Abstract
Background:
Patients with post-transplant cytopenias due to poor graft function or primary engraftment failure show poor prognosis with a high mortality rate mainly because of graft versus host disease (GVHD), infection and/or bleeding. Treatment options are scarce and a CD34+ stem cell boost or a second bone marrow transplantation may be required to restore adequate haematopoiesis.
Methods:
In the present study patients with primary engraftment failure (n = 1) and refractory poor graft function (n = 11) were treated with eltrombopag in a single centre. The reason for eltrombopag treatment was trilineage cytopenia in six patients, bilineage cytopenia in three patients and single lineage cytopenia in three patients. Eltrombopag was initiated at a median of 214 (range: 120–877) days after haematopoietic stem cell transplantation (HCST) and administered for a median time of 114 (range: 12 days to >490) days. In 8/12 patients eltrombopag was introduced at a dose of 75 mg/day and then increased to 150 mg/day after 1 week; 1 patient was given 50 mg eltrombopag per day, and 3 patients received 75 mg daily.
Results:
In 10/12 patients eltrombopag significantly enhanced blood count values and patients became transfusion independent. Once stable haematological response was obtained, treatment was tapered until final discontinuation in 9/10 responding patients. No grade 3 or 4 toxicities were observed. At time of last follow up, 3/12 patients were dead, 2 due to disease relapse, 1 due to GVHD and pneumonia. All patients except one maintained their complete response and remain transfusion independent at a median of 858 (range: 429–1119) days.
Conclusion:
These preliminary data confirm that eltrombopag is able to rescue multilineage haematopoiesis in patients with treatment-refractory cytopenias after allogeneic HSCT.
Introduction
Poor graft function is a multifactorial complication after allogeneic haematopoietic stem cell transplantation (HSCT), occurring in 7–21% of patients.1,2 Multiple pre- and post-transplantation factors have been associated, namely: (a) donor-recipient HLA-disparity; (b) low haematopoietic stem cell (HSC) dose; (c) T-cell depletion of the donor marrow; (d) intensity of the conditioning regimen; (e) post-transplant immunosuppression; (f) coexisting graft versus host disease (GVHD); (g) reactivation of cytomegalovirus (CMV) as well as its treatment with ganciclovir; (h) infections. A small proportion of patients (<3%) fail engraftment after transplant and therefore are classified as primary engraftment failure. 3
Several attempts to reconstitute donor haematopoiesis have included CD34+ selected stem cell boost, mesenchymal stem cell transfusion or a second marrow transplantation.4–6 Unfortunately patients achieve unsatisfactory results and their prognosis remains poor with a high mortality rate due to GVHD, infection and/or bleeding, especially in patients with primary engraftment failure.
Recent in vitro and in vivo data showed that stimulation of the c-MPL-signalling pathway by thrombopoietin (TPO) agonists may have a significant influence on maintenance and self-renewal of HSCs.7–10 Recovery of trilineage haematopoiesis after eltrombopag treatment was highly effective in patients with severe aplastic anaemia (SAA) and led to US Food and Drug Administration approval for treatment of SAA, immune thrombocytopenia and hepatitis-C-related thrombocytopenia.11–15
In the setting of HSCT, several authors investigated retrospectively the role of eltrombopag in refractory thrombocytopenia after allogeneic HSCT and reported promising results with significant platelet recovery until achieving transfusion independence.16–19 On the basis of these results current phase II trials using TPO agonists for prolonged thrombocytopenia after HSCT have been initiated, (NCT01791101, NCT01000051, NCT01927731).
Further, in vivo studies have demonstrated that TPO promotes even multilineage haematopoiesis and increases peripheral human platelets and white blood cell counts by recruiting and expanding HSC/haematopoietic progenitor cells from the bone marrow compartment of a NOD/SCID xenotransplant model.20,21
These observations led to the hypothesis that eltrombopag may have a role in haematopoietic recovery in single, bi- or even trilineage cytopenias in patients after allogeneic HSCT as recently reported.22–24 In the present analysis we report a single centre experience with eltrombopag in post-allogeneic transplant patients with refractory single and multilineage cytopenias due to poor graft function or primary engraftment failure.
Patients and methods
Between January 2015 and August 2018 a total of 216 patients underwent allogeneic HSCT at the Stem Cell Transplant Center, AOU Città della Salute e della Scienza in Turin, Italy. Overall, 12 (5.5%) patients developed refractory single or multilineage cytopenias after transplant due to graft failure (n = 1) or poor graft function (n = 11) and were included in the present analysis. Two patients have already been described in a previous study. 22 Poor graft function was defined as cytopenia in at least one lineage, namely platelets <20.000 × 106/L, haemoglobin <90 g/L, absolute neutrophil count (ANC) <1000 × 106/L and/or transfusion requirement of blood or platelets beyond day +28 after HSCT together with a full donor chimerism. Patients with severe GVHD, reactivation of CMV or other infections as well as patients receiving myelotoxic treatments at time of eltrombopag treatment were excluded. Three patients with full donor chimerism developed single (n = 1) and multilineage cytopenia (n = 2) at days +59, +105 and +131, respectively. They are still receiving eltrombopag after 30, 14 and 15 days, respectively. Interestingly, two of them showed significant improvement in their transfusion requirements. Given the short follow-up period the three patients were not included in the analysis.
Poor graft function was classified as ‘primary’ if association with obvious clinical events could be excluded. 23 Poor graft function was defined as ‘secondary’ in the following circumstances: (a) in case of association with immune-mediated events such as GVHD in the previous 4 weeks, or (b) in case of association with infectious complications and/or myelotoxic treatments such as CMV reactivation and anti-CMV treatment in the previous 4 weeks, or other clinically important infections. In the case of missing recovery of the blood count after HSCT the patient was defined as primary engraftment failure. 23
A bone marrow analysis showing a full donor chimerism and excluding disease relapse as well as showing impaired haematopoiesis was performed in all patients before therapy initiation. Before starting eltrombopag treatment and after individual approval by the Hospital Commission for Off-label Use of Drugs for each patient, personal informed consent for off-label treatment was signed by each patient. All patients received oral eltrombopag: in 8/12 patients eltrombopag was initiated at a dose of 75 mg/day and after 1 week was increased to 150 mg/day; 1 patient was given eltrombopag 50 mg/day, and 3 patients received 75 mg daily. Once stable haematological response was obtained, treatment was slowly tapered until final discontinuation.
Complete response or remission (CR) was defined as all of the following: (a) platelet count >50.000 × 106/L: (b) ANC >1500 × 106/L; (c) haemoglobin >100 g/L without blood cell transfusion or granulocyte colony-stimulating factor (G-CSF) support.
Treatment toxicities
Eltrombopag was well tolerated by all patients with no evidence of cataract, thrombosis or any other grade 3/4 toxicities, only grade 1–2 toxicities were observed. No endogenous TPO levels were measured in this patient cohort. A total of 11 patients started eltrombopag therapy at 75 mg daily in order to assess the initial safety profile. After 1 week, in eight patients, the dosage was increased to 150 mg daily, except in four patients. Patient no. 06 developed a grade 2 thrombocytosis up to 50,700 × 106/L when the dose was increased to 150 mg. Eltrombopag was immediately reduced to 75 mg and the platelet count normalized again. Patient no. 08 developed grade 1 nausea after enhancement to 150 mg and subsequently refused the 150 mg dosage. Patient no. 11 started eltrombopag therapy at 75 mg and obtained a CR with this dosage. Patient no. 12 started eltrombopag at 50 mg daily due to moderate liver enzyme enhancement. After a total of 12 days he developed significant increase in liver function tests, therefore eltrombopag was discontinued. He concomitantly developed severe venous occlusive disease.
Statistical analysis
A nonparametric Wilcoxon matched-pairs signed-rank test was used to determine the statistical significance of the single lineage upon eltrombopag treatment in patients with cytopenia considering the single cytopenia line. All statistical tests were two-tailed. Statistical analyses were performed using GraphPad Prism 8.4.1. for Windows (GraphPad Software, San Diego, CA, USA). Unadjusted p values less than 0.05 were considered significant.
Results
Transplant characteristics
Patients’ baseline transplant characteristics are summarised in Table 1. Indication for allogeneic transplantation was acute leukaemia (including one blast crisis of chronic myeloid leukaemia) in 11 patients and high-risk myelodysplastic syndrome in 1 patient. Median age of recipients and donors was 47.5 (range: 40–66) years and 37 (range: 17–59) years, respectively. The majority of patients (8/12) received grafts from an unrelated donor (8/8 HLA matched in 7 cases), 3 patients from a haploidentical family member and 1 patient from a matched sibling donor. A total of 7/12 patients showed an unfavourable CMV combination (donor negative/patient positive) whereas 5 donor/recipient pairs showed a major AB0 incompatibility. All patients received a T-cell depleted graft: in 5/12 patients the haematopoietic stem cell source was bone marrow while 7/12 patients received peripheral blood stem cells. Conditioning regimens were chosen according to standard guidelines on the basis of the underlying disease, the age of the patient and the donor type and are listed in Table 1. A myeloablative conditioning regimen was used in all cases except one and patients received a median of 3.7 × 108/kg (range: 1.77–13.3) total nucleated cells, a median of 6 × 106/kg (range: 1.8–19.2) CD34+ cells and a median of 2.37 × 108/kg (range: 0.25–3.2) CD3+ cells. GVHD prophylaxis included cyclosporine (CSA) and methotrexate (MTX) in matched sibling donor transplants, CSA-MTX and rabbit antithymocyte globulin in unrelated donor transplants and tacrolimus-mycophenolate mofetil combined with post-transplant cyclophosphamide in case of grafts from haploidentical donors.
Patient and transplant characteristics.
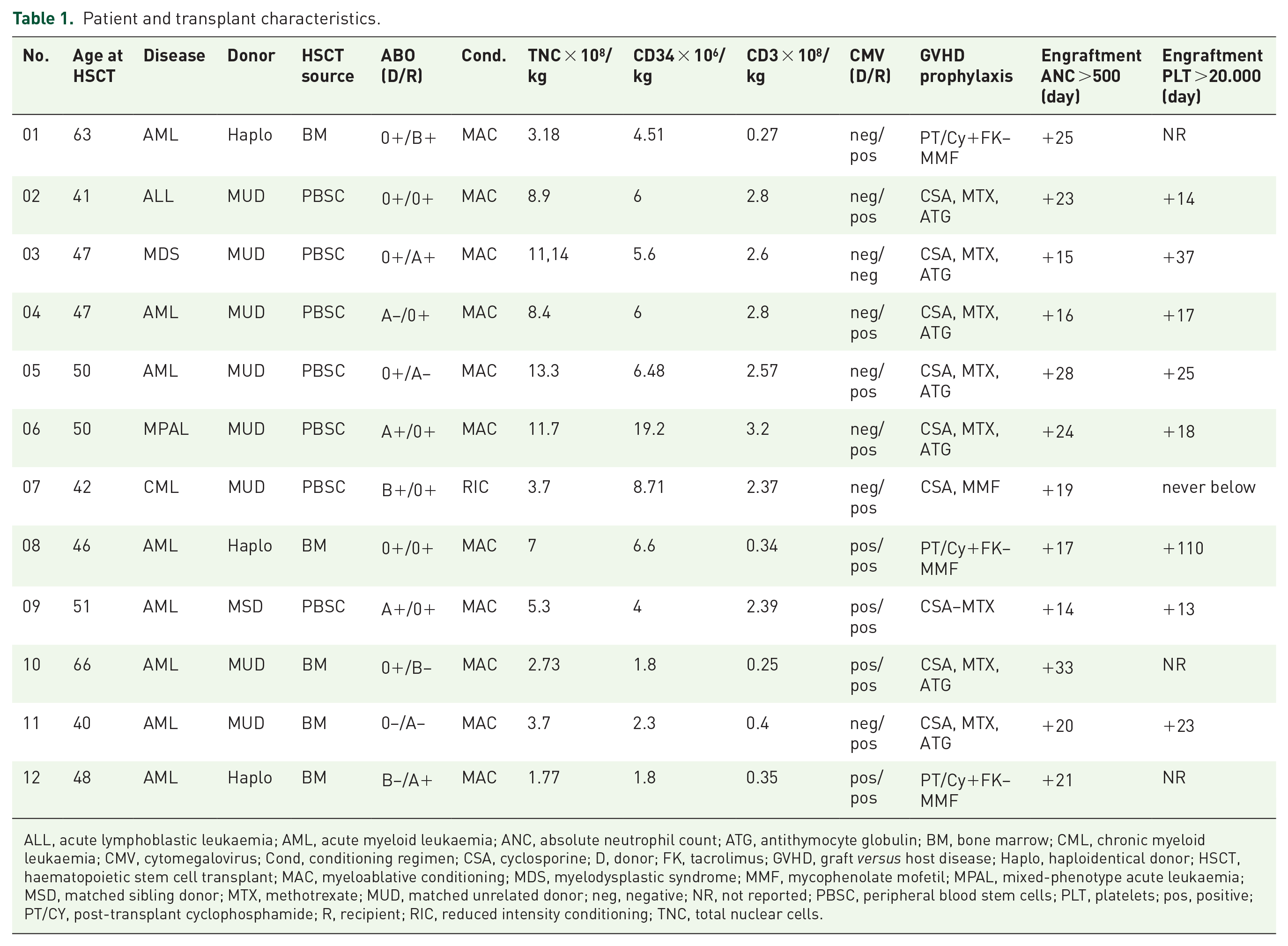
ALL, acute lymphoblastic leukaemia; AML, acute myeloid leukaemia; ANC, absolute neutrophil count; ATG, antithymocyte globulin; BM, bone marrow; CML, chronic myeloid leukaemia; CMV, cytomegalovirus; Cond, conditioning regimen; CSA, cyclosporine; D, donor; FK, tacrolimus; GVHD, graft versus host disease; Haplo, haploidentical donor; HSCT, haematopoietic stem cell transplant; MAC, myeloablative conditioning; MDS, myelodysplastic syndrome; MMF, mycophenolate mofetil; MPAL, mixed-phenotype acute leukaemia; MSD, matched sibling donor; MTX, methotrexate; MUD, matched unrelated donor; neg, negative; NR, not reported; PBSC, peripheral blood stem cells; PLT, platelets; pos, positive; PT/CY, post-transplant cyclophosphamide; R, recipient; RIC, reduced intensity conditioning; TNC, total nuclear cells.
A total of 11 patients achieved neutrophil recovery >500 × 106/L after a median of 20.5 (range: 15–33) days and 8 patients achieved platelets >20.000 × 106/L after a median of 20.5 (range: 13–37) days after transplant.
Cytopenias and treatment characteristics
Cytopenias and treatment characteristics are summarised in Table 2. Six patients developed primary poor graft function while in five patients poor graft function was secondary to GVHD (n = 2), previous bacterial sepsis (n = 2) and CMV reactivation requiring therapy up to 1 month before cytopenia appearance (n = 1). At time of eltrombopag initiation all patients showed no evidence of CMV infection. As part of the routine protocol for patients with refractory cytopenia at our centre, human herpesvirus 6 (HHV6) reactivation was controlled. None of the patients in the present study resulted positive.
Cytopenia and treatment characteristics.

ANC, absolute neutrophil count; BM, bone marrow; BRT, bortezomib; EDX, cyclophosphamide; EPO, erythropoietin; G-CSF, granulocyte colony-stimulating factor; HSCT, haematopoietic stem cell transplant; Ig, immunoglobulin; PEF, primary engraftment failure; PEX, plasma exchange; PGF, poor graft function; PLT, platelets, RBC, red blood cells; RTX, rituximab.
The reason for eltrombopag treatment was trilineage cytopenia in six patients, bilineage cytopenia in three patients and single lineage cytopenia in three patients, of whom n = 1 with isolated anaemia and n = 2 with isolated thrombocytopenia. All patients required recurrent transfusions of platelet and/or red blood cells. Antibody-mediated cytopenia was investigated at time of pure red cell aplasia diagnosis in three patients with anti-A immunoglobulin (Ig)G and IgM titres (no. 4: IgG 1:128, IgM 1:32; no. 6: IgG 1:2048, IgM 1:256; no. 9: IgG 1:32, IgM 1:4). Two patients (no. 6 and no. 9) did not show significant reduction of anti-A titres before eltrombopag initiation, while one (no. 4) did not determine anti-A titres before eltrombopag initiation. At the time of eltrombopag initiation, the median ferritin level was 3126 (range: 897–6770) ng/ml with 9/12 patients having a ferritin level >2000 ng/ml. Before treatment with eltrombopag patients received erythropoietin (n = 8), G-CSF (n = 8), plasma exchange (n = 2), rituximab (n = 4), immunoglobulins (n = 2), bortezomib (n = 1) and cyclophosphamide (n = 1). As indicated in Table 2, four patients received rituximab at days +216, +286, +161 and +209 post-transplant, respectively. Two underwent plasma exchange at day +173 and +125, whereas one patient received bortezomib at day +390 after transplant. Patients receiving rituximab therapy were treated after a total of 17, 257, 716 and 195 days, respectively with eltrombopag and all four responded with CR. Nevertheless a long-term effect of rituximab therapy cannot be excluded. Eltrombopag was initiated at a median of 214 (range: 120–877) days after HCST and was administered for a median time of 114 (range: 12–>490) days. In one case (no. 12) eltrombopag was discontinued after a total of 12 days and not resumed due to the presence of significant increase in liver function tests concomitantly with the development of severe venous occlusive disease. Interestingly, after discontinuation of eltrombopag the platelet count increased up to a maximum of 59,000 × 106/L, the haemoglobin value rose to a maximum of 113 g/L and the absolute neutrophil count up to a maximum of 1740 × 106/L. This improvement was temporary; 12.9 months after eltrombopag treatment the patient developed relapse of his acute myeloid leukaemia and died 3 weeks later.
In 10/12 patients eltrombopag significantly increased blood count values and patients became transfusion independent. Among all, the median platelet count of nine patients with persistent thrombocytopenia before eltrombopag treatment was 16,000 (range: 6000–22,000) × 106/L versus 77,000 (range: 50,000–218,000) × 106/L after eltrombopag treatment, p = 0.012 (Figure 1(a)). This maximum response of the platelet count was obtained a median of 66 (range: 44–425) days after eltrombopag initiation. A total of 10 patients with persistent anaemia showed a significant response with a rise in the haemoglobin value from a median value of 76 (range: 66–85) g/L to a median of 129 (range: 74–154) g/L after eltrombopag therapy; this response was obtained after a median of 141 (range: 6–291) days (p = 0.008) (Figure 1(b)). Finally, a total of eight patients with persistent neutropenia had a median absolute neutrophil count before eltrombopag treatment of 650 (range: 0–990) × 106/L versus 2325 (range: 0–4260) × 106/L after a median of 200 (21–379) days, p = 0.019 (Figure 1(c)).

Blood cell count before and after eltrombopag treatment. The analysis included all patients with cytopenia considering the single lineage. (a) Median platelet count was 16,000 (6000–22,000) versus 77,000 (50,000–218,000) × 106/L before and after treatment (p = 0.012). (b) Median haemoglobin was 76 (66–85) versus 129 (74–154) g/L (p = 0.008). (c) Median neutrophil count was 650 (0–990) versus 2325 (0–4260) × 106/L (p = 0.019). Nonparametric Wilcoxon matched-pairs signed-rank test.
Two patients did not respond to eltrombopag. The poor graft function of the nonresponding patient no. 10 was defined secondary probably due to a previous CMV reactivation and treatment. Eltrombopag treatment was maintained for up to 22 days at a maximum dose of 150 mg daily, however he developed a significant GVHD and eventually died of pneumonia. The second nonresponding patient (no. 7) was classified as a primary engraftment failure, she never recovered haematopoiesis after transplant and was treated with eltrombopag (maximum 150 mg/day) for a total of 30 days. No response was obtained, and she received a second bone marrow transplantation. She is still alive maintaining full donor chimerism and shows no signs of disease relapse.
Overall, at time of last follow up, 9/12 patients are still alive, of whom only 1 patient is still under eltrombopag treatment. In seven patients eltrombopag could be tapered and discontinued. All patients except one maintained their complete response and remain transfusion independent at a median of 858 (range: 429–1119) days. One patient, who initially responded with a CR lost the response 341 days after initiation of the treatment, and eltrombopag was stopped. A total of 3/12 patients were dead at time of last follow up, 2 due to disease relapse and 1 due to GVHD and pneumonia.
Discussion
The receptor for TPO, c-Mpl, is expressed mainly on HSCs. Eltrombopag initiates signal transduction by binding to c-Mpl, consequently Janus kinase 2 phosphorylates tyrosine residues, thereby stimulating the downstream cascade STATs, PI3K, the mitogen-activated protein kinases and extracellular signal-regulated kinases.25–27 Thereby, the c-Mpl/TPO signalling pathway is activated and regulates partially HSC cell-cycle progression and renewal. 28 Sun et al. could previously even demonstrate that eltrombopag induces in umbilical cord blood transplants multilineage responses through the expansion of bone marrow HSCs and haematopoietic progenitor cells. 21
On the basis of these results, the stimulation of residual HSCs by TPO agonists was postulated and attempted by using eltrombopag in the setting of persistent cytopenia after HSCT by several groups with promising results. Tang et al. have treated with eltrombopag a cohort of 12 post-HSCT patients, who responded poorly to standard treatments for secondary poor graft function. A total of 10 (83.3%) patients responded to the treatment; 8 achieved CR while 2 achieved a partial response. 24 The 12-month overall survival rate was 83.3% with all CR patients maintaining their response. A further study with a similar cohort size was reported by Marotta et al. using eltrombopag as first-line treatment in patients with primary engraftment failure and poor graft function (n = 13). 23 Overall, haematological response was reported in 7/13 (53.8%) patients, with 6 patients showing CR (46.2%). In both studies eltrombopag was well tolerated, and no signs of clonal evolution were reported. The dosages used in the study of Marotta et al. as well as in the study of Tang et al. were quite low (usually <100 mg/day) compared with the 150 mg/day used in aplastic anaemia. In our study the initial dose used was 75 mg and then increased to 150 mg daily in the majority of patients according to tolerance and response.
In the present cohort, a total of 10/12 patients (83.3%) obtained CR achieving transfusion independence. These results were obtained by oral eltrombopag monotherapy in patients that were deeply cytopenic that otherwise might require a CD34+-selected stem cell boost or a second allogeneic HSCT to restore an effective haematopoiesis.
Interestingly, analysis of the bone marrow biopsy before eltrombopag treatment in eight patients revealed the presence of infiltration of cytotoxic T lymphocytes (marked by CD3+ and cytotoxic granule-associated RNA binding protein TIA1) in the range of 10–25%. Whether the infiltration of cytotoxic T cells in the bone marrow may have an impact on poor graft function occurrence is currently unknown. Notably, Nakao et al. showed that in patients with aplastic anaemia a T-cell clone markedly inhibited the growth of haematopoietic progenitor cells by direct cell–cell contact and IFN-gamma secretion in response to CD34+cells. 29 They hypothesised that these cytotoxic T cells may even contribute to the pathogenesis of aplastic anaemia. 29
Further, iron overload has been shown to have a suppressive effect on the erythroblast differentiation of human CD34+ cells. It increases the level of the intracellular reactive oxygen species (ROS) promoting apoptosis of immature erythroblasts by suppressing BCL2 gene expression.30,31 Kong et al. recently reported that elevated ROS levels are associated with increased DNA damage and apoptosis, resulting in exhaustion of CD34+ cells in the bone marrow of patients with poor graft function. 32 These studies support the strong association of a high serum ferritin level with poor graft function. Iron-chelation therapy may lead to a significant haematopoietic response in some patients with aplastic anaemia.33,34 Similarly, Olivieri et al. described a patient with severe aplastic anaemia who was diagnosed with poor graft function after transplantation and achieved complete haematopoietic recovery mainly due to iron-chelation therapy. 35
In the present study the median ferritin level before eltrombopag treatment initiation was 3126 (range: 897–6770) ng/ml with 9/12 patients having a ferritin level >2000 ng/ml. This may be attributed to significant blood transfusion support as nearly all were patients with acute leukaemia. Iron chelation was performed in six out of nine patients with severe anaemia, and all except one (no. 10) became blood transfusion independent. On the basis of this preliminary result, a synergistic effect between eltrombopag and iron-chelation therapy may be hypothesised.
At time of last follow up, three patients were dead and one with primary engraftment failure was successfully retransplanted. Of the remaining eight patients seven maintained CR criteria and are transfusion independent. Our study has several limiting factors, namely the retrospective character, the small cohort size and the lack of a control group for the comparison of the drug effect. Defined criteria for eltrombopag initiation and discontinuation were lacking and were determined by the clinical course. A larger prospective study is warranted to confirm our preliminary results.
In conclusion, in the present study, a high overall response rate was achieved with the use of eltrombopag in heavily pretreated patients with post-HSCT cytopenias. These preliminary findings confirm the results from previous groups suggesting that patients with refractory cytopenias following HSCT might benefit from treatment with eltrombopag.
Footnotes
Authors contributions
SA collected data, performed analysis and wrote the manuscript. AB, CD, SM, LG, GI and BB analysed the data and wrote the manuscript. LG performed the bone marrow analysis.
Authors’ note
Semra Aydin is also affiliated with Department of Oncology, Hematology, Immuno-oncology and Rheumatology, University Hospital of Bonn, Bonn, Germany.
Conflict of interest statement
AB has been a speaker for Novartis.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
