Abstract
Background:
Older patients with acute myeloid leukemia (AML) and myelodysplastic syndromes (MDS) unfit for intensive chemotherapy are emergent for suitable treatment strategies. Hypomethylating agents and low-dose cytarabine have generated relevant benefits in the hematological malignancies over recent decades. We evaluated the efficacy and safety of the novel treatment regimen consisting of ultra-low-dose decitabine and low-dose cytarabine, with granulocyte colony-stimulating factor (G-CSF) in this population of patients.
Methods and materials:
Patients aged more than 60 years with newly diagnosed AML/MDS were enrolled to receive therapy combined of 300 µg subcutaneously per day for priming, decitabine 5.15–7.62 mg/m2/d intravenously and cytarabine 15 mg/m2/d twice a day subcutaneously and G-CSF for consecutive 10 days every 28 days. The study enrolled 28 patients unfit for standard intensive chemotherapy. The median age of patients was 68 years (range 60–83 years) and 20 (71.4%) patients harbored AML. The primary outcome was to evaluate overall response rate.
Results:
Overall, this novel ultra-low-dose treatment regimen was well tolerated, with 0% of both 4- and 8-week mortality occurrence. Objective response rate (CR + CRi + PR in AML and CR + mCR + PR in MDS) was 57.1% after the first treatment course. Responses of hematologic improvement (HI) aspect were achieved in 18 of 28 (64.3%) patients, 11 (39.3%), 12 (42.9%), and eight patients (28.6%) achieved HI-E, HI-P, HI-N, respectively.
Conclusions:
Untreated elderly with AML/MDS were well tolerated and benefited from this novel ultra-low-dose treatment regimen.
Keywords
Introduction
Myelodysplastic syndromes (MDS) are a heterogeneous group of myeloid disorders that have a variable risk of evolution into acute myeloid leukemia (AML). Both AML and MDS have generally resulted in adverse outcomes to patients, especially older patients.1–3 The main factors that result in a poor prognosis for AML/MDS patients ⩾60 years old are poor performance status, complex comorbidities, organ dysfunction and adverse cytogenetics.4,5 These characteristics mean that these older patients cannot tolerate conventional high-intensity chemotherapy, which results in higher early death rates, lower complete response rates and shorter median survival than in younger patients.6–8
Although the choice of treatment strategy for older patients with AML/MDS remains uncertain, low-intensity chemotherapies have been demonstrated to prolong survival compared with best supportive care. low-dose cytarabine (LDAC) and hypomethylating agents, including azacytidine and decitabine (2’-deoxy-5-azacytidine), have been used frequently.9–11 Novel combined induction treatment regimens consisting of novel agents or traditional chemotherapy with hypomethylating agents or LDAC have attempted to improve the response rate and survival of older patients with AML and intermediate- or high-risk MDS who are unfit for intensive chemotherapy.12–15
Hypomethylating agents are thought to reactivate hypermethylated tumor suppressor genes by inhibition of DNA methyltransferase. 16 Decitabine (DAC), which has the double effect of inducing differentiation of neoplastic cells at low dosage and cytotoxicity at high dosage, has been regarded as the frontline treatment for older AML/MDS patients according to National Cancer Center Network since 2016, especially those who are not suitable for standard intensive chemotherapy. 17 The 20 mg/m2 5-day schedule of DAC became the standard of care since showing more benefits and less toxicity compared with the original 45 mg/m2 3-day schedule of DAC. 18 Older patients with AML/MDS ineligible for intensive therapy could not tolerate well the standard schedule of DAC due to the frequent hematological toxicities. 19 A reduced schedule of 5 mg/m2 DAC for 7 or 10 days intravenously exhibited better outcome compared with the standard schedule. 20 To reduce the side effects of DAC and maintain the effects of demethylation in the meantime, ultra-low-dose decitabine has become one of the treatment choices of researchers in AML/MDS.19,21,22 Recent studies have shown that decitabine has synergistic effects with cytarabine on tumor cells and augments the regulation of natural killer cells.23,24
In this study, we used a novel induction regimen of granulocyte colony-stimulating factor (G-CSF) combined with ultra-low-dose decitabine and LDAC in older patients with AML/MDS ineligible for intensive therapy. The goal of this study was to evaluate the response, safety and survival benefits of the first treatment cycle.
Materials and methods
Study design
In this single-arm, prospective study, we enrolled patients ⩾60 years old with AML or MDS-refractive anemia with excess blasts (MDS-RAEB) who were unfit for high-intensity therapy including intensive chemotherapy and hematopoietic stem cell transplantation. This clinical program aimed to assess the efficacy and toxicity of a novel chemotherapy regimen consisting of G-CSF before low-intensity decitabine combined with LDAC. All patients recruited completed an initial routine evaluation including medical history, performance status, complete peripheral blood count, bleeding profile, blood chemistry, urinalysis, chest X-ray, electrocardiogram and echocardiography. Additional examinations required for diagnosis, such as morphology, immunophenotyping, cytogenetics, molecular cytogenetics and genome-wide analysis were also conducted in the pretreatment phase. A minimum of 20 metaphase cells from bone marrow were analyzed for their karyotype. Patients enrolled were required to undergo next-generation sequencing. The protocol was assessed by an external independent physician.
Patients
Patients aged ⩾60 years old were eligible for enrollment if they had previously untreated (1) AML (M0–M2 and M4–M7) defined by French–American–British subtypes or (2) MDS-RAEB with an International Prognostic Scoring System score of 1 or higher. The diagnoses of AML, MDS-EB-1 or MDS-EB-2 were according to the World Health Organization (WHO) 2016 classification. All patients were also required to have a performance status according to Eastern Cooperative Oncology Group (ECOG) of ⩽2, adequate organ function including an ejection fraction >50%, normal serum creatinine, liver transaminases no more than three times normal and a total serum bilirubin no more than two times normal. Assessment of comorbidities before therapy was performed according to the hematopoietic cell transplantation comorbidity index. Exclusion criteria were (1) acute promyelocytic leukemia or AML with cytogenetic abnormality t(15;17), (2) patients with prior exposure to decitabine and (3) patients who had contraindications to chemotherapy, such as uncontrolled infection or inadequate organ function. Our study was carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki). Written informed consent was obtained from all patients or their guardians before the initiation of therapy.
Treatment regimen
All patients meeting the condition were sequentially enrolled as to treatment. Patients were allocated to receive G-CSF 300 µg subcutaneously per day for priming (until their white blood cell count was 20 × 109/L). Treatment consisted of intravenous decitabine at 5.15–7.62 mg/m2/d over 1–2 h, plus subcutaneous cytarabine 15 mg/m2/d twice a day for 10 consecutive days every 28 days. The decitabine, produced by Chiatai Tianqing Pharma (China), was provided in 10 mg bottles. The overall designated dose of 10 mg decitabine per day was chosen with due consideration of cost. Best supportive care was provided as needed during the procedure. Cytomorphological and cytogenetic analyses of bone marrow were performed 4 weeks after finishing each cycle of chemotherapy. Peripheral blood counts were tested every 2 days during the therapy and per week during the treatment interval. The treatment interval was set at 4–8 weeks depending on blood count recovery, persistent disease and nonhematological toxicities. Patients would receive the subsequent treatment cycle after assessment of performance status and organ function relative to baseline values. Treatment was interrupted or delayed if patients suffered from severe nonhematological toxicity of grade 3 or worse. For patients in poor condition, a reduced dose or schedule for decitabine and/or cytarabine were permitted at their physician’s discretion. The decision to continue the treatment regimen was taken depending on the current status of the disease, severe toxicity or at the patient’s wishes.
Outcome and response criteria
The primary end point was the overall response to the first cycle of the treatment regimen. Secondary end points included safety and overall survival. The criteria for disease response were those defined by the International Working Groups for AML and MDS.25,26 The overall response included complete remission (CR), CR with incomplete hematological recovery (CRi) only for AML, marrow complete remission (mCR) for MDS and partial remission (PR). CR was defined as <5% marrow blasts without evidence of dysplasia and abnormalization of peripheral blood (a peripheral absolute neutrophil count of ⩾1 × 109/L and a platelet count of ⩾100 × 109/L). mCR was defined as <5% myeloblasts and decrease by ⩾50% over pretreatment of MDS in bone marrow. CRi was defined as CR without recovery of platelet count to 100 × 109/L or neutrophil count to 1 × 109/L. PR was defined as (1) marrow blasts >5% but decreased by at least 50% compared with pretreatment levels or (2) bone blasts ⩽5% with Auer rods. Hematologic improvement (HI) included specific responses of cytopenia lasting 8 weeks in the three hematopoietic lineages: erythroid (HI-E), platelet (HI-P) and neutrophil (HI-N). HI-E was evaluated only if patients had a pretreatment hemoglobin level <110 g/L and was defined as hemoglobin increase by ⩾15 g/L or a reduction in the requirement for transfusions of at least 4 units in 8 weeks compared with the pretreatment level. HI-P was evaluated when the pretreatment platelet count was <100 × 109/L. It was defined as an absolute increase of at least 30 × 109/L for patients with more than 20 × 109/L before therapy or an increase to >20 × 109/L and more than double the pretreatment level. HI-N was defined as at least a doubling and an absolute increase of 0.5 × 109/L when the pretreatment level of neutrophils was <1.0 × 109/L. The time to recovery of the major hematological lineages was evaluated. Recovery of neutrophils was defined as the earliest time at which the count reached 0.5 × 109/L for two consecutive days from the time at which neutropenia first appeared during the treatment. The CTEP version 5.0 of the NCI Common Terminology Criteria was utilized for reporting and grading the adverse events.
Statistical analysis
Efficacy and safety were evaluated by descriptive statistics and frequency tables. Version 22 of IBM SPSS Statistics marketed by IBM Corp., Armonk, NY, USA was used for the analysis of data. Survival curves of responders and non-responders were created using the Kaplan–Meier method. Values of p < 0.05 were considered significant.
Results
Baseline characteristics
Between January 2017 and May 2020, a total of 28 patients were enrolled in this clinical protocol, 20 (71.4%) diagnosed with AML and 8 (28.6%) with MDS-EB-1 and MDS-EB-2. The last patient was enrolled on 5 December 2019. Patient baseline characteristics are summarized in Table 1. Among the 28 patients enrolled, 21 patients (75.0%) were men and seven (25.0%) were women. The median age at enrollment was 68 years (range 60–83 years), with nine patients (33.3%) aged older than 70 years. The median ECOG performance status score was 1 (range 0–2). The median level of bone marrow blasts was 31%; 43.8% in the AML cohort and 9.8% in the MDS cohort.
Patient baseline characteristics.

AML, acute myeloid leukemia; IPSS, International Prognostic Scoring System; MDS, myelodysplastic syndromes; MDS-EB-1, MDS-refractive anemia with excess blasts-1; MDS-EB-2, MDS-refractive anemia with excess blasts-2; n, number.
Treatment administration
All patients received at least one cycle of treatment, although three of the 28 patients (10.7%) received a reduced dose of cytarabine at their physician’s discretion. The median cumulative doses of decitabine and cytarabine during the first cycle of treatment were 100 mg and 250 mg (range 100–300 mg), respectively.
Response
In all, four of the 28 patients (14.3%) achieved a CR, no CR in MDS, an AML patient (3.6%) achieved CRi, two MDS patients achieved a mCR (7.1%), nine (32.1%) achieved PR, and 12 (42.9%) had no response following the first cycle of therapy. The overall response rate (CR + CRi + PR in AML and CR + mCR + PR in MDS) was 57.1% after the first treatment course. HI responses were achieved in 18 of 28 (64.3%) patients; 11 (39.3%), 12 (42.9%) and eight patients (28.6%) achieved HI-E, HI-P and HI-N, respectively. Of the 12 (42.9%) patients not achieving CR or PR, four (33.3%) achieved improvements in their peripheral blood counts. Additional details of the responses and evaluation of hematological improvements are shown in Table 2. There was an overall response in 60% of AML patients; four of the 20 patients (20%) with AML achieved a CR, one (5%) a CRi, seven (35%) a PR and eight (40%) had no response. In this AML population, 12 patients (60%) achieved HI. Among the eight MDS patients, the overall response was 50%, with two patients (25%) achieving mCR, two PR (12.5%) and four (50%) no response. The rate of MDS patients who achieved HI was 75%. Two (7.1%) of these progressed to AML after the first cycle of treatment.
Response and Outcomes.
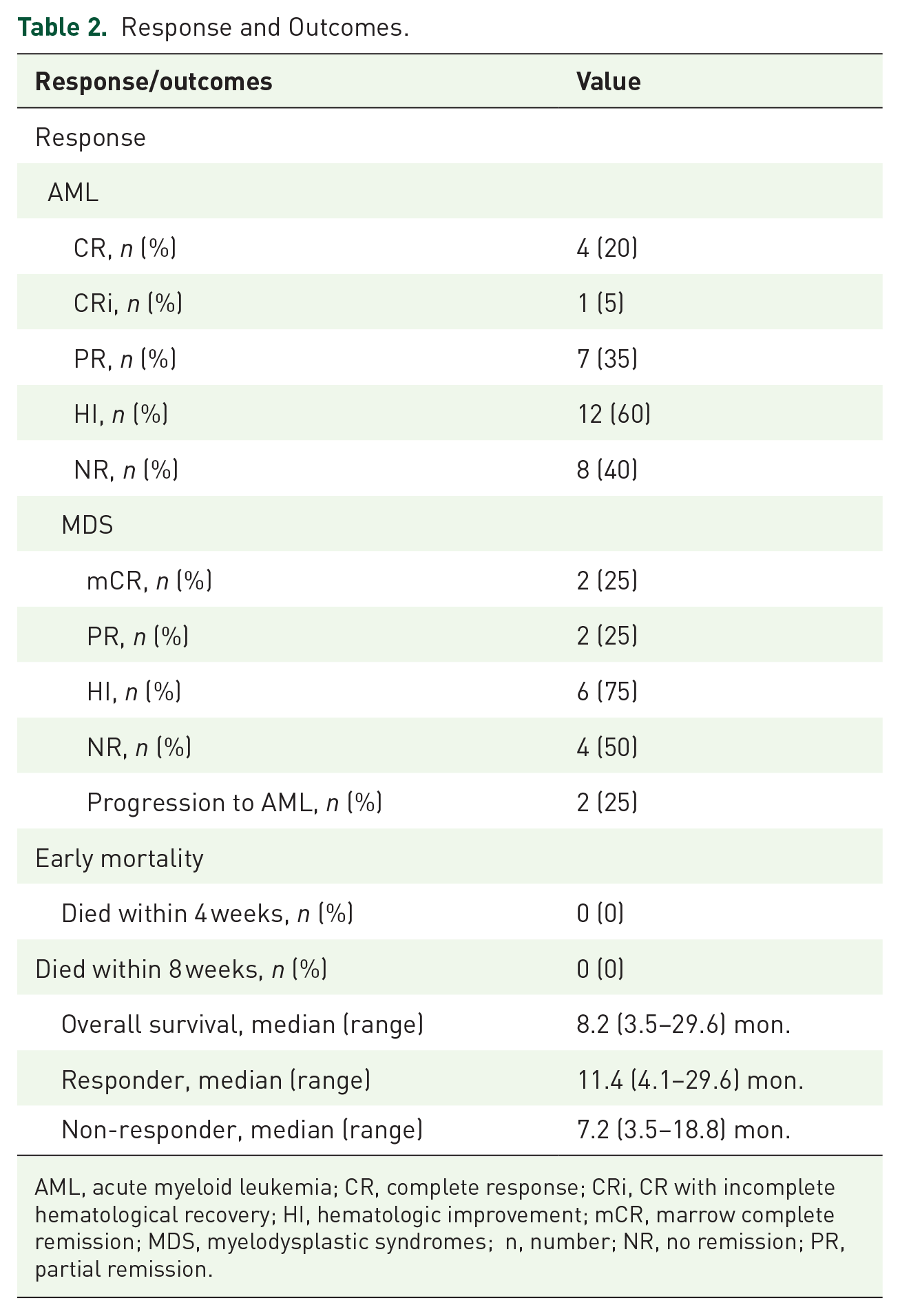
AML, acute myeloid leukemia; CR, complete response; CRi, CR with incomplete hematological recovery; HI, hematologic improvement; mCR, marrow complete remission; MDS, myelodysplastic syndromes; n, number; NR, no remission; PR, partial remission.
Safety and toxicity
All patients who finished the first cycle of treatment were evaluated for toxicity. Overall, this ultra-low-dose treatment regimen was well tolerated in the older patients unfit for intensive therapy, with 4- and 8-week mortality 0%. Patients experienced myelosuppression to some degree throughout the protocol. Nineteen (67.9%) patients had neutropenia in the pretreatment. The median pretreatment platelet count was 40.5 × 109/L and 17 (60.7%) patients were documented to have platelet counts <50 × 109/L. Initially, all patients suffered from anemia and 14 (50%) had hemoglobin ⩽80 g/L. After the first cycle of therapy, 78 instances of hematologic toxicities were recorded; 22 (28.2%) grade 3/4 thrombopenia, 14 (17.9%) anemia and 23 (29.5%) neutropenia. To assess the efficacy of the treatment regimen, hematopoietic recovery was evaluated as the proportion of neutropenia. The median time to achieve neutrophil recovery was 12 days (2–29 days) in the 19 patients with neutrophil <0.5 × 109/L. Overall, nine neutropenic responders achieved neutrophil recovery in a median time of 14 days (10–24 days). Hematological and nonhematological adverse events possibly related to treatment are summarized in Table 3. The most common nonhematological adverse events were febrile neutropenia, infection, fatigue and hemorrhage. Twenty-five incidences (40.3%) of nonhematological adverse events grade 3 or 4 were documented, among which were febrile neutropenia 12 (19.4%), infections 10 (16.1%), hemorrhage two (3.2%) and epilepsy one (1.6%). Transient grade 1 or 2 elevated total bilirubin, alanine aminotransferase and aspartate aminotransferase were documented in two (3.2%), three (4.8%) and three (4.8%) instances, respectively. No patients demonstrated increased creatinine.
Adverse events possibly related hematologic and nonhematologic toxicities.

Discussion
The majority of AML patients are older individuals, who have worse performance status, poor organ function, more comorbidities, adverse karyotype and are less willing to undergo treatment than younger patients. These factors influence the inferior response rate, more severe toxicity and even the lower overall survival rate of 10% for older patients receiving intensive chemotherapy, compared with younger AML patients.27,28 MDS, a malignant disease that has a high risk of transforming to AML, has the similar characteristics of clinical and genetic heterogeneous disorders with myeloid stem cells. 29 Therapy of older patients with AML/MDS has been actually challenging, thus less-aggressive therapies have attached scientists’ eyes. LDAC and low-dose hypomethylating agents have been considered as the frontline therapy for elderly patients with AML/MDS not candidates for intensive therapy. 30 We developed this novel therapy to improve the overall response rate and survival quality of these patients after the first cycle of induction therapy.
With the recognition of the increasing importance of epigenetics in hematological malignancies, decitabine, an inhibitor of DNA methyltransferase, has played an increasingly influential role in the treatment of older patients with AML/MDS.31–37 Recent studies have shown a trend toward the use of low-dose decitabine in AML/MDS because of its reduced toxicities.18,20,38 In addition to its therapeutic effect, decitabine has a synergistic effect on many other chemotherapeutic drugs, such as cytarabine.39,40 Based on the findings of previous studies, we developed a novel treatment regimen consisting of G-CSF combined with ultra-low dose of decitabine and low-dose cytarabine.
In this study, this novel treatment regimen was generally well tolerated in older patients with AML/MDS. We observed an overall response rate of 57.1% and an HI rate of 64.3%. The myeloid suppression was short-lived, with a median neutrophil recovery time of 14 days. The incidence of grade 3 or 4 nonhematological adverse events was 43.1%. There was zero 4- and 8-week mortality. However, because we could not conduct a suitable control arm, these data may include bias. Most of the older patients with newly diagnosed AML/MDS were unable to continue the novel therapy regimen for more than four cycles, mostly due to disease resistance, expensive cost and the inability to undergo regular treatment.
The intention of our work was to provide non-intensive treatment, prolong the overall survival and improve quality of life for patients in this older population. We recorded a median overall survival of 8.2 (3.5–29.6) months with selected subsequent treatment regimens after the first cycle, as detailed in Table 4. The overall survival of responders to the first treatment cycle (shown in Figure 1) tended to be better, but this difference was not significant (p = 0.1556), probably because the sample size was limited and the subsequent treatment differed. According to a previous study, the elderly patients with AML received LDAC of 20 mg twice daily subcutaneously for 10 days achieved a survival of 4% at 2 years. 41 LDAC has been explored in AML/MDS for more than two decades, and the most suitable dose needs to be studied sequentially.
Patient disease details, treatment and survival.

AA, regimen of Adriamycin and cytarabine; AML, acute myeloid leukemia; CAG, regimen of low-dose cytosine arabinoside, aclarubicin and G-CSF; DAC, decitabine; EA, regimen of etopol and cytarabine; HA, regimen of homoharringtonine and cytarabine; HAG, regimen of homoharringtonine, cytarabine and G-CSF; IA, regimen of idarubicin and cytarabine; LDAC, low-dose cytarabine; MA, regimen of mitoxantrone and cytarabine; MDS, myelodysplastic syndromes; none = no more subsequent treatment; the number in “()” referring to the number of treatment cycle.

Overall survival in responders and non-responders after first cycle of treatment.
Further studies are warranted to evaluate the significance of this treatment regimen consisting of G-CSF combined with ultra-low-dose decitabine and low-dose cytarabine.
Footnotes
Acknowledgements
The authors would like to express our gratitude to all those who helped us during this work.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval/patient consent
Our study achieved ethical approval from the Ethics Committee of The Third Affiliated Hospital of Soochow University. Written informed consent was obtained for all participants.
