostatic (in addition to inflammatory) pathways. Fostamatinib is an orally administered SYK inhibitor approved in the USA and Europe for treatment of chronic ITP in adults.
Methods:
The phase III and extension studies included heavily pretreated patients with long-standing ITP, many of whom had risk factors for thrombosis prior to initiating fostamatinib. This report describes long-term safety and efficacy of fostamatinib in 146 patients with up to 5 years of treatment, a total of 229 patient-years, and assesses the incidence of thromboembolic events (by standardized MedDRA query).
Results:
Platelet counts ⩾50,000/µL were achieved in 54% of patients and the safety profile was as described in the phase III clinical studies with no new toxicities observed over the 5 years of follow-up. The only TEE occurred in one patient (0.7%, or 0.44/100 patient-years), who experienced a mild transient ischemic attack. This is a much lower rate than might be expected in ITP patients.
Conclusion:
This report demonstrates durable efficacy and a very low incidence of TEE in patients receiving long-term treatment of ITP with the SYK inhibitor fostamatinib.
ClinicalTrials.gov identifiers:
NCT02076399, NCT02076412, and NCT02077192.
Keywords
Introduction
Immune thrombocytopenia (ITP) is characterized by increased bruising and bleeding events, attributed to low platelet counts. Paradoxically, patients with ITP have a small increased risk of both venous and arterial thromboembolic events (TEEs) compared with the general population.1–3 The risk of TEE appears to be further increased in ITP patients who are over 60 years of age, male, and/or obese (BMI ⩾ 30), as well as in those with prothrombotic comorbid conditions such as cancer, hyperlipidemia, diabetes, hypertension, coronary artery disease, and chronic kidney disease. In other words, the same risk factors are operative in patients with ITP as in patients who do not have ITP. Increased risk of TEEs is also associated with the presence of antiphospholipid antibodies or autoimmune hemolysis (AIHA).3–6 For ITP patients treated with thrombopoietin receptor agonists (TPO-RAs), both arterial and venous thrombotic events are increased (venous more than arterial). The platelet count itself does not influence the risk of venous thromboembolism but arterial events tend to occur at near normal, normal, or high platelet counts.7,8 Other treatments for chronic ITP, beyond TPO-RAs, have not been well studied except for an increased risk of TEE after splenectomy.2,9
Spleen tyrosine kinase (SYK), a 72-kDa signaling protein, is important in the thrombocytopenic events associated with ITP. 10 SYK is required for FcγRIIIA, FcγRI, and FcγRIIA receptor signaling in macrophages, which is integral to phagocytosis of antibody-coated platelets.10–14 In a mouse model of ITP, administration of the active metabolite of fostamatinib (R406) blocked reduction in platelet count caused by administration of an anti-platelet antibody similar to findings with intravenous immunoglobulin. 15 Use of fostamatinib in ITP has been based on this finding and the accompanying phase II pilot study.
SYK is also involved in platelet activation and clot formation. 16 Blocking collagen-induced platelet glycoprotein VI (GPVI) signaling in SYK-deficient mice or with a SYK inhibitor reduced platelet activation and decreased the occurrence of thrombosis in several models of arterial thrombosis, including FeCl3 injury, mechanical injury of the aorta, and carotid artery photochemical injury.17–20 Both SYK inhibition and SYK deficiency reduced the size of brain infarctions with minimal effect on normal hemostasis. 20 Thus, SYK signaling plays a role in the onset of TEEs, and blocking it may reduce thrombosis without a significant increase in bleeding.21,22 Of note, a decrease in bleeding events was observed with fostamatinib in the phase III and long-term extension studies.23–25
Fostamatinib is an orally administered, potent SYK inhibitor approved in the USA and Europe for treatment of thrombocytopenia in adults with chronic ITP.13,23–25 Treatment with fostamatinib has also demonstrated activity in rheumatoid arthritis (RA), chronic lymphocytic leukemia (CLL), and warm antibody AIHA.26–30 Furthermore, three studies of fostamatinib for treatment of COVID-19 have been launched unrelated to the effects on thrombocytopenia. These and other findings emphasize the strong anti-inflammatory effects of inhibition of SYK.
The studies cited above in RA and CLL have established a substantial database of the safety profile of fostamatinib in over 4000 patients. Clinical trials in ITP thus far have included a phase II clinical trial of 16 patients; 15 two randomized, double-blind, placebo-controlled, phase III, multicenter clinical trials of 75 patients each; 24 and an ongoing, open-label extension study for patients from the phase III trials. 25 A recent report demonstrated that response rates were better in patients earlier in their ITP course and/or who had received fostamatinib as second-line treatment. 23
The report here describes long-term safety and efficacy in patients with up to 5 years of continuous fostamatinib treatment. In particular, we describe the remarkably infrequent occurrence of TEEs during the fostamatinib clinical trials in 146 patients with ITP based on 229 patient-years of treatment experience.
Methods
This report includes patients who received fostamatinib during one of two randomized, double-blind, placebo-controlled, phase III, multicenter trials and those in the long-term, open-label extension. The data cutoff date was December 2019. All patients provided written informed consent. These studies were approved by independent ethics committees and implemented in accordance with the Declaration of Helsinki of 1975 and 2008. The methods and patient population have been described elsewhere and will be briefly summarized below.24,25
Patients
The randomized trials included adult patients with ITP who had failed ⩾1 prior ITP treatment. The extension study enrolled patients who completed 24 weeks of treatment or who completed ⩾12 weeks of double-blind treatment and discontinued due to platelet count <50,000/µL.
Patients were excluded if they had a known history of coagulopathy including prothrombotic conditions such as Factor V Leiden, Activated Protein C resistance, Antithrombin-III deficiency and lupus anticoagulant, or arterial or deep venous thrombosis within 6 months prior to randomization. In patients with deep venous thrombosis greater than 6 months prior to randomization, anticoagulants must have been discontinued for at least 30 days and subsequent D-dimer must be within normal limits.
Treatment
Patients started fostamatinib at 100 mg b.i.d. and could increase the dosage to 150 mg b.i.d. after week 4 or later, based on platelet count and drug tolerability. The dose could also be tapered if needed for tolerability.
Endpoints and statistical analysis
Endpoints included the number and percentage of patients with an increase in platelet counts that met set thresholds (⩾50,000/µL and ⩾30,000/µL); who received rescue therapy; and who had adverse events. Adverse events were coded using the Medical Dictionary for Regulatory Activities (MedDRA), and standardized MedDRA queries (SMQ) were used to search the safety database for specific types of events, such as TEE, bleeding-related adverse events (hemorrhage), hypertension, and diarrhea. Three different Embolic and Thrombotic Events SMQs were used, including 1248 terms. Reporting of specific patient events was based on the detail and verbatim terms provided by the clinical study sites.
Results
One hundred forty-six patients received fostamatinib in the phase III and open-label extension studies, with a total of 229.4 patient-years of exposure to fostamatinib as of December 2019. At baseline, median age was 53 years, median duration of ITP 8 years, and median platelet count 16,000/µL. All patients had received at least one, median 5, prior ITP treatments including corticosteroids (94%), thrombopoietic agents (47%), rituximab (32%), and splenectomy (35%). The mean duration of fostamatinib exposure was 19 months (range <1–61.7 months), and 58 (40%) patients had ⩾12 months of treatment, with 36 ongoing as of December 2019. Reasons for study discontinuation included lack of/loss of response in 50 (34%), adverse events in 30 (21%), subject decision in 14 (10%), non-compliance in three (2%), investigator decision in two (1%), lost to follow-up in one (<1%), and other in 10 (7%).
Efficacy
At least one platelet count ⩾50,000/µL attributable only to fostamatinib was achieved by 79 (54%) of 145 evaluable patients, including 64 (44%) whose platelet count exceeded 50,000/µL by week 12. The median platelet count of on-study patients increased over time and remained 50,000–150,000/µL for the duration of treatment (up to 52 months as of data cutoff) (Figure 1). A platelet count ⩾30,000/µL was achieved by 101 (70%) patients, 91 (63%) by week 12. The 37 patients with platelet counts exceeding 30,000/µL (but not 50,000/µL by week 12) had a more gradual response to treatment, and median post-baseline platelet counts tended to stabilize between 30,000/µL and 50,000/µL (Figure 1). The remaining 44 (31%) patients had very low baseline platelet counts (median 5,000/µL), received limited or no benefit from treatment, and discontinued usually within 6 months.

Platelet count over time in patients with an early response (platelets ⩾50,000/µL by week 12) in dark blue, patients who showed clinical benefit without an early response (platelets ⩾30,000/µL) in green, and patients with limited or no benefit (platelets <30,000/µL) in red. Shaded areas represent the 25th to 75th percentiles. Platelet counts affected by rescue therapy are not included. The number of patients contributing data at each time point is shown below the graph. Data points with ⩽5 patients are shown by a dotted line.
Comparing baseline characteristics between the 79 who achieved platelet counts ⩾50,000/µL (“Responders”) and the 66 who did not, responders had a shorter duration of ITP (6 versus 11 years), higher baseline median platelet counts (20,000/µL versus 10,000/µL), and less prior exposure to TPO-RA (37% versus 60%), rituximab (24% versus 42%), and splenectomy (20% versus 52%).
Thirty-six patients continue to receive treatment, 34 of whom have platelet counts exceeding 50,000/µL, with median duration of fostamatinib treatment of 49 months (range 42–62 months) and median post-baseline platelet count of 83,250/µL. Twenty-two patients discontinued therapy after at least 1 year of fostamatinib: nine (6%) discontinued due to an adverse event, seven (5%) due to subject decision, five (3%) due to loss of response, and one (<1%) due to non-compliance.
Safety: thromboembolic events
Risk factors for thrombosis were identified in 127 (87%) patients (Table 1), and 85 had multiple risk factors (median 2; range 0–7). Three patients had a prior TEE, including two deep vein thromboses (DVTs) and one lower limb thrombosis. Seventeen patients (12%) were on hormonal therapies, and one each had chronic kidney disease (stage 3) and positive antiphospholipid and anticardiolipin antibodies. History of (or current) smoking was not recorded; other factors are presented in Table 1.
Baseline patient characteristics and risk factors for thromboembolic events (TEEs).

Fifteen (10%) males were over age 65 years.
Breast cancer in three, endometrial cancer in two, colorectal cancer, non-melanoma skin cancer in five.
ITP, immune thrombocytopenia.
Despite TEE risk factors in many of the 146 patients treated with fostamatinib for up to 5 years with 229 patient-years total, only one minor (0.7%) TEE was observed.
A 61-year-old female patient (BMI >30) had a mild transient ischemic attack (TIA) on day 80 with a platelet count of 245,000/µL judged by the site investigator to be unrelated to study drug ongoing at a 100-mg single daily dose of fostamatinib. She received IV fluids and increased dose of antihypertensive medication with rapid TIA resolution. The patient had a 10-year history of ITP with splenectomy 5 years earlier and also had coxarthrosis, atherosclerosis, hypertension, and chronic obstructive pulmonary disease. Apart from mild nasopharyngitis (days 1131–1135) and the TIA, the patient had no other adverse events (AEs) during the study. The last platelet count was 262,000/µL at month 42, above 100,000/µL at 91% of visits during 42 months of fostamatinib treatment at 100 mg QD.
No other TEEs were identified among the 146 ITP patients with 229 patient-years of ongoing fostamatinib exposure.
Safety: other adverse events
Serious AEs occurred in 45 (31%) patients including four fatal AEs (Supplemental material Table S1 online): pneumonia, sepsis, plasma cell myeloma (described previously),24,25 and endocarditis (described below); none of the four fatalities were considered by the investigator to be related to treatment.
A 92-year-old patient had been hospitalized on day 1491 following a fall at home, which was accompanied by fever (102°F). She was found to have chronic microvascular ischemic changes on computed tomogram of the head (day 1501), not reported as an adverse event; the scan also revealed a 5-mm acute right frontal lobe hemorrhage. The bleed was managed with platelet transfusion, and aspirin was initiated for the ischemic changes. During hospitalization, she developed urosepsis, bacteremia secondary to endocarditis, cellulitis, acute kidney injury, atrial fibrillation, and pneumonia and died due to Morganella bacteremia endocarditis (day 1517).
Serious AEs to April 2017 were previously described; 25 additional serious AEs since that time include thrombocytopenia (two patients), sepsis (one patient), transaminases increased (one patient), and other events in one patient each.
AEs were reported in 89% of patients, of which 69% were mild to moderate. The most common AEs were diarrhea, hypertension, nausea, and epistaxis; 10% had elevated alanine aminotransferase (ALT) (Table 2). Treatment-related AEs led to dose reduction in 13 (9%) patients and dose interruption in 24 (16%). Dose modifications were most commonly due to diarrhea, increased ALT, and hypertension. Thrombocytosis was reported in two patients, resulting in dose reduction in one patient (397,000/µL) and dose interruption in the other patient (440,000/µL).
Common (⩾10% of patients) adverse events by severity in the fostamatinib exposure population (229 patient-years of fostamatinib) and in the randomized population (29 patient-years of fostamatinib and 12 patient-years of placebo).

AE, adverse event; ALT, alanine aminotransferase; Pt-years; patient-years.
The rate of AEs did not proportionally increase with the increased duration of fostamatinib treatment during the extension trial (up to 62 months of treatment and 229 patient-years). No novel toxicities developed during long-term treatment with fostamatinib.
Most bleeding AEs were mild, with epistaxis, petechiae, and contusion most common (Table 2); as in other ITP studies, bleeding AEs were generally related to the platelet count (Figure 2). Rescue therapy was administered to 61 (41.8%) patients. The rescue therapy exposure adjusted rate was 0.38 per 100 patient-years of exposure in the 79 patients whose platelet counts exceeded 50,000/µL and 2.28 per 100 patient-years in the other 67 patients.
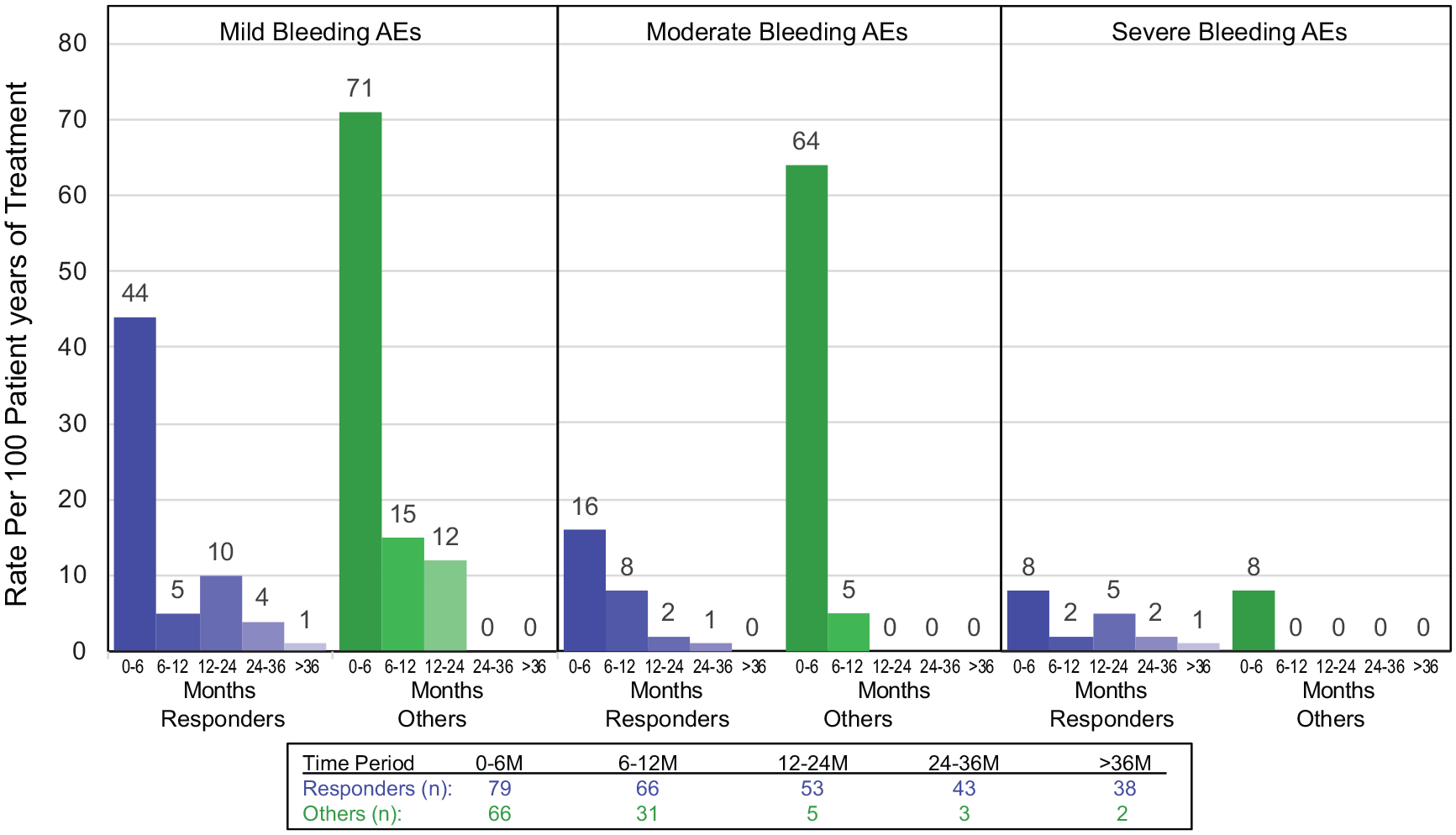
Exposure-adjusted rate of bleeding events by standard MedDRA query in responders (platelet count ⩾50,000/µL) and others (all counts <50,000/µL). Rate is per 100 patient-years and is shown in 6-month increments.
Discussion
Fostamatinib is a SYK inhibitor approved for treatment of ITP in the United States, Canada, and Europe with a different mechanism of action than other treatments. The 5-year efficacy and safety outcomes presented herein demonstrate that fostamatinib provides effective, long-term treatment for ITP without cumulative toxicity. A striking finding of this long-term extension study is the virtual absence of TEE among ITP patients treated with fostamatinib despite substantial platelet increases. The incidence of TEEs in 146 patients treated with fostamatinib for up to 5 years (229 patient-years) was 0.7%, or one episode (0.44 per 100 patient-years), and this one episode was a TIA, not a DVT, pulmonary embolism, or myocardial infarction.
Inhibition of SYK is thought to increase platelets in patients with ITP primarily by inhibiting macrophage phagocytosis. 15 SYK mediates signaling by immunoreceptor tyrosine-based activation motif receptors and is important for activation of numerous cell types in the immune system, including platelets. SYK phosphorylation and activation have been observed following stimulation of (a) Fc receptors, which mediate NETosis in neutrophils; cytokine release in macrophages, dendritic cells, monocytes, endothelial cells; and activation of platelets; (b) platelet GPVI; and (c) CLEC-2 (a C-type lectin-like receptor), which support thrombus formation.16,22,31–34 Thus, SYK is positioned at the nexus of inflammatory and thrombotic responses (Figure 3).17,35 Post-hoc meta-analyses of trials involving inhibitors of other tyrosine kinases that form part of the same pathways (namely, BTK and SRC) have also shown similar reductions in both arterial and venous thrombosis compared with placebo-treated controls.36,37
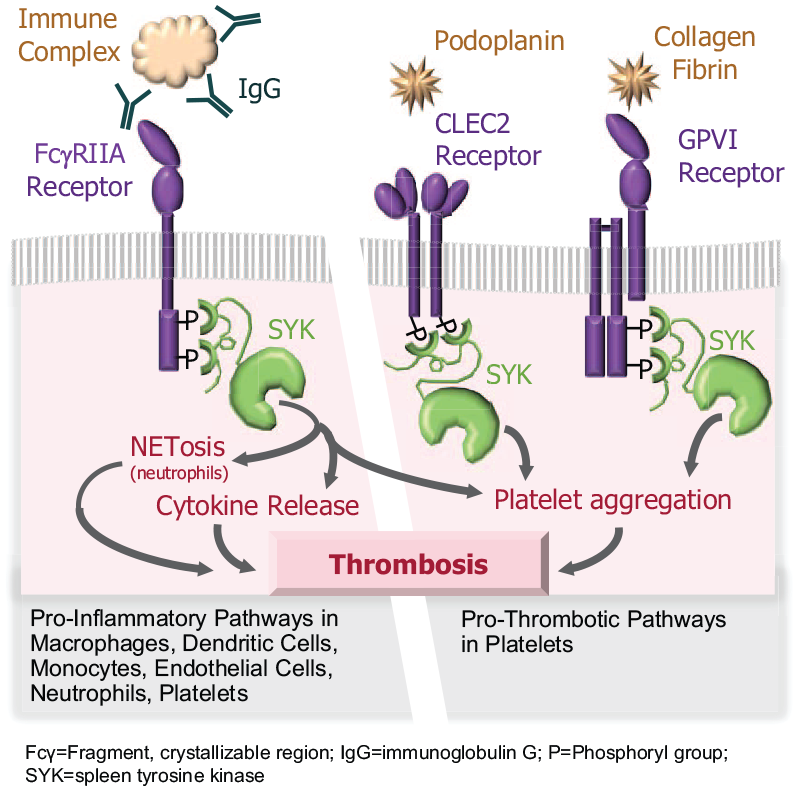
SYK-mediated pro-inflammatory and pro-thrombotic pathways in the immune system.
GPVI and CLEC-2 signal primarily via SYK; patients with TIA and stroke had significantly elevated levels of platelet GPVI expression compared with patients with non-ischemic events. 38 Conversely, patients with genetic deficiency in GPVI did not have significant disruption of hemostasis or other platelet activities.39,40 In vivo experiments have shown that SYK deficiency or administration of the active metabolite of fostamatinib (R406) and other SYK inhibitors has a negligible effect on hemostasis.20,41 Importantly, in platelet-mediated hemostasis, platelet activation and aggregation are also stimulated by thrombin, thromboxane A2, and ADP, which all signal through G protein-coupled receptors that are not dependent on signaling via SYK.21,33,42 The maintenance of hemostatic function of platelets despite SYK inhibition is likely due to these compensatory G protein-coupled signaling pathways. Therefore, suppression of SYK signaling with fostamatinib may reduce occurrence of TEEs by blocking multiple pathways contributing to NETosis, cytokine release, and platelet aggregation without substantially affecting platelet function.
ITP itself is a mildly prothrombotic disease; ITP patients have elevated endothelial cell activation and neutrophil extracellular trap (NET) generation as well as increased P-selectin and/or prothrombin fragments 1 + 2 and D-dimer, all associated with a hypercoagulable state.1–3,43,44 Certain treatments are associated with a further increase in thromboembolic risk.2,9,45 Eleven publications of TPO-RA treatment of ITP (Table 3) have reported randomized, controlled registrational clinical trials and/or their long-term rollover studies in ITP. The incidence of TEEs reported in these studies of TPO-RA ranged from 0% to 9.4% in studies up to 7 months in duration and from 2.6% to 8.9% in studies of 2–8 years’ duration (Table 3). The TEE rate per 100 patient-years was reported in three of these studies as 2.7 per 100 patient-years with eltrombopag, 46 3.1 and 3.9 per 100 patient-years with romiplostim,47,48 and a pooled analysis of 13 studies with romiplostim reported the TEE rate as 7.5 per 100 patient-years. 49 The mechanism of action of TPO-RAs is to upregulate bone marrow production of platelets, thus increasing the number and percentage of circulating “young” platelets. 50 Some studies have shown that TPO-RA may increase platelet apoptosis, formation of microparticles, and levels of both PAI-1 and P-selectin, all of which favor TEE development,43,44 especially with increased platelet production by TPO-RAs. 7 Other treatments for ITP, including corticosteroids and anti-CD20 antibody, have not been evaluated in registrational trials in ITP patients; however, long-term studies of splenectomy based on registries have demonstrated a significantly increased risk of stroke.1,51
Incidence shown as number of patients with events out of number of patients in the study.
Most patients in the fostamatinib studies had additional risk factors for TEEs (as described in Table 2). The rate of thrombosis was very low, 0.44 per 100 patient-years, when compared with the ITP studies using TPO-RAs and not due to selective difference in entry criteria (Supplemental Table S2).46–48,52–60 It should be noted that the fostamatinib studies and nearly all of the TPO-RA studies excluded patients with certain risk factors for thrombosis, including a history of thromboembolic events, cardiovascular disease, and in some studies hereditary thrombophilias; thus, rates of thrombosis in a real world population of treated ITP patients may differ. It is important to note that the low rate of thrombosis seen in patients with ITP on fostamatinib was not achieved at the expense of increased bleeding secondary to effects of fostamatinib on platelets.
The platelet response to fostamatinib combined with the very low rate of thrombosis is consistent with the mechanism of action, which is to abrogate the destruction of platelets rather than to stimulate platelet production. Blocking destruction would be expected to reduce platelet microparticles. In addition, the inhibition of platelet signaling and anti-inflammatory effects of fostamatinib both directly inhibit thrombosis. Unlike for drugs stimulating platelet production, blocking platelet destruction also minimizes the issue of thrombocytosis. Current studies of fostamatinib include a phase III trial for the treatment of warm antibody AIHA (NCT03764618), a phase III trial for hospitalized patients with COVID-19 (NCT04629703), and two open-label studies for the treatment of COVID-19 (NCT04579393 and NCT04581954); both AIHA and COVID-19 patients have increased risks of thrombosis in addition to being pro-inflammatory disorders. 37
This report highlights durable efficacy of up to 5 years of treatment with fostamatinib. Despite numerous risk factors for TEE, in addition to having ITP and 34/36 patients with ongoing fostamatinib having platelet counts greater than 50,000/µL, the incidence of TEE for patients on fostamatinib was remarkably low when compared with other ITP treatments. These data suggest the unique and novel mechanism of action of fostamatinib may offer treatment for ITP that provides increased hemostasis as a result of increased platelet count while simultaneously reducing the risk of thrombosis.
Supplemental Material
sj-pdf-1-tah-10.1177_20406207211010875 – Supplemental material for Assessment of thrombotic risk during long-term treatment of immune thrombocytopenia with fostamatinib
Supplemental material, sj-pdf-1-tah-10.1177_20406207211010875 for Assessment of thrombotic risk during long-term treatment of immune thrombocytopenia with fostamatinib by Nichola Cooper, Ivy Altomare, Mark R. Thomas, Phillip L. R. Nicolson, Steve P. Watson, Vadim Markovtsov, Leslie K. Todd, Esteban Masuda and James B. Bussel in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
Author contributions
All authors made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work. All authors drafted and/or made critical revisions to the manuscript as well as approving the final version. All authors agree to be accountable for all aspects of the work.
Conflict of interest statement
NC: Amgen: Honoraria, Speakers Bureau; Novartis: Honoraria, Speakers Bureau.
IA: Employed by Flatiron Health, Inc., an independent subsidiary of the Roche group; Bayer: Consultancy; Rigel: Consultancy; Amgen: Consultancy; Incyte: Consultancy, Research Funding; Novartis: Consultancy.
PLRN, SPW, and MRT have received a research grant from Rigel. SPW is supported by the British Heart Foundation (03/003). PLRN received research grants from Novartis and Principia Biopharma; and honoraria from Bayer.
EM, VM, and LKT are employed by Rigel Pharmaceuticals.
JBB: Novartis: Consultancy; Argenx: Consultancy; UCB: Consultancy; CSLBehring: Consultancy; Shionogi: Consultancy; Regeneron: Consultancy; 3SBios: Consultancy; Dova: Consultancy; Principia: Consultancy; Rigel: Consultancy; Momenta: Consultancy; RallyBio: Consultancy; Amgen: Consultancy.
Ethics statement
These studies were approved by independent ethics committees at participating centers and were performed in accordance with the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were funded by Rigel Pharmaceuticals, Inc. Editorial support and technical contributions were provided by Dheepika Weerasinghe and funded by Rigel Pharmaceuticals.
Informed consent
All patients provided written informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

