Abstract
Individuals with sickle cell disease (SCD) are living further into adulthood in high-resource countries. However, despite increased quantity of life, recurrent, acute painful episodes cause significant morbidity for affected individuals. These SCD-related painful episodes, also referred to as vaso-occlusive crises (VOCs), have multifactorial causes, and they often occur as a result of multicellular aggregation and vascular adherence of red blood cells, neutrophils, and platelets, leading to recurrent and unpredictable occlusion of the microcirculation. In addition to severe pain, long-term complications of vaso-occlusion may include damage to muscle and/or bone, in addition to vital organs such as the liver, spleen, kidneys, and brain. Severe pain associated with VOCs also has a substantial detrimental impact on quality of life for individuals with SCD, and is associated with increased health care utilization, financial hardship, and impairments in education and vocation attainment. Previous treatments have targeted primarily SCD symptom management, or were broad nontargeted therapies, and include oral or parenteral hydration, analgesics (including opioids), nonsteroidal anti-inflammatory agents, and various other types of nonpharmacologic pain management strategies to treat the pain associated with VOC. With increased understanding of the pathophysiology of VOCs, there are several new potential therapies that specifically target the pathologic process of vaso-occlusion. These new therapies may reduce cell adhesion and inflammation, leading to decreased incidence of VOCs and prevention of end-organ damage. In this review, we consider the benefits and limitations of current treatments to reduce the occurrence of VOCs in individuals with SCD and the potential impact of emerging treatments on future disease management.
Introduction
Sickle cell disease (SCD) is an autosomal recessive disorder caused by mutations in the
The hallmark of SCD is pain, necessitating recurrent emergency department (ED) and/or hospital visits.3,4 An acute vaso-occlusive crisis (VOC) is an important driver of this pain, and is a major cause of morbidity and acute care utilization in this population.5–8 However, the unpredictably of painful events, use of different pain definitions, the subjective nature of pain, and challenges differentiating acute and chronic pain highlight the limitations in assigning specific pain events to an underlying cause in SCD. The principal cause of VOCs is microvascular occlusion, leading to increased inflammation and tissue ischemia-reperfusion injury.9,10 Treatments for VOC have focused largely on the symptomatic management of the acute painful episode as opposed to prevention. However, specific therapies are needed to reduce the occurrence of these episodes and decrease the associated tissue and organ damage. Several investigational treatments are being studied that target the pathologic mechanism of the vaso-occlusive process and that have the potential to reduce the frequency and severity of VOC. A detailed description of all agents that target the vaso-occlusive patho-biologic process have been the subject of several recent reviews, including one that examines the pathophysiology and development of new agents. 11 In this review, we specifically discuss the current and emerging therapeutic options for reducing VOC in SCD.
Pathogenesis of acute VOCs: adhesion and inflammatory processes
The pathophysiology of SCD is initiated by the presence of HbS, which polymerizes under conditions of reduced oxygenation, causing deformation and damage to the RBC membrane.3,12 Hemolysis caused by unstable HbS results in release of free hemoglobin, labile iron, and oxidative stress (Figure 1).
13
This, in turn, causes inflammation and activation of neutrophils, platelets, and endothelial cells through promotion of release of placenta growth factor and endothelin I, and the activation of toll-like receptor 4 and NALP inflammasome signaling.
13
Ultimately, this leads to adhesion of RBCs, neutrophils, and platelets to the endothelium, resulting in vaso-occlusion.13,14 Free hemoglobin also directly consumes nitric oxide, leading to endothelial dysfunction.15,16 In addition, hemolysis also causes release of arginase I, which depletes plasma

Contribution of intravascular hemolysis to vasculopathy and vaso-occlusion. Intravascular hemolysis produces free hemoglobin, which drives Fenton reactions to produce oxidants and scavenges NO by a deoxygenation reaction. Intravascular hemolysis also releases red cell arginase 1 into plasma, where it can deplete plasma
Thus, the processes underlying VOC in SCD are multifactorial, with no single mechanism dominating the adhesion interactions. Consequently, if a therapy is targeted to one specific interaction, vaso-occlusion may still occur through multiple other mechanisms, suggesting the need for multimodal therapy to reduce the occurrence of VOC. 20 An understanding of the pro-adhesive role of the endothelium and the inflammation resulting from ongoing hemolysis and recurrent/chronic ischemia-reperfusion injury has led to the development and investigation of newer mechanistic treatment approaches aimed at reducing their occurrence (Figure 2). New therapies are aimed at both inhibiting the adhesion of cells to the activated endothelium and preventing hemolysis and sickling through direct targeting of the hemoglobin within RBCs.
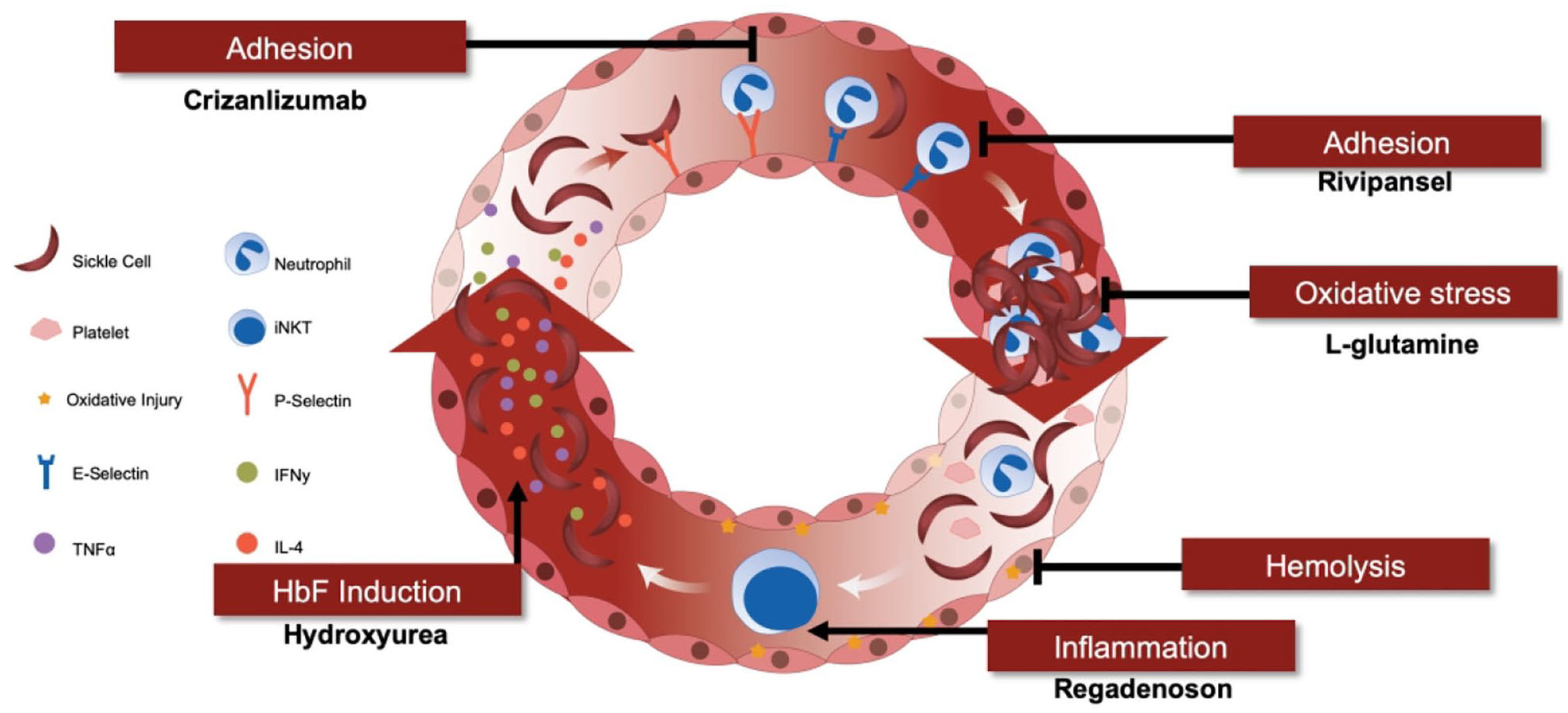
Sickle cell pain crises: role of pharmaceutical interventions in sequential pathogenic mechanisms, including adhesion, oxidative stress, inflammation, hemolysis, and HbF induction.
Current strategies to reduce the occurrence of VOCs
Prior to 2019, there were four available treatment modalities to mitigate the acute and chronic complications of SCD: hydroxyurea (HU), RBC transfusions,
Hydroxyurea
Until recently, the only medication approved to reduce the occurrence of VOCs was HU, the first drug approved by the United States Food and Drug Administration (FDA) for treatment of SCD, and transfusion therapy. HU is believed to be effective for several reasons: induction of fetal hemoglobin (HbF) to prevent RBC sickling; myelosuppression (reducing the overall number/quantity of potentially activated blood cells) leading to decreased available white blood cells, platelets, and reticulocytes; and potentially improving nitric oxide donor bioavailability. In preclinical models, HU was demonstrated to reduce the heterotypic cellular adhesive interactions, and, thus, has multiple beneficial effects in SCD.21,22
The phase III Multicenter Study of Hydroxyurea (MSH study) demonstrated significant reduction in rates of acute VOC, acute chest syndrome, and the need for transfusions among patients ⩾18 years of age with SCA (HbSS or HbSB0) receiving HU, which led to HU gaining FDA approval in 1998.23,24 In 2010, the long-term benefits and risks of HU in SCA were reported in a 17.5-year follow-up study.
25
Results showed persistently high overall mortality rates for SCD of 43.1%, but reduced mortality among those individuals with long-term HU exposure, as 87% of these deaths occurred among individuals who never took HU or had less than 5 years of exposure. The BABY-HUG trial conducted in children aged 9–18 months at randomization also showed a significant reduction in reports of pain [hazard ratio (HR) 0.59, 95% confidence interval (CI): 0.42–0.83;
While HU is highly effective in reducing pain crisis for some individuals with SCD, many individuals continue to have VOC despite taking HU, attesting to myriad mechanistic pathways that may be responsible for VOC that may not be fully mitigated with HU. HU also has several side effects requiring frequent monitoring, and can cause hair thinning, nail bed/skin color changes, and stomach upset. Thus, many individuals with SCD are opposed to taking HU, or are intolerant of the medication. A recent Cochrane review concluded that HU was effective in reducing pain crises for individuals with SCD based on four trials conducted in 577 individuals with SCD that compared HU and placebo. Increases in HbF and reductions in neutrophil counts were also observed.
27
Contrary to other smaller studies, this review did not show a difference between individuals receiving HU
Chronic transfusion therapy
Chronic transfusion therapy is used primarily to reduce the risk of stroke in high-risk individuals with SCD.31,32 However, it can also be used to reduce the levels of circulating sickled RBCs, and thereby decrease the number of VOCs when used as prophylactic therapy. 33 Complications of chronic transfusion therapy include the risk of allo-immunization, iron overload, and its related complications. The utility of chronic transfusion therapy in SCD is limited by the availability of a compatible blood donor population. Individuals undergoing transfusion for SCD may also generate antibodies against the transfused RBCs, resulting in unpredictable, delayed hemolytic transfusion reactions and thus require phenotypically matched blood. 34
Despite improved implementation of HU and chronic transfusion therapy for disease management in SCD, recurrent VOCs continue to be associated with disease severity and mortality. Of greater concern is the increased risk of early mortality in individuals with more frequent VOCs. In a study of 264 subjects with SCD, there was a significantly younger age of death (55.8 years
l -Glutamine
Hematopoietic stem cell transplant
HSCT is a curative approach that has traditionally been reserved only for those patients with severe SCD and complications such as stroke, recurrent acute chest syndrome, impaired neuropsychologic function, recurrent VOC, sickle lung disease, or nephropathy.
42
The first successful HSCT in a patient with SCD was reported in 1984.
43
Since that first transplant using cyclophosphamide and fractionated whole body irradiation, outcomes for HSCT have improved with the introduction of other conditioning regimens, including busulfan, fludarabine, and anti-thymocyte globulin.44,45 Despite being potentially curative, HSCT is optimized in individuals with a matched sibling donor, which is not universally available. Furthermore, the risks of HSCT, including infertility and graft-
New approaches: recent investigations
In this review, we summarize some of the recent mechanistic approaches for the treatment of patients with SCD.
Oxygen affinity agent
Voxelotor, formerly known as GBT440, was approved by the FDA in 2019 for the treatment of SCD in adults and pediatric patients 12 years of age or older.
48
This treatment is a first-in-class orally administered agent that increases the affinity of hemoglobin for oxygen, thereby inhibiting the polymerization of HbS.
49
In a phase I/II randomized, double-blinded, placebo-controlled study, the safety, tolerability, pharmacokinetics, and pharmacodynamics of voxelotor were evaluated in subjects with SCD. Subjects treated for ⩾28 days experienced improvements in hemoglobin and mild decreases in hemolysis, and the drug was well tolerated.
49
In other studies of voxelotor in healthy volunteers (
Fetal hemoglobin inducers
Higher levels of HbF have been shown to be protective against VOC and other pathological consequences of SCD due to its effect on inhibiting HbS polymerization.52,53 As previously discussed, HU has been shown to increase levels of HbF, demonstrating improved outcomes and lower incidence of VOCs.23,54 Other small molecule inducers have been shown to increase HbF expression for individuals with SCD. One such molecule, HQK-1001, was tested in a phase II study. Unfortunately, the results of the study were negative, with no significant increase in HbF and a trend for more pain crises in the HQK-1001 group. 55 Due to the modest impact of this agent, it is not being investigated further.
Decitabine and tetrahydrouridine
A recent phase I study examined the impact of decitabine in combination with tetrahydrouridine. 56 Results from this study showed significant increases in both HbF and total hemoglobin concentrations, and accompanying improvements in biomarkers of hemolysis, coagulation, and inflammation. The treatment was well tolerated, and the combination therapy is undergoing further clinical development for individuals with SCD [ClinicalTrials.gov identifier: NCT04055818]. To date, there are no data on the impact of decitabine plus tetrahydrouridine on clinical endpoints, including VOC.
Anti-inflammatory agents
Inflammation is a key step in the cascade of events leading to vaso-occlusion and VOC. As a result, agents targeting inflammatory processes specific to SCD as well as anti-inflammatory agents with a broader scope are of interest.
Regadenoson
Regadenoson is an adenosine A2A receptor agonist that mediates the anti-inflammatory effects of invariant natural killer T (iNKT) cells – a subset of lymphocytes that have been implicated in the pathogenesis of SCD.
57
In a phase II, randomized, placebo-controlled trial, subjects with SCD admitted to the hospital for a VOC (
NKTT120
NKTT120 is a humanized monoclonal antibody that has been shown to cause rapid and sustained deletion of iNKT cells in SCD patients without infusion-related toxicity or serious AEs in an open-label study. 58 This agent may offer the advantage of dosing every 3 months by infusion as opposed to daily dosing. Next steps for this agent are a randomized controlled clinical trial to determine its efficacy in reducing VOCs and determining the safety of prolonged iNKT cell depletion. 58
Simvastatin
The cholesterol-lowering agent simvastatin may have a beneficial impact in SCD by virtue of its effects on improving endothelial function, restoring nitric oxide production, and suppressing inflammation; although the exact mechanism of action is not clear, short-term treatment with simvastatin in patients with SCD has been shown to improve soluble biomarkers of inflammation.59,60 In a single-center pilot study of 19 individuals with sickle cell anemia, treatment with simvastatin for up to 3 months resulted in reduced sickle cell-related pain, oral analgesic use, and improvement in biomarkers of inflammation, with no simvastatin-related safety concerns or AEs reported in the study. 60 Despite the potential for myalgia associated with statin therapy, no clinical safety concerns or simvastatin-related AEs were observed. 60 Notably, the greatest reductions in pain and inflammatory markers were observed in those individuals who also received HU, suggesting a synergistic effect of combining these two therapies. 60 In light of these encouraging results, larger controlled studies are expected to evaluate the long-term efficacy and safety of simvastatin (alone or in combination with HU) in subjects with SCD.
Omega-3 fatty acids
Omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) have a number of pleiotropic effects that may have a role in SCD. In addition to their anti-inflammatory effects
The phase II Sickle Cell Omega-3 Treatment (SCOT trial) evaluated the use of SC411, a DHA ethyl ester formulation, in a multicenter population of 67 children with SCD.
64
The trial met its primary endpoint, demonstrating a significant increase in RBC membrane DHA and EPA (
Antiplatelet agents
Platelets likely contribute to the vaso-occlusive process both as procoagulants and inflammatory mediators. The impact of modulators of platelet activation and their receptor-ligand interactions with other cells is currently under investigation to reduce vascular inflammatory processes and prevent VOCs in individuals with SCD.19,65
Prasugrel
Prasugrel is a third-generation thienopyridine that irreversibly inhibits ADP-mediated platelet activation and aggregation. This agent is approved for use in adults with acute coronary syndrome managed with percutaneous coronary intervention, and was investigated for the prevention of VOCs in the phase III, placebo-controlled DOVE trial.29,66 Children 2–17 years old with SCD (
Ticagrelor
Ticagrelor is an inhibitor of platelet aggregation approved for use in adults with acute coronary syndrome and following myocardial infarction.
67
As with prasugrel, the rationale for the use of ticagrelor in SCD relates to the increased activation state of platelets in SCD and their further activation during VOCs.
67
In a two-part, phase II, multicenter dose-finding study (HESTIA1), a favorable dose-exposure-response relationship was demonstrated in a population of children with SCD, and the agent was well tolerated, with no increased risk of bleeding.
67
In another phase IIb study of ticagrelor in adult subjects (
Anti-adhesion agents
Molecules that inhibit selectin binding and adhesion are a potential method for reducing or improving vaso-occlusion. These molecules are especially appealing given their potential for use in combination with RBC-targeting agents described above.
Crizanlizumab
P-Selectin is one of several targets implicated in the increased adhesion activity associated with VOCs.
19
The impact of crizanlizumab, a humanized monoclonal antibody that binds P-selectin, thereby blocking its interaction with PSGL-1, has been evaluated with or without HU therapy in the phase II SUSTAIN trial.
28
In this study, subjects with SCD aged 16–65 years and 2–10 pain crises within the past 12 months (
Additional agents under investigation
Vitamin D
A recent study has examined environmental and dietary factors, including vitamin D levels, and their association with crisis events in patients with SCD. The results showed that diets high in fish, milk, cheese, and eggs were associated with reduced hospital/ED visits in individuals with SCD.
71
The study also showed statistically fewer crisis-related hospitalizations for individuals with vitamin D levels >24 ng/ml; among 102 individuals with SCD, mean hospitalizations over 12 months were higher for those with severe vitamin D deficiency (0–19 ng/ml, 9 days) and mild to moderate deficiency (20–29 ng/ml, 5 days) compared with those who were not deficient (>30 ng/ml, <3 days;
Potential targets in vascular pathways
The interaction of endothelin-1 (ET-1), a mediator of neutrophil recruitment and activation, with the endothelin B receptor (ETB) has been shown to be an important mediator of neutrophil recruitment and adhesion to activated endothelium in experimental studies of SCD mice.
74
The ET-1/ETB axis is thus under investigation as a pro-inflammatory pathway in SCD, and ET receptor inhibitors, if proven safe and efficacious in humans, may be another means of preventing VOCs in individuals with SCD.
74
Activation of the alternative complement pathway, characterized by microvascular deposition of C5b-9, has also been observed in the skin of individuals with SCD (but not healthy controls), and deposition of C3b has been observed on RBC membranes in individuals with SCD.
75
Factor H, an inhibitor of the alternative complement pathway, inhibits the interaction of sickled erythrocytes with the endothelium
Anti-neutrophil agents
There is evidence that a reduction in neutrophils has a beneficial impact on the occurrence of VOCs in individuals with SCD, particularly those receiving HU therapy.24,27 Thus, reducing neutrophil interactions with the activated endothelium is another avenue of investigation for the prevention of VOCs in individuals with SCD. 74
cGMP-modulating agents
Cyclic guanosine-3′5′-monophosphate (cGMP) promotes the production of HbF in erythroid cells, reduces leukocyte adhesion mechanisms, decreases endothelial adhesion molecule expression, and improves vasorelaxation. 76 cGMP production is regulated by nitric oxide binding to soluble guanylate cyclase.16,77 As already discussed, cell-free hemoglobin and arginase I reduce nitric oxide, resulting in endothelial dysfunction.13,15,16 As a result, decreased nitric oxide may result in reduced cGMP. A number of products in early clinical development have been shown to increase cGMP and therefore may have a role in mitigating the vasculopathy and nitric oxide depletion in SCD. Olinciguat stimulates soluble guanylyl cyclase, an enzyme that catalyzes the conversion of guanosine-5′-phosphate (GTP) to cGMP. 76 This compound demonstrated increased cGMP and decreased blood pressure in phase I trials with no renal clearance, suggesting it may be appropriate for use in patients with renal impairment. 76 Olinciguat is currently being studied in a phase II clinical trial in patients with SCD [ClinicalTrials.gov identifier: NCT03285178].
Another possible mechanism of cGMP in SCD is through activation of protein kinase G and induction of HbF.16,76,78 IMR-687 is a phosphodiesterase-9 inhibitor (PDE9) that acts by preventing degradation of cGMP by PDE9. It has also demonstrated induction of HbF in animal models and human cell lines. 78 Interim results from a phase II study presented at the European Hematology Association Congress in 2019 showed a trend toward increased HbF and reduced hemolysis and cellular adhesion factors in SCD patients treated with IMR-687 100 mg orally once a day. 79
cGMP-amplifying agents have been shown to intensify the nitric oxide-mediated effects of HU in animal models. 22 As a result, it is possible that all of these agents may act synergistically with HU.
Gene therapy
In several trials [ClinicalTrials.gov identifiers: NCT02140554, NCT02151526, NCT04293185, NCT02633943], gene addition therapy is performed using an
Gene editing studies using the CRISPR/CAS-9 technology are also underway. The majority of these studies are using CRISPR to disrupt genes that fetal hemoglobin production by targeting genes such as BCL11A.82,83 These studies, also in phase I/II clinical trials, are also demonstrating very exciting potential for disease-modifying effect.
Intravenous immunoglobulin
Intravenous immunoglobulin (IVIG) inhibits Mac-1 dependent capture of RBCs by neutrophils, and has been shown to reduce neutrophil adhesion to post-capillary venular endothelium and interactions with circulating RBCs in murine models of sickle cell acute pain crisis.84–86 In a phase I study of patients with SCD with acute pain crisis, IVIG-treatment resulted in a significant decrease in Mac-1 function. 87 An ongoing randomized, placebo-controlled, phase I/II trial in SCD is ongoing to evaluate whether IVIG reduces the duration of VOC [ClinicalTrials.gov identifier: NCT01757418].
Conclusion
Acute pain remains the primary reason for which individuals with SCD currently seek acute care. While frequency, severity, and duration may vary, VOC affects many individuals with SCD.2,88 Therapies including HU,
At this time, it remains unclear if any therapy will be useful in shortening or stopping a VOC once it is initiated. Several studies have failed in this regard in recent years. While it is possible that these large studies have not met their primary endpoints due to flawed study designs and difficulty in quantifying or defining the end of a VOC, it is also possible that the vascular occlusive process cannot be altered once initiated. Indeed, the definition of pain events and pain crisis is a limitation for all of the studies of therapies discussed here, especially if dependent on patient report. This may be particularly complicated in adults who may also suffer from complex chronic pain.
As individuals are living longer with SCD, an integrative and likely multimodal approach to preventing and treating VOCs is needed. Whereas acute management of pain associated with VOCs will likely remain an essential component of treatment, the risk of opioid tolerance, reduced efficacy, and dependency over the long term remains. Furthermore, while other nonsteroidal medications are also used to decrease inflammation, their use is limited by renal dysfunction and bleeding as well as gastrointestinal complications. Other non-opioid pain therapies remain in use but have not demonstrated significant efficacy. Similarly, while they may improve patient coping/management strategies for VOCs, cognitive and/or behavioral approaches to recurrent pain associated with VOCs are limited by insufficient funding for mental health providers to administer these therapies and disparate access to sickle cell centers that would typically have these services available. Of greatest concern for the development of therapies to decrease the length of a VOC is the lack of biomarkers outside of a subjective pain report. Furthermore, there are no predictive biomarkers to identify the highest-risk subjects and/or to prioritize the use of these therapies. Lastly, although they remain to be proven in larger numbers of subjects, gene therapies have the potential to reverse or halt the disease process in SCD by introducing wild-type hemoglobin. As these therapies develop, it will be important to have initiatives that allow for greater access to specialized care and treatment once they become available.
Footnotes
Acknowledgements
Editorial support in the preparation of this manuscript was provided by Jennifer Lee of Phase Five Communications, supported by Novartis Pharmaceuticals Corporation. The authors were responsible for all content and editorial decisions and received no honoraria related to the development of this manuscript.
Conflict of interest statement
IO has received research funding from Health Resources and Services Administration, North Carolina Division of Public Health, and Patient-Centered Outcomes Research Institute; served as a consultant for Cyclerion Therapeutics Inc., Global Blood Therapeutics, Novartis Pharmaceuticals Corp., and Pfizer Inc; participated on advisory boards for Acceleron Biopharma, Forma Therapeutics, Global Blood Therapeutics, Novartis Pharmaceuticals Corp., and Pfizer Inc; participated on the speakers’ bureau for Global Blood Therapeutics, Novartis Pharmaceutics Corp., and Terumo Medical Corporation; and is a member of the Data and Safety Monitoring Board for Micelle BioPharma Inc.; currently serves as Editor in Chief of
DM has received research funding from bluebird bio and Global Blood Therapeutics, and served as a consultant for Global Blood Therapeutics, Novartis Pharmaceuticals Corp., and Pfizer Inc.
JK has received research funding from Health Resources and Services Administration, Center for Disease Control, and National Institute of Health as well as from bluebird bio and Novartis Pharmaceuticals Corp.; served as a consultant for bluebird bio, Imara Inc., Modus Therapeutics, Novartis Pharmaceuticals Corp., Agios, Beam Therapeutics, and Sanofi; and received honoraria from Bluebird Bio, Global Blood Therapeutics and Terumo Medical Corporation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
