Abstract
Background:
Although many acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS) patients have been treated with hypomethylating agents (HMAs) as a substitute for intensive chemotherapy in recent years, the incidence of invasive fungal infections (IFIs) and the efficacy of posaconazole as antifungal prophylaxis in these patients are not well known to date.
Methods:
We retrospectively analyzed 280 AML and MDS patients treated with HMAs to identify IFI incidence and posaconazole efficacy as antifungal prophylaxis in these patients.
Results:
The overall incidence of probable or proven IFIs was 7.9% (22/280 patients): 11.5% in the no-use group (17/148 patients) and 3.8% in the posaconazole group (5/132 patients). Most IFIs occurred during the early cycles of the HMAs (median: 3 cycles; range: 1–8 cycles), especially in patients who had neutropenia or did not respond to HMAs. Posaconazole significantly lowered IFI incidence compared with that in the no-use group in univariate and multivariate analyses. Moreover, patients who had reduced liver function at HMA initiation, were treated with decitabine therapy, and did not respond to HMA chemotherapy were independently associated with a higher IFI risk. In subgroup analysis, posaconazole appeared to be more beneficial for patients with good Eastern Cooperative Oncology Group performance score or liver function at HMA initiation.
Conclusion:
Thus, in AML and MDS patients receiving HMAs, IFI risk may be high during the early cycles, especially when the underlying disease is not controlled. Posaconazole could represent antifungal prophylaxis in these patients; further studies are needed for its appropriate indications.
Introduction
Invasive fungal infection (IFI) is a major cause of non-relapse mortality during the treatment of hematologic malignancy. The incidence of IFIs has been reported to be approximately 5–20% in acute myeloid leukemia (AML)1–5 and 2–10% in myelodysplastic syndrome (MDS),6–9 but the mortality rate has been reported to be as high as 70%.6,10–13 For this reason, the current guidelines recommend the use of antifungal prophylaxis during the treatment of patients with AML or MDS, especially in high-risk patients undergoing intensive chemotherapy or in cases of anticipated prolonged and profound neutropenia.14–17
In recent years, an increasing number of AML and MDS patients were treated with hypomethylating agents (HMAs) as a substitute for best supportive care or even conventional intensive chemotherapy. 18 Although the incidence of prolonged severe neutropenia is not as high as that of intensive chemotherapy, HMAs may cause cytopenia, including neutropenia, and increase transfusion demand.19,20 In addition, considering that AML and MDS themselves are risk factors for febrile neutropenia, 15 the use of antimicrobial prophylaxis during HMA treatment needs to be discussed. However, unfortunately, there is a lack of information on the risk of infection, including IFI, and the use of antifungal prophylaxis in patients treated with HMAs for AML and MDS.
Posaconazole is an oral azole with a wide spectrum, including species of
Methods
Study design and patients
This was a non-interventional comparative cohort study, in which we retrospectively analyzed the data of patients who were consecutively enrolled in the AML and MDS Registry from January 2006 to April 2020. Three affiliated tertiary hospitals (Anam, Guro, and Ansan hospitals) located in the metropolitan area participated in the registry. The study was approved by the institutional review board, and all data were fully anonymized (IRB No. 2020AN0211, 2020GR0237, and 2020AS0137).
Patients who met the following inclusion criteria were selected: (1) patients with a diagnosis of AML or MDS according to the World Health Organization classification; 23 (2) patients first exposed to HMAs (azacytidine or decitabine) as first-line chemotherapy; and (3) patients who received at least one full cycle of either drug. Azacytidine and decitabine were administered at the recommended dose of 75 mg/m2 subcutaneously or intravenously (IV) daily for 7 days every 4 weeks and 20 mg/m2 IV daily for 5 days every 4 weeks, respectively. The use of posaconazole was determined at the discretion of the participating physicians according to the patients’ insurance coverage and the risk of IFI as judged by a physician, and posaconazole was administered at the recommended dose of 300 mg once a day (maintenance dose) after 300 mg twice a day (loading dose) from chemotherapy day 1 until neutropenia recovery.
Clinical endpoints
The primary endpoints were the incidence of IFIs in the AML and MDS patients treated with the HMAs and the efficacy of posaconazole as antifungal prophylaxis. We counted probable or proven IFIs that occurred during treatment with the HMAs (from the time of HMA initiation to before second-line chemotherapy or allogeneic hematopoietic stem cell transplantation), and each IFI was defined according to the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group criteria. 24 In brief, a proven IFI was defined as a case that satisfied any one of the following criteria: (1) identification of fungal elements (molds or yeasts) through microscopic analysis with sterile material; (2) the detection of fungal elements (molds or yeasts) through culture with sterile material or blood; and (3) identification of fungal elements (applies only to yeasts) through serological analysis of cerebrospinal fluid. A probable IFI was defined as a case that satisfied all three of the following conditions: the presence of a host factor, a clinical criterion, and a mycological criterion. Details are presented in Supplemental Material Table 1 online.
The secondary endpoint was the characteristics of patients who would benefit from posaconazole use. The following data were collected: age, diagnosis of AML or MDS, cytogenetics–molecular risk stratification of AML classified according to the 2017 European LeukemiaNet criteria, 25 prognosis risk groups of MDS divided according to the revised international prognostic scoring system, 26 Eastern Cooperative Oncology Group performance score (ECOG PS), 27 absolute neutrophil count (ANC), Child–Pugh score (CPS), 28 chronic kidney disease (CKD) stage 29 based on estimated glomerular filtration rate (eGFR) using the Chronic Kidney Disease Epidemiology Collaboration creatinine equation at the initiation of HMAs,30,31 type of HMAs, use of antibacterial prophylaxis, and treatment responses to HMAs. Responses to the HMAs were evaluated according to the International Working Group.32–34 Patients who responded to treatment were defined as achieving a complete response (CR) or partial response (PR) in AML and CR or PR or hematologic improvement in MDS.
Statistical analysis
Baseline characteristics were compared between the groups using the Mann–Whitney
Results
Patient characteristics
In total, 280 AML or MDS patients treated with HMAs were analyzed in this study (no-use group:
Baseline characteristics at the initiation of hypomethylating agents.

Bold indicates statistical significance.
Cytogenetics-molecular risk stratification of acute myelogenous leukemia was classified according to the 2017 European LeukemiaNet criteria.
Prognosis risk groups of myelodysplastic syndrome were divided according to the revised international prognostic scoring system.
Estimated glomerular filtration rate was calculated using the Chronic Kidney Disease Epidemiology Collaboration creatinine equation.
The analysis was conducted except for the missing value of 7.9% (total 22/280 patients, no use:
CKD, chronic kidney disease; ECOG PS, Eastern Cooperative Oncology Group performance score; eGFR, estimated glomerular filtration rate.
IFIs
The median number of HMA chemotherapy cycles was five cycles (range: 1–72 cycles) in the no-use group and four cycles (range: 1–28 cycles) in the posaconazole group. In total, 22 out of 280 patients were identified with probable or proven IFIs (overall incidence of IFIs: 7.9%) and the details of each IFI are summarized in Tables 2 and 3. The overall incidence of probable or proven IFIs was 11.5% in the no-use group (17/148 patients) and 3.8% in the posaconazole group (5/132 patients); of these, the incidence of invasive mold infections was 8.1% in the no-use group (12/148 patients) and 3.8% in the posaconazole group (5/132 patients). The patients with probable or proven IFIs showed a median age of 69.5 years (range: 21–82 years) and 27.3% of the patients had AML. The median values of the HMA chemotherapy cycle and ANC at the time of IFI diagnosis were 3 (range: 1–6 cycles) and 100/μL (range: 0–2700/μL), respectively. The changes in the ANCs of each patient at the time of diagnosis, the start of HMA treatment, the time of IFI diagnosis, and over the 14 days before IFI diagnosis are summarized in Supplemental Figure 1, as is febrile neutropenia. At the time of IFI diagnosis, the proportion of patients who did not respond to HMAs and the incidence of IFI-related death were 72.7% and 50%, respectively.
Cases of IFIs during treatment with HMAs.

Febrile neutropenia was defined according to the National Comprehensive Cancer Network guidelines (prevention and treatment of cancer-related infections, version 2.2020). Microbiologic criteria for probable infection with
AML, acute myeloid leukemia; ANC, absolute neutrophil count; CR, complete remission; EORTC/MSG, European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group; HI, hematological improvement; HMA, hypomethylating agent; IC, immature cell in peripheral blood; IFI, invasive fungal infection; MDS, myelodysplastic syndrome; NA, not applicable; PD, progressive disease; PR, partial remission; SD, stable disease; WHO, World Health Organization.
Patient characteristics at the time of IFI diagnosis.
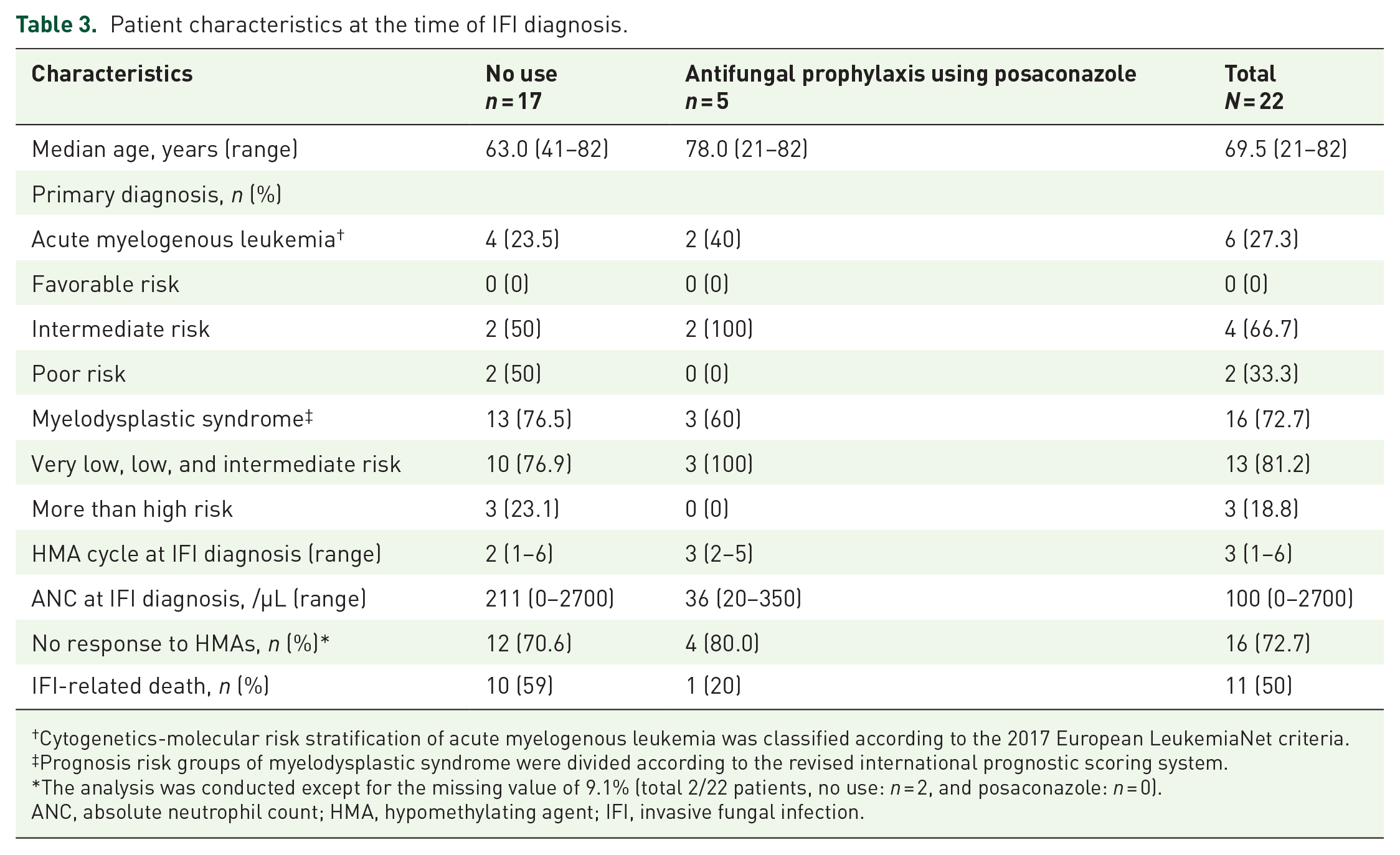
Cytogenetics-molecular risk stratification of acute myelogenous leukemia was classified according to the 2017 European LeukemiaNet criteria.
Prognosis risk groups of myelodysplastic syndrome were divided according to the revised international prognostic scoring system.
The analysis was conducted except for the missing value of 9.1% (total 2/22 patients, no use:
ANC, absolute neutrophil count; HMA, hypomethylating agent; IFI, invasive fungal infection.
In the univariate analysis, the posaconazole group showed a significantly lower incidence of IFIs than the no-use group [no use
Univariate and multivariate analyses of the incidence of invasive fungal infections.

These characteristics were measured at the initiation of hypomethylating agents.
eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration creatinine equation.
ANC, absolute neutrophil count; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance score; eGFR, estimated glomerular filtration rate; OR, odds ratio.
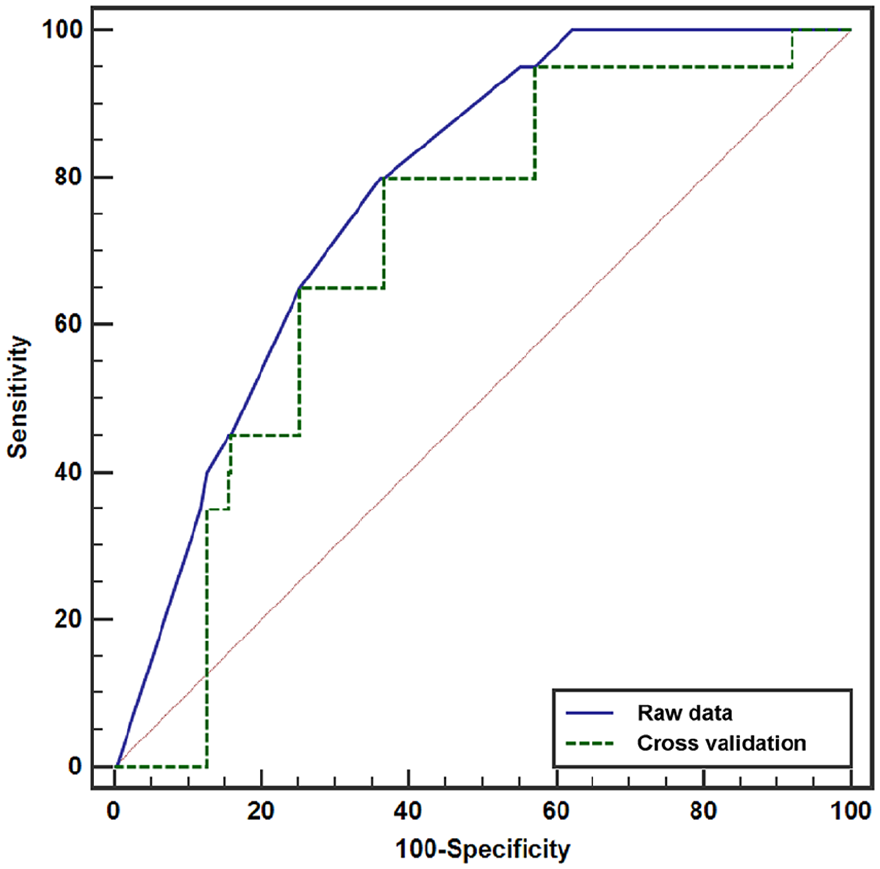
Receiver operating characteristic curve for the raw data and cross validation based on the logistic regression model.
Subgroup analysis for the incidence of IFIs according to the use of posaconazole as antifungal prophylaxis
To further investigate which subgroups would benefit from the use of posaconazole as antifungal prophylaxis, subgroup analysis of the following factors was performed: age, diagnosis, characteristics at HMA initiation (ECOG PS, ANC, CPS, and eGFR), type of HMA, use of antibacterial prophylaxis, and treatment responses to HMAs (Figure 2). In the subgroup analysis, the use of posaconazole significantly decreased the incidence of IFIs in the groups with 0–1 ECOG PS and in the groups with 5–6 CPS (OR: 0.230, 95% CI: 0.061–0.866,

Subgroup analysis of IFI incidence according to posaconazole use as antifungal prophylaxis.
In addition, we further analyzed whether there was medical history that could increase the risk of IFI in patients with AML and MDS. In the univariate logistic regression analyses, there were no histories that raised the risk of IFI, including hypertension (yes: 108
Adverse events leading to the discontinuation of posaconazole
There were no reports of grade 3 or higher adverse events requiring the interruption or discontinuation of posaconazole treatment.
Discussion
In this study, the overall incidence of probable or proven IFIs in AML and MDS patients treated with HMAs as first-line chemotherapy was 7.9%: 11.5% in the no-use group and 3.8% in the posaconazole group. Among the IFIs, the overall incidence of invasive mold infection was 6.1%: 8.1% in the no-use group and 3.8% in the posaconazole group. Although the posaconazole group had a higher median age and proportion of patients with AML diagnosis and lower liver or kidney function at HMA initiation than the no-use group, the use of posaconazole as antifungal prophylaxis during HMA treatment significantly reduced the incidence of IFIs. In the subgroup analysis, this advantage of posaconazole appeared to be greater in patients with good ECOG PS or liver function at HMA initiation. Moreover, this study suggested other factors associated with a higher risk of IFIs, including reduced liver function at HMA initiation, the use of decitabine, and a poor response to HMAs.
Previous studies on the incidence of IFIs in patients treated with HMAs are summarized in Table 5.6,35–37 In these studies, the incidence of IFIs ranged from 3.3% to 12.5%, which is consistent with the incidence range obtained in the present study (overall incidence: 7.9%). As most previous studies did not provide information on the use of antifungal prophylaxis or used antifungal prophylaxis in less than 30% of patients, it is difficult to directly compare the efficacy of posaconazole as antifungal prophylaxis. However, considering that the median value of IFI incidence in the previous studies was 8.8% (range: 3.3–12.5%), the 3.8% IFI incidence in the posaconazole group in the present study was considered relatively lower than that in the previous studies. In addition, in the present study, the overall incidence of invasive mold infections in the no-use group was 8.1% (12/148 patients), which exceeds the 8% threshold of the incidence of invasive mold infections set by the European Conference on Infections for the recommendation of antifungal prophylaxis. 17 Collectively, the results of the present study suggest that the use of antifungal prophylaxis in AML or MDS patients treated with HMAs is reasonable, and one of the options could be posaconazole.
Previous studies on the incidence of IFIs in patients treated with HMAs.

AML, acute myeloid leukemia; ANC, absolute neutrophil count; AZA, azacytidine; DAC, decitabine; HMA, hypomethylating agent; IFI, invasive fungal infection; IPSS-R, revised International Prognostic Scoring System; MDS, myelodysplastic syndrome; PLT, platelet; UK, unknown.
In this study, the characteristics at the time of IFI diagnosis were further summarized to identify patients with a risk of IFIs (Table 3). First, the proportion of AML patients was 27.3% (no-use group: 23.5%, posaconazole group: 40%), suggesting that the risk of developing IFIs could be considered even in MDS patients treated with HMAs. Second, most cases of IFIs in the present study occurred during the early cycles of HMA treatment (median: 3 cycles; range: 1–6 cycles), consistent with previously published results (Table 5). Third, in most cases, ANC was less than 1000/μL at the time of IFI diagnosis. In half of the cases with ANC more than 1000/μL, immature cells were observed in peripheral blood. Neutropenia or immature cells could indicate that the underlying disease was not controlled at the time of IFI diagnosis. In addition, the response to HMA treatment was independently associated with a risk of IFIs in the present study (Table 4). Based on these results, during the early cycles of HMAs, especially until the confirmation of the improvement of cytopenia or response to treatment, the risk of IFIs may be high and the possibility of IFIs among causes of infections should be considered even in MDS patients treated with HMAs.
Posaconazole use as antifungal prophylaxis should be limited to well-accepted indications to avoid excessive cost, toxicity, and antimicrobial resistance. 38 To find the patient groups that benefit more from posaconazole, we conducted a subgroup analysis. In the present study, posaconazole as antifungal prophylaxis significantly decreased the incidence of IFIs in the groups with 0–1 ECOG PS and groups with 5–6 CPS at the initiation of HMAs. As posaconazole is an oral medication partly metabolized in the liver, it may lead to hepatotoxicity.39,40 Thus, posaconazole might not be beneficial for patients with poor compliance or reduced liver function. However, there was no definitive evidence supporting these hypotheses. Rather, considering that the risk of invasive infection is generally high when the performance status is poor41–43 and that delayed diagnosis and treatment are commonly caused by insufficient clinical symptoms in patients with reduced liver function,44,45 antifungal prophylaxis alone may not lower the risk of IFIs in patients with poor ECOG PS or reduced liver function at HMA initiation. The finding that reduced liver function at HMA initiation was an independent risk factor for IFIs in the present study may also support this hypothesis. However, the results of the present study alone are not enough to identify a subgroup that will obtain greater benefit from posaconazole; therefore, further studies are warranted.
The present study was conducted retrospectively with a small number of patients. Thus, the results of this study alone are insufficient to conclude whether the use of posaconazole as antifungal prophylaxis is recommended in AML or MDS patients treated with HMAs or to suggest a subgroup that will obtain greater benefit from posaconazole. In addition, as posaconazole was not compared with fluconazole or itraconazole, which can be generally used as a prophylactic antifungal agent, it is necessary to verify the cost-effectiveness of posaconazole compared with other antifungal agents. Nevertheless, this study provided meaningful insights into the incidence and characteristics of IFIs as well as the efficacy of posaconazole as antifungal prophylaxis in AML or MDS patients treated with HMAs.
Conclusion
In conclusion, in AML or MDS patients receiving treatment with HMAs, the risk of IFIs may be high during the early cycles of HMA treatment, especially when the underlying disease is not controlled. Posaconazole could be an option for antifungal prophylaxis in these patients, and further studies are needed to determine its appropriate indications.
Supplemental Material
10_Supplementary_figure_1_20200909 – Supplemental material for Efficacy of posaconazole prophylaxis in acute myeloid leukemia and myelodysplastic syndrome patients treated with hypomethylating agents
Supplemental material, 10_Supplementary_figure_1_20200909 for Efficacy of posaconazole prophylaxis in acute myeloid leukemia and myelodysplastic syndrome patients treated with hypomethylating agents by Ka-Won Kang, Byung-Hyun Lee, Min Ji Jeon, Eun Sang Yu, Dae Sik Kim, Se Ryeon Lee, Hwa Jung Sung, Chul Won Choi, Yong Park and Byung Soo Kim in Therapeutic Advances in Hematology
Supplemental Material
9_Supplementary_table_1_20200909 – Supplemental material for Efficacy of posaconazole prophylaxis in acute myeloid leukemia and myelodysplastic syndrome patients treated with hypomethylating agents
Supplemental material, 9_Supplementary_table_1_20200909 for Efficacy of posaconazole prophylaxis in acute myeloid leukemia and myelodysplastic syndrome patients treated with hypomethylating agents by Ka-Won Kang, Byung-Hyun Lee, Min Ji Jeon, Eun Sang Yu, Dae Sik Kim, Se Ryeon Lee, Hwa Jung Sung, Chul Won Choi, Yong Park and Byung Soo Kim in Therapeutic Advances in Hematology
Footnotes
Author contributions
BSK and KWK designed the study. KWK analyzed the registry, summarized the results, and wrote the manuscript. KWK, BHL, MJJ, ESY, DSK, SRL, HJS, CWC, YP, and BSK. performed patient management and maintained the registry. All authors approved the final version of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Ethics approval
The study was approved by the institutional review board, and all data were fully anonymized (IRB No. 2020AN0211, 2020GR0237, and 2020AS0137).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from the Korea Health Technology R&D Project from the Korea Health Industry Development Institute, funded by the Ministry of Health & Welfare, South Korea (grant number: HI17C2072).
Informed consent statement
Informed consent was waived by the institutional review board because of the retrospective nature of the study and because the analysis used anonymous clinical data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
