Abstract
Background:
Thrombopoietin-receptor agonists (TPO-RAs) are used to treat immune thrombocytopenia (ITP), a disorder characterized by prolonged low platelet counts (PCs) that pose a risk of serious bleeding episodes. Avatrombopag (AVA) is the most recently approved TPO-RA for the treatment of chronic ITP. A high proportion of patients responded to AVA in clinical trials, and treatment was well-tolerated; however, limited real-world effectiveness data have been reported to date.
Objectives:
To describe demographic and clinical characteristics, treatment patterns, and outcomes following the initiation of AVA in patients with ITP in the United States.
Design:
This is a retrospective study using administrative claims data from the Komodo Healthcare Map (1 February 2017 to 28 February 2022) linked with PC laboratory data.
Methods:
Patients with ⩾1 diagnosis of ITP, ⩾1 paid prescription for AVA (index date), and ⩾1 month of pharmacy coverage after AVA initiation were selected. Baseline characteristics and follow-up steroid, immunosuppressant, and rescue medication use were described. The percentage of patients achieving clinically meaningful PC thresholds (⩾30 × 109/l) were assessed among patients with ⩾1 PC following AVA initiation and prior to AVA discontinuation/switch (effectiveness subgroup).
Results:
A total of 205 patients met eligibility criteria and 49% reported TPO-RA use in the prior 6 months. Approximately 70% and 93% of patients did not require use of steroid or immunoglobulin rescue medication during follow-up, respectively. Among patients with concomitant steroid (n = 75) or immunosuppressant (n = 7) use at AVA initiation, 35% and 57% discontinued those treatments, respectively. Of the 21 patients in the effectiveness subgroup, 81% achieved clinically meaningful PC thresholds.
Conclusion:
A high proportion of evaluable patients with ITP in this real-world study achieved clinically meaningful PCs, without requiring rescue medication during AVA treatment, with many able to discontinue baseline concomitant steroid or immunosuppressant utilization. Despite limited availability of PC data, these results are consistent with results from the AVA pivotal clinical trials.
Keywords
Introduction
Background
Immune thrombocytopenia (ITP) is an autoimmune disorder characterized by a reduction in platelet counts (PCs) in the blood, estimated to impact 5.3 per 100,000 children and 9.5 per 100,000 adults in the United States.1,2 ITP is caused by both impaired platelet production and peripheral platelet destruction. While normal PC ranges from 150 × 109/l to 450 × 109/l, PCs for ITP patients often fall below 100 × 109/l. 1 This reduction in platelets contributes to sudden increases in bleeding manifestations, which can range in severity from mild skin bruises to life-threatening intracranial hemorrhage. 3 According to the International Consensus Report on ITP and American Society of Hematology 2019 guidelines, treatment is recommended for ITP patients with PCs below 20 × 109/l to 30 × 109/l.4,5 The clinical goal of ITP treatment is to increase PCs to a level that enables patients to maintain their daily activities with a low risk of bleeding. Treatment response is measured in terms of increased PC, with response typically defined as a PC of 30 × 109/l to 50 × 109/l (and may require doubling of baseline PC) and complete response defined as 100 × 109/l.4–6
Corticosteroids and immunoglobulin therapy are recommended as first-line treatments. 5 These therapies have variable and often temporary efficacy, and long-term corticosteroid use is not recommended due to the significant side effects that often outweigh the potential benefits, with guidelines generally recommending steroid discontinuation within 6 weeks of initiation. 4 Furthermore, most adult patients relapse upon cessation of steroid treatment, and for such patients, subsequent treatment is recommended. Patients may use rescue medications such as intravenous immunoglobulin (IVIg) and steroids (i.e. dexamethasone and prednisone) to treat acute symptoms, but neither are considered long-term treatments. 7 Prior to 2008, the most common subsequent treatments for ITP patients were splenectomy and rituximab, both of which exhibit variable efficacy and can be associated with rare but life-threatening adverse events. 5
The US Food and Drug Administration (FDA) has approved three thrombopoietin-receptor agonists (TPO-RAs) for subsequent treatment of ITP, which have substantially improved the outlook for ITP patients who have not responded to first-line treatment.8,9 TPO-RAs increase platelet production by mimicking the action of endogenous thrombopoietin (TPO) on megakaryocytes and megakaryocyte precursors. 10 Eltrombopag (ELT; PROMACTA®) is an oral medication approved by the FDA in 2008. ELT is compromised when administered with polyvalent cations so it must be taken at least 2 h before or 4 h after the following: certain medications (e.g. antacids), foods containing >50 mg calcium, or supplements containing polyvalent cations.10,11 ELT also carries a boxed warning for hepatotoxicity and requires monthly hepatic monitoring and potential statin dosing adjustment. 10 Romiplostim (ROMI; Nplate®), also approved by the FDA in 2008, is administered in a medical office via weekly subcutaneous injection. 10 Avatrombopag (AVA; Doptelet®) was more recently approved by the FDA in June 2019 for the treatment of adult patients with chronic ITP who had an insufficient response to a previous treatment and has demonstrated comparable efficacy to other TPO-RAs.12–14 AVA is an oral medication taken once-daily that does not have food restrictions, hepatic monitoring requirements, or statin dose adjustment requirements. 2
All three TPO-RAs approved for the treatment of ITP have response rates >60% with persistent response over time and low rates of adverse events and thus have become mainstays of ITP treatment. 5 Given the relatively recent approval of AVA, however, there is limited real-world evidence and longitudinal data on its use and effectiveness for the treatment of ITP.
Study objectives
The purpose of this noninterventional, retrospective cohort study was to describe the real-world demographic and clinical characteristics, treatment patterns, and outcomes of patients with ITP treated with AVA in the United States.
Methods
Data source
This cohort study used administrative claims data (i.e. diagnoses, procedures, medications administered at point of care, prescription pharmacy fills) from the Komodo Healthcare Map, which encompasses 330 million patients across over 500 unique commercial, Medicare, and Medicaid payers. 15 These data included linked PC laboratory records from a large, nationally represented laboratory, Quest Diagnostics, 16 and included data over a 5-year period (February 2017 to February 2022). This is a retrospective analysis of de-identified claims data; thus, no institutional board review was required. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 17
Patient selection
For the overall sample, patients were included if they had ⩾1 paid prescription for AVA; the date of the first paid AVA prescription was defined as the index date. Patients were additionally required to have ⩾1 diagnosis for ITP (ICD-9-CM: 287.31; ICD-10-CM: D69.3) prior to the index date and pharmacy coverage on and for ⩾1 month after the index date. Patients with any paid prescriptions for AVA with 5 days of supply were excluded because a 5-day course of AVA is typically administered to patients with chronic liver disease who are scheduled to undergo a procedure and not for the treatment of ITP that was the focus on this analysis. 18 A subgroup of the overall sample, dubbed the effectiveness subgroup, included patients with ⩾1 PC available after the index date and before the first occurrence of AVA discontinuation and switch to an alternative ITP treatment.
Additional definitions
The baseline period was defined as the 6-month period before the index date. The follow-up period was defined as the period from the index date until the earliest of (1) the end of the pharmacy eligibility period that overlapped with the index date or (2) patient’s last AVA claim in the study period plus days of supply plus 90 days, due to the titration schedule with AVA, which allows for an effective dose as low as one 20 mg tablet per week. The postindex period was defined as the follow-up period, with the exclusion of the index date.
Study measures
Patient demographics were described as of the index date. For the subgroup of patients with at least 6 months of baseline medical and pharmacy coverage prior to the index date, Charlson Comorbidity Index (CCI), comorbidities, and ITP treatment use during the baseline period were described. Prior ITP treatments considered were TPO-RAs, steroids (prednisone and dexamethasone), IVIg, anti-D immune globulin, fostamatinib disodium, rituximab, and splenectomy.
Treatment switch to an alternative ITP treatment (i.e. ELT, ROMI, fostamatinib disodium, or rituximab) was assessed, with treatment switch defined as at least one claim for one of the alternative ITP treatments of interest during the follow-up period. Time to treatment switch was assessed among patients who switched. Use of rescue medications (i.e. new treatments of ⩾3-day course of dexamethasone, ⩾7-day course of prednisone, or IVIg) during the follow-up period was described, including time to first rescue medication. Use of steroids and immunosuppressants in the postindex period were evaluated. Among patients with medical and pharmacy coverage during the 1-month period before the index date, patients with concomitant steroid or immunosuppressant use were defined as patients with a steroid or immunosuppressant claim during the 1-month window up to and including the index date. Among patients with concomitant steroids or immunosuppressants, discontinuation of steroids or immunosuppressants was defined as having no claims for steroids or immunosuppressants during the postindex period.
AVA effectiveness was assessed among the effectiveness subgroup using PCs from the index date until the earliest of AVA discontinuation, treatment switch, and end of follow-up; PCs that occurred within 4 weeks after IVIg or 8 weeks after steroid rescue medication were censored as they may be inflated by these rescue medications. The following measures were assessed: (1) achievement of clinically meaningful PC thresholds (⩾30 × 109/l, ⩾ 50 × 109/l, ⩾75 × 109/l, ⩾100 × 109/l) during follow-up and before AVA discontinuation or treatment switch and (2) achievement of clinically meaningful PC thresholds among patients with ⩾2 or ⩾4 PCs during the follow-up period.
Statistical analyses
Outcomes were described using frequencies and percentages for categorical variables and mean values, standard deviations (SDs), medians, and interquartile ranges (IQRs) for continuous variables. All analyses was conducted using SAS Enterprise Guide Software Version 7.1. 19
Results
Sample selection
Of the 1260 patients in the linked database with ⩾1 paid prescription for AVA, 205 patients met the eligibility criteria (Figure 1). Among these patients, 21 patients were included in the effectiveness subgroup (Figure 1).
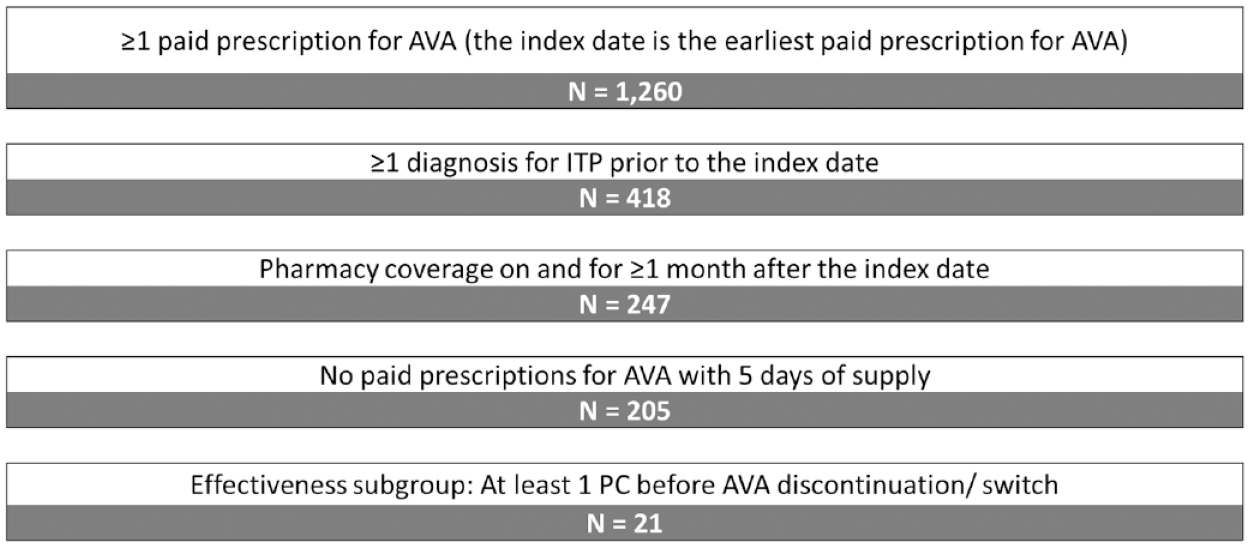
Sample selection.
Baseline characteristics
The overall study sample (n = 205) had a mean (SD) age of 52 (18) years, and 54% were female (Table 1). In this group, the most common types of health insurance were commercial (43%) and Medicaid (30%). Mean (SD) follow-up time was 246 (184) days. Patients with at least 6 months of baseline medical and pharmacy coverage (n = 172) demonstrated a mean (SD) CCI of 2 (2), and the most common CCI comorbidities were chronic pulmonary disease (17%), diabetes without chronic complications (16.3%), and rheumatologic disease (14%). Of the prior ITP treatments considered, 56% used one treatment during the 6-month baseline period, and 16% used two or more treatments. Nearly half (49%) received a TPO-RA in the 6-month period before initiating AVA, with 30% using ROMI, 25% using ELT, and 6% receiving both. Other common ITP treatments included prednisone (50%), dexamethasone (38%), and IVIg (18%).
Patient baseline characteristics.
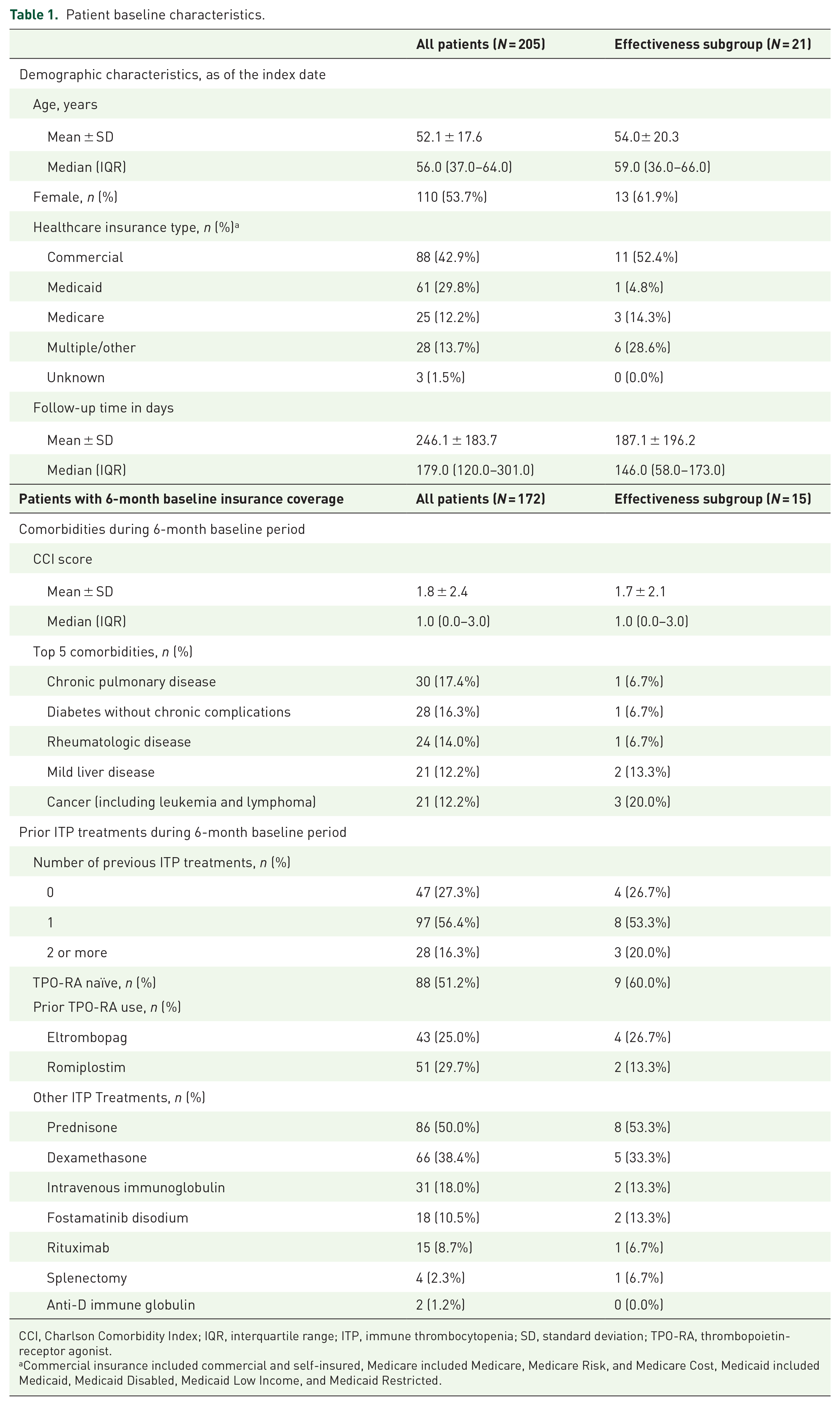
CCI, Charlson Comorbidity Index; IQR, interquartile range; ITP, immune thrombocytopenia; SD, standard deviation; TPO-RA, thrombopoietin-receptor agonist.
Commercial insurance included commercial and self-insured, Medicare included Medicare, Medicare Risk, and Medicare Cost, Medicaid included Medicaid, Medicaid Disabled, Medicaid Low Income, and Medicaid Restricted.
In the effectiveness subgroup (n = 21), the mean (SD) age was 54 (20) years, and 62% were female. The most common type of health insurance was commercial (52%); 29% had multiple insurance types. The mean (SD) follow-up time was 187 (196) days.
Patients in the effectiveness subgroup with at least 6 months of baseline medical and pharmacy coverage (n = 15) had a mean (SD) CCI of 2 (2), and the most common CCI comorbidities were renal disease (27%), peripheral vascular disease (20%), and cancer (including leukemia and lymphoma) (20%). Of the prior ITP treatments considered, 53% used one treatment during the 6-month baseline period, and 20% used 2 or more treatments. 40% had prior TPO-RA use in the 6-month period before initiating AVA, with 13% using ROMI and 27% using ELT. Other common ITP treatments in this subgroup included prednisone (53%), dexamethasone (33%), IVIg (13%), and fostamatinib disodium (13%) (Table 1).
Treatment patterns
Among the overall sample (N = 205), 76% of patients did not switch to another ITP treatment during follow-up (Table 2). Of the 49 (24%) patients that switched to another ITP treatment during follow-up, the most common was ROMI (32/49; 65%) followed by ELT (7/49; 14%) and rituximab (6/49; 12%). Among patients who switched, the median time from index date to treatment switch was 13 days (IQR = 4–56).
ITP treatment switch and rescue medication use during the follow-up period.

ELT, eltrombopag; IQR, interquartile range; ITP, immune thrombocytopenia; IVIg, intravenous immunoglobulin; ROMI, romiplostim; SD, standard deviation.
Rescue medications included new treatments of IVIg, ⩾3-day course of dexamethasone, and ⩾7-day course of prednisone.
Approximately 67% of patients did not require the use of any rescue medications during follow-up. Among the patients who required a rescue medication, 6% and 31% had evidence of IVIg and steroid rescue medication use, respectively. The median time from index to IVIg and steroid rescue medication use were 119 and 59 days, respectively (Table 2).
In the postindex period, 49% of patients did not require use of any steroids or immunosuppressant medication (Table 3); 48% used steroids and 9% used immunosuppressants. Of total, 185 patients had pharmacy and medical coverage for the 1-month period before the index date and were available to assess discontinuation of steroids and immunosuppressants (Figure 2). Among patients with concomitant steroid (n = 75) or immunosuppressants (n = 7) use at index, 35% (n = 26) discontinued steroid use while 57% (n = 4) discontinued immunosuppressant use in the postindex period (Table 3 and Figure 2).
Steroid and immunosuppressant use postindex.

Steroid medications of interest included dexamethasone and prednisone. Immunosuppressant medications included azathioprine, mycophenolate mofetil, cyclosporine (including cyclosporine A), cyclophosphamide, vinca alkaloids (vinblastine, vinorelbine, vincristine), danazol, and dapsone.

Steroid and immunosuppressant discontinuation postindex.
AVA effectiveness among effectiveness subgroup
Among the 21 patients in the effectiveness subgroup, 17 (81%) reached a PC ⩾ 30 × 109/l; 16 (76%) reached a PC ⩾50 × 109/l; 15 (71%) reached a PC ⩾75 × 109/l; and 13 (62%) reached a PC ⩾100 × 109/l, as shown in Figure 3. Among patients with ⩾2 PCs during follow-up (n = 13), 92% (n = 12) reached a PC ⩾50 × 109/l, and 85% (n = 11) reached a PC ⩾100 × 109/l. All patients with four or more PCs (n = 10) achieved a PC ⩾100 × 109/l during follow-up. As a sensitivity analysis, effectiveness analyses were conducted for the subgroup of patients with no TPO-RA use in the 6 months before the index date. Of these 9 patients, 8 (89%) reached a PC ⩾30 × 109/l; 7 (78%) reached a PC ⩾50 × 109/l; 7 (78%) reached a PC ⩾75 × 109/l; and 6 (67%) reached a PC ⩾100 × 109/l (results not shown).

Avatrombopag effectiveness during follow-up among the effectiveness subgroup.
Discussion
In this study, approximately half of all patients with ITP treated with AVA used another TPO-RA in the 6 months before initiating AVA, which may indicate more refractory ITP as well as a high proportion of patients undergoing TPO-RA class switch to AVA. The reason for switch was not captured in this retrospective real-world data set, but potential clinical reasons include lack of effectiveness, desire for improved convenience, costs, and adverse events experienced with a prior TPO-RA. After initiating AVA, the majority of patients did not switch to an alternative ITP treatment or require the use of rescue medications that may be indicative of a robust and sustained response to AVA. Patients who did require IVIg or steroid rescue medication did so within a median of 119 and 59 days, respectively. Overall, 24% of patients (49/205) initiating AVA switched treatment, often within the TPO-RA class (65% of patients who switched did so to ROMI and 14% to ELT). The median time to switch was 13 days, which was rather short for an adequate evaluation of response before initiating a switch in treatment, as the AVA prescribing information suggests titration to a higher dose occurs after 14 days of treatment when insufficient effectiveness is achieved. While the database lacked information on reasons for switch, cost-related impact due to type of insurance coverage (i.e. injectables are typically covered under medical benefits as opposed to pharmacy benefits) and tolerability-related issues may be plausible. Among patients with concomitant steroid use at AVA initiation, over 1/3 were able to discontinue steroids, suggestive of improved effectiveness with AVA administration, versus treatment regimens prior to AVA initiation. Prolonged steroid use has been linked to lower quality of life in ITP patients and treatment guidelines have recommended discontinuing steroids in a timely fashion.4,5 Of the patients with available PC data, more than 80% reached clinically meaningful PC thresholds.
These results are broadly consistent with results from prior studies of AVA. In a phase III study, among the 15 patients with chronic ITP treated with AVA who had concomitant ITP medication at baseline, 33.3% were able to reduce the use of these medications after AVA initiation, similar to the 35% who discontinued steroids and 57% who discontinued immunosuppressants in this study. In the phase III study, 0% of patients who received placebo were able to reduce their concomitant ITP medications. 13 The phase III study also demonstrated that, during the core phase, 43% of patients were able to discontinue or reduce the dose of concomitant steroids with AVA treatment, with 36% of patients discontinuing entirely. In addition, 71% of placebo patients initiating AVA in the extension phase of the study were able to reduce the dosage or discontinue steroid utilization during the extension phase, with 43% discontinuing. 15 The results from this real-world analysis were similar, as 35% of patients discontinued steroids entirely. Due to data limitations, however, this study could not evaluate reduction in steroid dose. In the phase III study, 90.6% of patients treated with AVA achieved PC ⩾30 × 109/l at any time during the study, compared with 35.3% of patients receiving placebo. 20 In one of the few real-world studies of AVA, which used data abstracted from patient charts to assess PCs of patients with ITP who switched from ELT or ROMI to AVA, 93% of patients who switched to AVA achieved PC ⩾50 × 109/l and 86% achieved PC ⩾100 × 109/l. In addition, 57% of patients were able to discontinue the use of concomitant ITP treatments after switching to AVA. 21 The present real-world evidence study demonstrated >80% of patients with available data were able to achieve a clinically relevant PC, although longitudinal laboratory data were sporadic. AVA, in this initial study, appears to be effective in real-world use in the United States; however, further studies are needed to better characterize its use and effectiveness.
Several limitations should be considered when interpreting findings from this study. Given less than 3 years of market availability of AVA, the sample size was limited; this limited sample size influenced some study design decisions. For example, while the 6-month baseline period provides an incomplete view of the patient journey, requiring additional preindex claims data would decrease the sample size. Similarly, only one ITP diagnosis preindex and 1 month of pharmacy coverage postindex were required for inclusion due to sample size considerations. The linked lab data do not provide a comprehensive view of all patients’ PCs, as the results of labs drawn at other clinics or facilities were not available. In addition, in the real world (as opposed to a clinical trial setting), PCs may not be checked at regular intervals. Both of these limitations can cause an underestimation of the treatment response. The effectiveness results are assessed in a small subset of AVA patients, as not all patients had linked or available PC information in the relevant time period. In addition, the lack of complete accessibility to PCs precluded our ability to evaluate time to response and durability of response. Finally, some limitations are due to the nature of claims data. For example, sample selection and treatment pattern analyses rely on pharmacy claims; however, pharmacy claims for a filled prescription do not guarantee the actual use of the medication by the patient. Claims data also do not include information on reasons for treatment switch, so whether patients switched to alternative ITP treatments for efficacy, cost, or other reasons could not be assessed. For the purposes of this study, concomitant steroid or immunosuppressant at index was defined as having a steroid or immunosuppressant claim during the 1-month window up to and including the index date; however, it is possible that some of these patients were no longer using the steroid or immunosuppressant therapy at index, which may bias our estimates of steroid or immunosuppressant discontinuation. Finally, changes in steroid dose, which could be an indicator for treatment effectiveness, could not be assessed due to data limitations.
Conclusion
This is the first real-world analysis of patients with ITP initiating AVA utilizing data from an administrative healthcare claims database linked with laboratory data. Patients were diverse in terms of age, comorbidity burden, and use of previous ITP treatments, with approximately half having used another TPO-RA in the previous 6 months. More than 80% of patients with available PC data in this real-world study achieved clinically meaningful PCs following AVA initiation. A large share of patients with concomitant steroid or immunosuppressant use was able to discontinue these treatments following AVA initiation. Furthermore, rescue medications were sparingly used among the study population. While this study was limited by the availability of PC data, the results demonstrated that AVA may be effective in real-world use that is consistent with results from the pivotal clinical trials of AVA.
