Abstract
Background:
The pivotal trial on venetoclax and hypomethylating agents in unfit elderly acute myeloid leukaemia (AML) has got FDA approval. However, Asian patients were under-represented and showed no survival advantage.
Objective:
We aimed to compare overall survival and healthcare resource utilisation in elderly patients newly diagnosed with AML, receiving venetoclax with hypomethylating agents versus hypomethylating agents alone.
Design:
Target trial emulation.
Methods:
Propensity score matching balanced the baseline characteristics. Kaplan-Meier curve and Cox regression compared overall survival. Negative binomial regression assessed healthcare resource utilisation with time offset.
Results:
Venetoclax plus hypomethylating agents treatment conferred superior overall survival in patients with AML ⩾60 years old and significantly reduced transfusion requirement compared to those receiving hypomethylating agents alone. The difference was particularly prominent among patients ⩾75 years old.
Conclusion:
Compared to hypomethylating agent alone, venetoclax plus hypomethylating agent benefits elderly patients with AML on overall survival and healthcare resource utilisation.

Introduction
Acute myeloid leukaemia (AML) is a group of heterogenous diseases with distinct clinicopathologic, cytogenetic and genetic features, sharing in common an abnormal increase in blasts in blood and bone marrow. 1 Intensive chemotherapy and allogeneic haematopoietic stem cell transplantation are the mainstays of treatment. Elderly and unfit patients with AML who are unfit for these treatments used to have a dismal outcome. Hypomethylating agents (HMAs), including azacitidine or decitabine, inhibit DNA methyltransferases and result in DNA demethylation, 2 whereas the BCL-2 inhibitor venetoclax (VEN) induces apoptosis by blocking anti-apoptotic BCL-2. 3 A pivotal randomised control trial has demonstrated survival advantage conferred by the combination of VEN and azacitidine over azacitidine alone. 3 However, the randomised control trial recruited less than 10% of patients in Asia, and it is unclear if such survival benefits are applicable to Asian patients. We emulated the target trial 3 using territory-wide electronic health records in Hong Kong, which provides medical service to over 90% of the population and evaluated if the combination has benefited these patients over HMA alone in the real world. Such information might ascertain the benefits of combination therapy in developing and resource restricted Asian regions.
Methods
Data source
This is a multi-centre study. We retrospectively retrieved clinical data from the Clinical Data Analysis and Reporting System (CDARS), a territory-wide electronic health record database covering more than seven million eligible residents in Hong Kong4,5 under the care of the Hospital Authority (HA). CDARS includes all public hospital and ambulatory clinic records, providing real-time demographics, diagnoses, prescriptions, laboratory results and mortality data for research and auditing. 6 Disease diagnoses were encoded by the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM), and their accurate coding and entry are considered mandatory in all HA hospitals. Patient information was anonymised by unique patient identifiers.
Study design and eligibility criteria
The study was a target trial emulation.3,5,7 The target population was elderly patients who were newly diagnosed with AML (ICD-9-CM code: 205.0, Table S1) between 2018 and 2023. Inclusion criteria included patients ⩾60 years old who were first diagnosed with AML and have received HMAs, singly or in combination with venetoclax during the recruitment period. Exclusion criteria were adopted from the pivotal trial (ClinicalTrials.gov identifier: NCT02993523): (1) previous treatment with target therapy or chemotherapy for myelodysplastic syndrome (MDS); (2) previous diagnosis of acute promyelocytic leukaemia, malignant neoplasm of lymphatic and haematopoietic tissue or HIV; or (3) patients with any malignancies 2 years before the index date, except basal or squamous cell carcinoma of the skin, or carcinoma in situ of the cervix or the breast (Table S2). Two treatment arms were combination (VEN + HMA) and monotherapy (HMA). Specifications of the target and emulated trials were shown in Table S2. The index date was defined as the first record of prescription of VEN or HMA or the diagnosis date of AML, whichever earlier. Patients would have to receive the specified treatment within 30 days after the index date, and another 30 days were allowed to define the combination therapy (Figure 1).

Inclusion of subjects leading to the final analyses.
A 1:1 propensity score matching, including age, sex, Charlson Comorbidity Index and laboratory tests within 30 days before the index date, was conducted with a caliper of ⩽0.2 to balance the baseline characteristics of patients who received combination or monotherapy with standardised mean difference ⩽0.1. 8 Intention-to-treat (ITT) and per-protocol analysis (PPA) were performed to account for potential non-adherence to the assigned treatments. In ITT, the follow-up ended at death or 30th June 2024 (study end date), whichever earlier. In PPA, the follow-up ended at the time of treatment alteration, discontinuation ⩾90 days, death or study end date, whichever earlier.
Sample size
Sample size was calculated based on the effect size reported in the target trial (hazard ratio (HR) = 0.66, 1 − β = 0.80, α = 0.05). The minimum number of patients required to differentiate overall survival (OS) between the combination and monotherapy arms was calculated using the Sample Size Calculator 9 and was estimated to be 142 for each group. The formula used in the calculator was shown in the Supplementary Method. 10
Outcomes
The primary outcome was OS. Secondary outcomes were healthcare resource utilisation (HRU), including annualised number of transfusions, length of stay in hospital and frequency of hospitalisation, unplanned hospital admission and emergency admission. We also analysed the occurrence of sepsis and 30-day antibiotics usage (Tables S1 and S3) following treatments.
Statistical analyses
Multiple imputation by chained equations was used for imputing variables with missing percentage less than 10%. OS was illustrated by Kaplan–Meier curves and compared using Cox regression. When proportional hazard assumption was violated, segmented Cox regression was used as the alternative. Negative binomial regression was used for between-group HRU comparison. Subgroup analysis by age (60–74 years, and ⩾75 years) was conducted. p Value less than 0.05 was considered as statistically significant. The STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) reporting guidelines 11 were consulted for the transparent reporting of the cohort study (Supplemental Material).
Results
Patient characteristics
We identified 1958 patients with newly diagnosed AML during the recruitment period. Based on the inclusion and exclusion criteria, 186 and 193 patients (mean age: 74 years; male: 61%) received combination or monotherapy (Table 1). There was a significant trend towards increased prescription of combination and decreased prescription of monotherapy from 2018 to 2023 (Figure 2). Patients receiving combination had more medical conditions, resulting in greater Charlson Comorbidity Index than the monotherapy arm (mean ± standard deviation: 1.14 ± 0.96 vs 1.10 ± 1.10). After propensity score matching, 159 patients were assigned to each treatment arm with all standardised mean differences ⩽0.1 (Figure 1 and Table 1).
Baseline characteristics of the patients with acute myeloid leukaemia – before and after propensity score matching.
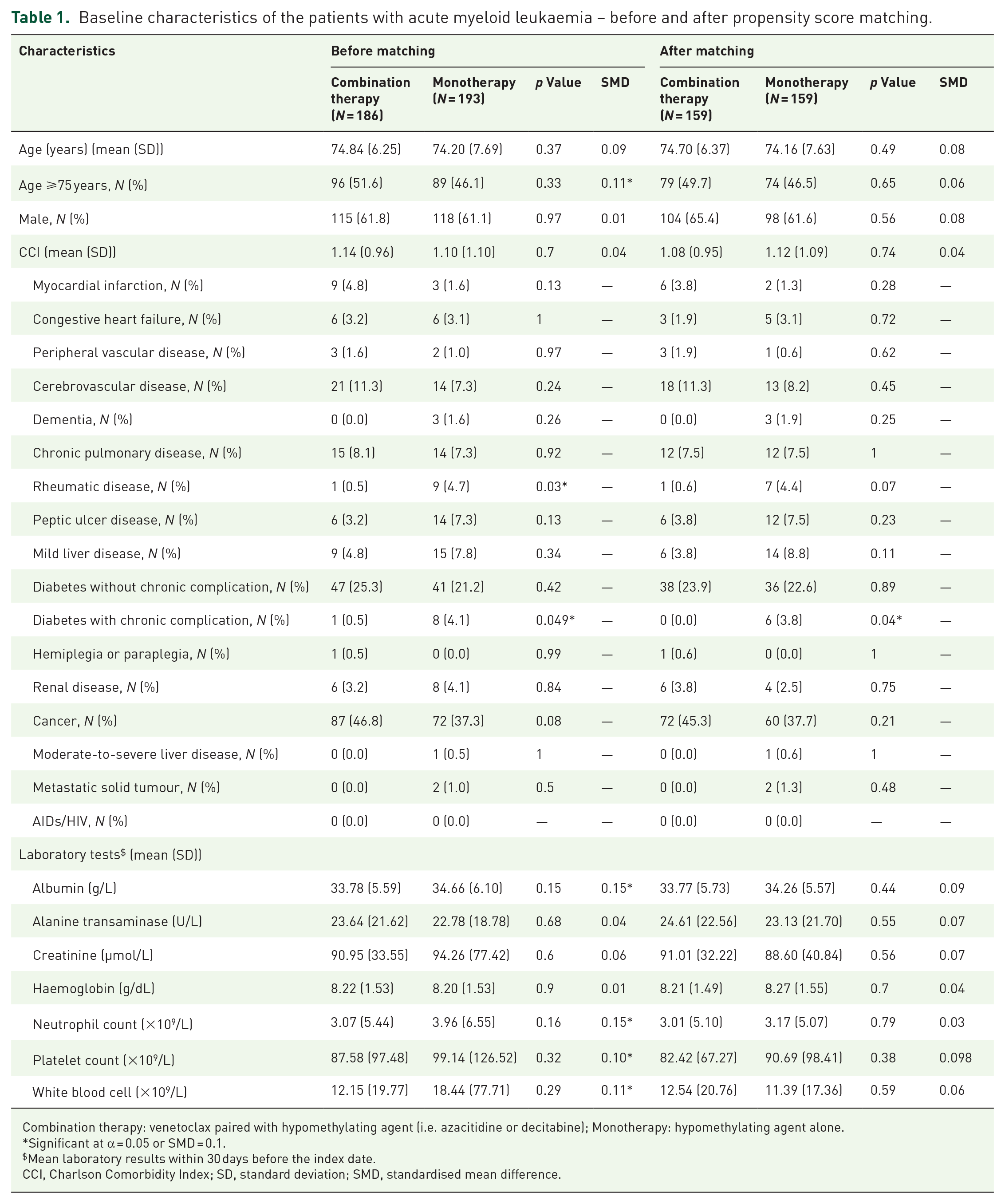
Combination therapy: venetoclax paired with hypomethylating agent (i.e. azacitidine or decitabine); Monotherapy: hypomethylating agent alone.
Significant at α = 0.05 or SMD = 0.1.
Mean laboratory results within 30 days before the index date.
CCI, Charlson Comorbidity Index; SD, standard deviation; SMD, standardised mean difference.

Prescription practice of haematologists territory-wide from 2018 to 2023.
Overall survival
In the ITT analysis, the median durations of follow-up in the combination and monotherapy arms were 11.40 months (interquartile range (IQR) = 13.22 months) and 12.50 months (IQR = 14.60 months). Patients receiving combination showed superior OS compared to those who received monotherapy (HR = 0.72, 95% confidence interval (CI) (0.57, 0.93), p = 0.01; Figure 3(a)), with median OS of 13.16 months (95% CI (11.86, 16.97)) and 12.63 months (95% CI (11.13, 13.90)), respectively. In both arms, there were 13 out of 159 early deaths, defined as death within 60 days after the first targeted prescription. In the PPA, the median durations of follow-up were similar between patients receiving combination and those who received monotherapy (8.13 (IQR = 9.02) months vs 7.30 (IQR = 9.67) months; Figure S1(A)). The HR in Cox regression was 0.80 (95% CI (0.56, 1.14), p = 0.22; Figure S1(A)) and that of segmented Cox regression were 0.49 after 10 months (95% CI (0.24, 1.00), p = 0.049; Table S4). Among patients ⩾75 years (n = 75 each arm), combination conferred superior OS than those receiving monotherapy based on ITT analysis (HR = 0.68, 95% CI (0.47, 0.98), p = 0.04; Figure 3(b)) but not PPA (Figure S1B). Among patients aged 60–74 years (n = 69 each arm), there was no significant difference in OS between combination and monotherapy based on ITT (Figure S2) or PPA analysis (Figure S1(C)).

Overall survival of patients based on intention-to-treat analysis. The number of patients at risk at each time point was shown at the bottom of each panel. The dashed line indicates a 50% overall survival probability, and the tick marks indicate censored data. (a) Entire cohort of patients ⩾60 years old; (b) patients ⩾75 years old.
Healthcare resource utilisation
In the ITT analysis, combination arm (26.98 units per person-year) required significantly less pack cell transfusion compared to the monotherapy arm (35.20 units per person-year, incidence rate ratio (IRR) = 0.70, 95% CI (0.56, 0.87), p = 0.001; Figure 4(a)). Similar results were observed in the PPA analysis (IRR = 0.76, 95% CI (0.60, 0.96), p = 0.02; Figure S3(A)). The rate of transfusion independence, defined as the percentage of patients who had not required blood transfusion within eight consecutive weeks 12 after commencement of the first targeted prescription, showed no significant difference between the two arms (combination: 10.7% (17/159) vs monotherapy: 9.4% (15/159), p = 0.85). In the PPA, a shorter average length of stay was observed in the combination arm (6.19 days vs 7.92 days per person-episode, IRR = 0.79, 95% CI (0.63, 0.99), p = 0.03; Figure S3(A)). There was no significant difference in other HRUs based on ITT (Figure 4(a)) or PPA analyses (Figure S3(A)). Risk of sepsis and 30-day antibiotics use were similar between combination and monotherapy arms in both ITT and PPA analyses (Table S5). Among patients ⩾75 years (n = 75 each arm), those receiving combination also required significantly less pack cell transfusion in the ITT analysis (IRR = 0.72, 95% CI (0.53, 0.98), p = 0.04; Figure 4(b)) and shorter length of stay per person-episode in the PPA (IRR = 0.67, 95% CI (0.49, 0.93), p = 0.01; Figure S3(B)). Among patients aged 60–74 years (n = 69 each arm), there was no significant difference in HRU between combination and monotherapy based on ITT (Figure S4) or PPA analysis (Figure S3(C)).

Healthcare resource utilisation in the intention-to-treat analysis. Values in the lines under ‘Combination’ and ‘Monotherapy’ are the absolute mean value per person-year of each healthcare resource utilisation in each arm. Psychiatric ward accounts for a very small proportion of ‘General and psychiatric ward’. As the standardised mean difference was >0.1 after propensity score matching, age and albumin were added to the regression as covariates in the ⩾75 years group. (a) Entire cohort of patients ⩾60 years old; (b) patients ⩾75 years old.
Discussion
We identified nearly 2000 AML patients in Hong Kong, of whom over 90% are ethnic Chinese, and evaluated the clinical effectiveness of VEN/HMA combinations in 159 new diagnosed elderly patients and compared it with a matched population of patients receiving HMA monotherapy. The database was based on objective evaluation of disease diagnosis, prescription records and clinical endpoints in the HA system, which has shown high coding accuracy. 13 HA provides leukaemia service for over 90% 14 of patients in Hong Kong, and the data represented a comprehensive capture of treatment scenarios territory-wide. Data were based on information input by physicians in different hospitals who were independent of the study. 15 To our knowledge, this is the largest comparative study in Asian patients. Previous studies in Asian patients were limited by small sample size16,17 or having only single arm of treatment. 18 Our results showed that VEN/HMA combination significantly prolonged patient survival and reduced blood transfusion requirements, when compared with a matched population receiving HMA alone. The increase in VEN/HMA prescription over HMA monotherapy also reflected the changing haematology practice territory-wide. Specifically, our real-world data (RWD) based on the emulated trial design demonstrated a longer median OS conferred by VEN/HMA. The benefit was seen predominantly in elderly patients ⩾75 years and was more prominent after 1 year in the PPA analysis, reaffirming that this population with hitherto dismal outcomes have achieved a meaningful remission. This is in contrast to the apparent lack of OS benefits among Asian patients in the pivotal randomised control trial, which has enrolled only 10% patients of Chinese and Japanese ethnicity. Furthermore, patients receiving combination required less pack cell transfusion and addition of VEN to HMA was not associated with increase in sepsis, prescription of antibiotics or unexpected hospital stays. These observations reflected better haematologic recovery as more patients in the combination cohort would have achieved morphologic remission. 3 The results also suggested that combination treatment in elderly would alleviate transfusion requirements; an issue of particular relevance to the reduced motivation of blood donation after the COVID-19 epidemics. 19 Finally, segmented Cox-regression analysis showed significant OS benefits of VEN/HMA after about 1 year of diagnosis with evidence of plateauing over nearly 6 years, supporting the proposition that some of these patients might have achieved a very sustainable remission. It should be noted that the difference in median OS between combination and monotherapy (13.16 months vs 12.63 months) was smaller than that reported in the VIALE-A trial 3 (14.7 months vs 9.6 months). Discrepancies in outcomes between randomised control trial and RWD is well recognised 20 and might be due to difference in demographics, unadjusted confounding factors and variation in treatment doses and frequencies in the real-world setting. It should not negate the significance of our findings but rather underscored the importance of post-marketing RWD in ascertaining the effectiveness of novel treatment. In fact, the median OS of patients receiving combination therapy in this study was longer than the 6.0 months reported in another observational study by Bouligny et al. 21
Limitations
Our datasets lacked the granularity required for detailed evaluation of patient characteristics, including their mutation profiles, treatment response and the dose and regimen of HMA and VEN, precluding detailed outcome comparison with those of the pivotal trial. Long-term follow-up of the target trial showed that combination therapy conferred survival benefit to AML carrying mutations of isocitrate dehydrogenase (IDH1/2) but not those with TP53 mutation. 22 Such analyses were beyond the scope of this study. In an attempt to provide a filter for adverse genetic risk associated with secondary AML, patients with prior diagnoses of MDS/MPN (myeloproliferative neoplasms), and those who have received chemotherapy for other malignancies were excluded from the analysis. In addition, the actual drug costs to HA and potential alleviation of healthcare burden were not available, precluding detailed evaluation of the health economy effectiveness. These shortcomings notwithstanding, our data represented one of the largest territory-wide dataset in Asian patients and reaffirmed the clinical benefits of VEN/HMA combination in elderly patients who were newly diagnosed with AML in clinical practice.
Conclusion
Compared to HMA monotherapy, VEN/HMA combination effectively prolonged the OS and reduced the need in the pack cell transfusion for patients with AML, especially in the group ⩾75 years.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251346914 – Supplemental material for Hypomethylating agent versus venetoclax combination: an electronic health records-based target trial emulation among Asian elderly patients with newly diagnosed acute myeloid leukaemia in Hong Kong
Supplemental material, sj-docx-1-tah-10.1177_20406207251346914 for Hypomethylating agent versus venetoclax combination: an electronic health records-based target trial emulation among Asian elderly patients with newly diagnosed acute myeloid leukaemia in Hong Kong by Qiwen Fang, Chi Yeung Fung, Jiaqi Wang, Wing Hei Lai, Raymond S. M. Wong, Bonnie C. S. Kho, June S. M. Lau, Vivien W. M. Mak, Chung Yin Ha, Xue Li and Anskar Yu-Hung Leung in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251346914 – Supplemental material for Hypomethylating agent versus venetoclax combination: an electronic health records-based target trial emulation among Asian elderly patients with newly diagnosed acute myeloid leukaemia in Hong Kong
Supplemental material, sj-docx-2-tah-10.1177_20406207251346914 for Hypomethylating agent versus venetoclax combination: an electronic health records-based target trial emulation among Asian elderly patients with newly diagnosed acute myeloid leukaemia in Hong Kong by Qiwen Fang, Chi Yeung Fung, Jiaqi Wang, Wing Hei Lai, Raymond S. M. Wong, Bonnie C. S. Kho, June S. M. Lau, Vivien W. M. Mak, Chung Yin Ha, Xue Li and Anskar Yu-Hung Leung in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
