Abstract
Epigenetics refers to the regulation of gene expression mainly by changes in DNA methylation and modifications of histone proteins without altering the actual DNA sequence. While epigenetic modifications are essential for normal cell differentiation, several driver mutations in leukemic pathogenesis have been identified in genes that affect epigenetic processes, such as DNA methylation and histone acetylation. Several therapeutic options to target epigenetic alterations in acute myeloid leukemia (AML) have been successfully tested in preclinical studies and various drugs have already been approved for use in clinical practice. Among these already approved therapeutics are hypomethylating agents (azacitidine and decitabine) and isocitrate dehydrogenase inhibitors (ivosidenib, enasidenib). Other agents such as bromodomain-containing epigenetic reader proteins and histone methylation (e.g. DOT1L) inhibitors are currently in advanced clinical testing. As several epigenetic therapies have only limited efficacy when used as single agents, combination therapies that target AML pathogenesis at different levels and exploit synergistic mechanisms are also in clinical trials. Combinations of either epigenetic therapies with conventional chemotherapy, different forms of epigenetic therapies, or epigenetic therapies with immunotherapy are showing promising early results. In this review we summarize the underlying pathophysiology and rationale for epigenetically-based combination therapies, review current preclinical and clinical data and discuss the future directions of epigenetic therapy combinations in AML.
Keywords
Introduction
Acute myeloid leukemia (AML) is the most common form of acute leukemias in adults and, while pathogenetically heterogenous, it is typically caused by genetic events affecting hematopoietic progenitor or stem cells leading to the clonal proliferation of abnormally differentiated or undifferentiated myeloid cells. 1 Despite the identification of several genetic abnormalities underlying AML, the mainstay of AML therapy for fit patients for several decades has been intensive induction chemotherapy with anthracyclines and cytarabine, followed by consolidation chemotherapy or allogeneic hematopoietic stem cell transplantation.2,3 However, intensive chemotherapy is generally only an option for younger and medically fit patients. Unfortunately, the majority of AML patients are older than 65 years with multiple comorbidities and are often ineligible for intensive chemotherapy with long-term survival rates of 10% or less.4–6
Histone proteins provide a scaffold for storage of DNA within the nucleus of eukaryotic cells. The macromolecular complex of DNA and histone proteins is referred to as chromatin.
7
Chromatin modification altering interactions between DNA and histones is a highly dynamic process in cells affecting transcription, DNA repair and replication.
7
Epigenetics are most commonly defined as changes to the chromatin structure which can occur in the form of DNA methylation, histone modifications, and changes to higher-order chromatin structures that ultimately affect gene expression.8,9 Epigenetic regulators can be broadly classified as ‘writers’ (e.g. DNA and histone methyltransferases), ‘erasers’ (e.g. histone deacetylase) or ‘readers’ (e.g. bromodomain-containing proteins).
10
The importance of epigenetics in cancer development has been well documented for several decades for both solid and hematologic malignancies.11,12 Especially in AML, several specific mutations affecting epigenetic processes such as histone modification [Enhancer of Zeste Homologue 2 (
DNA methylation and DNA hydroxymethylation are key epigenetic pathways that have been linked to malignant transformation by inactivating tumor suppressor genes.7,16 Mutations in genes affecting DNA methylation (e.g.
Besides DNA methylation, histone acetylation is a highly dynamic process of modifying gene transcription that is tightly regulated by the competing activity of histone lysine acetyltransferases and histone deacetylases (HDACs) with histone acetylation often leading to a more accessible chromatin structure that promotes gene transcription.
7
Mutations in other genes affecting histone modifying enzymes such as
Overall, the therapeutic efficacy of HMAs and HDAC inhibitors are limited when used as single agents. Combination strategies of epigenetic therapy with either conventional chemotherapy, immunotherapy, or other forms of targeted therapies such as fms-related tyrosine kinase 3 (

Overview of epigenetic mechanisms and selected therapeutic interventions.
Combination of HMAs with other epigenetic therapy
Combinations of HMAs and HDAC inhibitors
Overview of selected clinical trials for combination of HMAs and HDAC inhibitors in AML treatment.
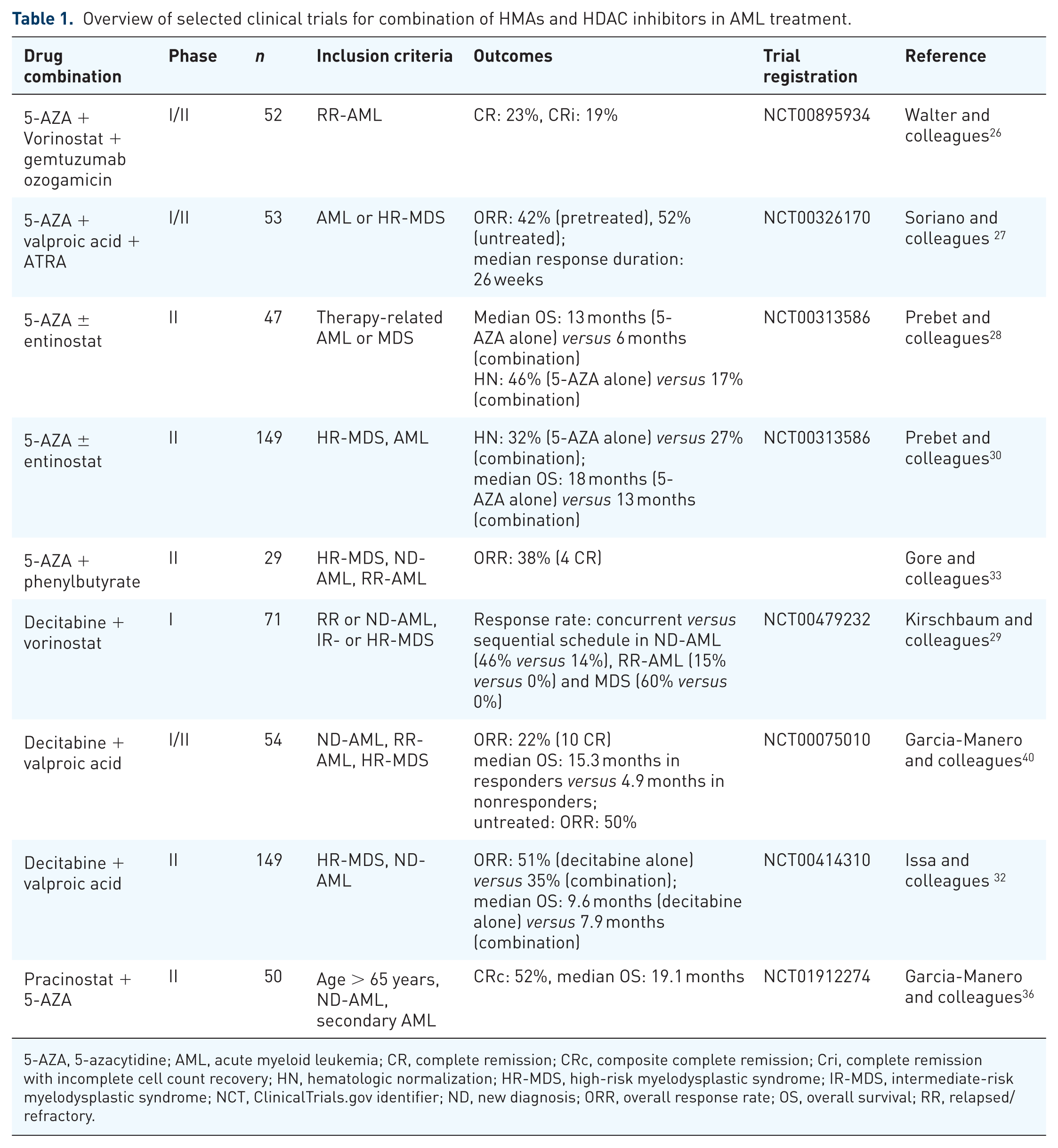
5-AZA, 5-azacytidine; AML, acute myeloid leukemia; CR, complete remission; CRc, composite complete remission; Cri, complete remission with incomplete cell count recovery; HN, hematologic normalization; HR-MDS, high-risk myelodysplastic syndrome; IR-MDS, intermediate-risk myelodysplastic syndrome; NCT, ClinicalTrials.gov identifier; ND, new diagnosis; ORR, overall response rate; OS, overall survival; RR, relapsed/refractory.
Combination of HMAs and isocitrate dehydrogenase inhibitors
Methylation of cytosine-guanine dinucleotides (CpG) within the DNA is one of the key epigenetic mechanisms that regulate gene transcription and is mediated mainly by a tightly-controlled enzyme called DNA methyltransferase.11,41 On the other hand, 5-hydroxymethylation of these cytosine residues by α-ketoglutarate-dependent enzymes such as ten eleven translocation (TET) DNA methylases leads to DNA demethylation.42,43 Under physiological conditions, isocitrate dehydrogenase (IDH) catalyzes the conversion of isocitrate to α-ketoglutarate in the Krebs cycle. However, mutations in
Since one of the mechanisms by which IDH mutations contribute to leukemogenesis is DNA hypermethylation, combination therapy of IDH inhibitors and HMAs seems to be a promising therapeutic strategy. Preclinical data have suggested a synergistic effect of enasidenib and 5-AZA leading to the initiation of a clinical phase I/II trial of enasidenib and 5-AZA (ClinicalTrials.gov identifier: NCT02677922) and a phase III trial of ivosidenib and 5-AZA for newly diagnosed AML patients ineligible for standard intensive chemotherapy (ClinicalTrials.gov identifier: NCT03173248).45,52,53 While neither of these trials has been published in a peer-reviewed journal yet, data available in abstract form seem promising with an ORR of 73% [
Combination of HMAs and novel epigenetic therapies
So-called ‘epigenetic readers’ comprise a class of proteins that specifically bind to distinct DNA or histone modifications and constitute the third class of epigenetic regulators besides ‘epigenetic writers’ and ‘epigenetic erasers’.
10
Rearrangements of the
Similar to DNA methylation, histone methylation and demethylation are highly dynamic processes that can lead to either activation or repression of transcription depending on the specific location of the modified lysine residue. 74 Histone methylation is catalyzed by specific methyltransferases such as MLL1/2, DOT1L, and EZH1/2, while histone demethylation is mainly regulated by the activity of lysine-specific histone demethylase (LSD) 1 and LSD2.74,75 Mutations in the involved genes and resulting overexpression of the encoded enzymes have been documented in both primary and secondary AML. 74 Pharmacological inhibition of LSD1 (ORY-1001, GSK2879552, IMG-7289) has been shown in animal models to induce differentiation of AML blasts and has been tested in phase I/II clinical trials (ClinicalTrials.gov identifiers: NCT02177812, NCT02842827, EUDRACT no. 2013-002447-29).76–80 While a phase I trial of the LSD1 inhibitor GSK2879552 has been stopped recently due to a negative risk-benefit assessment, preclinical data suggest synergistic effects of the combination of LSD1 inhibitors with the HDAC inhibitor panobinostat and all-trans-retinoic acid (ATRA). 81 LSD1 inhibition has been shown to increase HDAC inhibition and to block activity of the transcription factor c-Myc which can be further enhanced by addition of HDAC inhibitors.77,81
EZH1 and EZH2 are epigenetic regulators that methylate histones (H3K27) and repress transcription of target genes. Dysfunction of EZH1/2 has been associated with unregulated self-renewal of leukemic stem cells and a poor prognosis of AML and MDS patients harboring these mutations.82,83 Recent preclinical studies showed synergistic effects of EZH2 inhibition with the HMA decitabine and the HDAC inhibitor panobinostat.83–85 The underlying mechanism is the enhanced effect on gene silencing by simultaneously preventing DNA methylation by HMAs or histone acetylation with HDAC inhibitors in combination with increasing the pro-transcriptional effect of histone methylation by EZH2 inhibition. 83 Additionally, trimethylation of histone H3K27 by EZH2 has been shown to mark genes for silencing by DNA methylation and is one of the mechanisms that has been linked with decitabine resistance.86–88 However, neither of these combinations has yet been tested in clinical trials.
Combination of epigenetic therapy with conventional chemotherapy
Combination of HMAs and conventional chemotherapy
Several
Overview of selected clinical trials for combination of epigenetic therapies with conventional chemotherapy in AML treatment.

5-AZA, 5-azacitidine; AML, acute myeloid leukemia; CML, chronic myelogenous leukemia; CR, complete remission; CRi, complete remission with incomplete cell count recovery; HR-MDS, high-risk myelodysplastic syndrome; HSCT, hematopoietic stem cell transplant; NCT, ClinicalTrials.gov identifier; ND, new diagnosis; ORR, overall response rate; OS, overall survival; Ref, reference; RR, relapsed/refractory.
Combination of HDAC inhibitors and conventional chemotherapy
While having only moderate antileukemic effects as single agents, HDAC inhibitors have shown synergistic activity in combination with various forms of conventional chemotherapy such as nucleoside analogues (cytarabine, fludarabine), anthracyclines, and topoisomerase inhibitors (etoposide).40,103–105 The underlying mechanism for this synergy is not completely understood, but it is hypothesized that HDAC inhibitors promote a more open chromatin structure that might allow for better access of topoisomerase inhibitors to the DNA and therefore higher efficacy of chemotherapeutics. 105 Other studies investigating the synergy between doxorubicin and panobinostat implicated DNA double-strand breaks and activation of caspase-dependent apoptosis pathways in the antileukemic efficacy. 104
This concept has also been tested in various clinical trials. In a phase II trial of vorinostat in combination with cytarabine and idarubicin of 75 newly diagnosed AML or high-risk MDS patients ORRs were 85% and addition of vorinostat did not lead to an increase in toxicity.
99
Interestingly, 100% of patients with
Combination of epigenetic therapy with targeted therapy
Combination of epigenetic therapy and venetoclax
B-cell leukemia/lymphoma-2 (BCL-2) is an anti-apoptotic protein that inhibits cell death by blocking permeability of the mitochondrial outer membrane. 106 Though first identified in follicular lymphoma, BCL-2 is also overexpressed in other hematologic malignancies including AML and has been implicated in leukemia stem cell survival.107,108 BCL-2 activity is regulated by a group of small molecules known as BH3-mimetics that bind to and inhibit the BH3 domain of BCL-2 proteins which releases proapoptotic factors from their BCL-2 binding site and triggers apoptosis. 109
Venetoclax (ABT-199) is an oral, highly-specific BCL-2 inhibitor which is United States Food and Drug Administration (US FDA)-approved for the treatment of chronic lymphocytic leukemia.110,111 Venetoclax has recently been shown to be modestly effective as a single agent in the treatment of RR-AML or newly diagnosed AML patients who are ineligible for intensive chemotherapy. 112 However, early-phase clinical data have shown both an impressive synergistic effect and acceptable safety profile of venetoclax in combination with HMAs (ClinicalTrials.gov identifier: NCT02203773; composite CR: 61%, median OS: 17.5 months) or low-dose cytarabine (ClinicalTrials.gov identifier: NCT02287233; CR/CRi: 62%; median OS: 11.4 months) in the frontline setting in elderly AML patients ineligible for intensive chemotherapy.107,113,114 Acknowledging the remarkable response rates that exceed historical outcomes with 5-AZA alone (composite CR: 28%, median OS: 10.4 months), 115 the combination of 5-AZA and venetoclax received breakthrough designation by the US FDA and is currently tested in a phase III clinical trial against 5-AZA monotherapy. 106 A similar trial of cytarabine in combination with venetoclax is also currently recruiting (ClinicalTrials.gov identifier: NCT03069352).
The combination of venetoclax and either HMAs or low-dose cytarabine appears to have activity in the AML salvage therapy setting based on data which reported objective response rates of 21% and a median OS of 3.0 months. 107 While these numbers seem modest at a first glance, it must be kept in mind that 94% of these patients had been pretreated and 41% of the patients had failed ⩾3 previous lines of therapy. 112
On a molecular basis this synergy can be explained by the fact that resistance to venetoclax is mediated by the antiapoptotic proteins BCL-XL and MCL1 which can be overcome by combination therapy with HMAs, daunorubicin or cytarabine.116,117 On the other hand, resistance of leukemic blasts to chemotherapy has been linked to overexpression of BCL-2.
118
Therefore, combining venetoclax with HMAs targets resistance mechanisms that have hampered monotherapy with either of these agents and seems to make this combination highly effective.
107
Of note, subgroup analysis has shown that patients with
Combination of epigenetic therapy and FLT3 inhibitors
Addition of the multikinase inhibitor midostaurin to intensive induction chemotherapy was recently shown to yield a higher response rate than chemotherapy alone leading to its US FDA-approval for the treatment of
In addition to midostaurin, several more specific
Overview of selected clinical trials for combination of hypomethylating agents and targeted therapies in AML treatment.

5-AZA, azacitidine; AML, acute myeloid leukemia; CMML, chronic myelomonocytic leukemia; CR, complete remission; Cri, complete remission with incomplete cell count recovery; HR-MDS, high-risk myelodysplastic syndrome; LDAC, low-dose cytarabine; NCT, ClinicalTrials.gov identifier; ND, new diagnosis; ORR, overall response rate; OS, overall survival; Ref, reference; RR, relapsed/refractory; s-AML, secondary AML.
Combination of epigenetic therapy and immunotherapy
Evasion of immunosurveillance by epigenetic silencing of genes involved in immune recognition and effector T-cell function is an essential feature of cancer cells and contributes to the immunosuppressive tumor microenvironment. 133 HMAs have been shown to increase the expression of leukemia-associated antigens such as NY-ESO-1 and MAGE-A that can potentially trigger an antileukemia immune response.134–138 Additionally, they contribute to immune system activation by increasing the expression of major histocompatibility complex (MHC)-I and co-stimulatory molecules (ICAM1, CD80, CD86).135,138,139 However, at the same time HMAs are also hampering antitumor immune response by upregulating the expression of immune checkpoint molecules such as programmed cell death (PD)-1/programmed death ligand (PD-L)1 and cytotoxic T-lymphocyte-associated protein (CTLA)-4 which may contribute to treatment failure with HMAs.140–142 However, the increased expression of PD-1/PD-L1 with HMA treatment may lead to a greater susceptibility of cancer cells to treatment with PD-1/PD-L1 inhibitors. Preclinical experiments have shown that pretreatment with 5-AZA led to an enhanced response to treatment with immunotherapeutics such as CTLA-4 inhibitors. 143 Studies in non-small cell lung cancer patients have provided additional proof of principle that combing HMAs with PD-1/PD-L1 inhibitors sensitized patients to treatment with PD-1 inhibitors.
These findings have inspired various clinical trials that combine HMAs with various PD-1 inhibitors [nivolumab (ClinicalTrials.gov identifier: NCT02397720), and pembrolizumab (ClinicalTrials.gov identifier: NCT02845297)], PD-L1 inhibitors [durvalumab (ClinicalTrials.gov identifier: NCT02775903), and atezolizumab (ClinicalTrials.gov identifier: NCT02508870)], or the CTLA-4 inhibitor ipilimumab (ClinicalTrials.gov identifiers: NCT02890329, NCT02397720). One study demonstrated a 33% ORR (22% CR/CRi) and median OS of 6.3 months in RR-AML patients treated with nivolumab and 5-AZA combination therapy (
Future directions
Our understanding of the epigenetic processes underlying AML is still incomplete. With further improvement in and wider availability of diagnostic techniques such as next-generation sequencing, an increasing number of mutations affecting epigenetic regulators is being discovered, but their precise impact on AML development and their suitability as therapeutic targets remains to be elucidated in preclinical experiments. However, identification of these mutations has already led to the development and approval of drugs that specifically target these mutations which has paved the way to a more individualized treatment approach for AML patients. Nevertheless, several important questions remain to be answered.
One of the major challenges remaining is the appropriate selection of patients who are most likely to benefit from an intervention. While targeted therapies such as IDH inhibition with ivosidenib and enasidenib only work in patients with IDH mutations, predicting treatment response to HMAs or HDAC inhibitors is difficult. Methylation of CpG islands has been suggested to be a biomarker predicting response to HMAs. 145 However, subsequent studies showed that methylation status is dynamic while patients are undergoing treatment and that it depends rather on the methylation status of certain individual genes than on the overall CpG methylation status.146,147 Additionally, epigenetic therapies such as DOT1L and BET inhibition can be successful even if the target gene is not mutated at all which questions the utility of simple gene mutation testing and underlines the need for broader drug sensitivity testing. 13 The combination of different mutations can predict treatment response as well which is seen in the higher response rate of patients with concomitant IDH mutations who are treated with venetoclax. Further studies are warranted to guide appropriate drug selection. 112
Various studies have shown that targeting a single genetic mutation such as
Finally, preleukemic hematopoietic stem cells already harbor some mutations in genes that are involved in epigenetic changes such as DNA methylation and histone modification.
148
Interestingly, these progenitor cells can survive induction chemotherapy and have been linked to disease relapse.
149
As some of the earliest mutations occurring during the transformation of normal HSCs to leukemia clones are seen in
Conclusion
Epigenetic therapy in AML is still in its infancy but is a rapidly evolving and highly promising field. While early forms of epigenetic therapies like the HMAs 5-AZA and decitabine have shown modest effects, more targeted therapies such as IDH inhibitors have shown a better response and survival rates in appropriately selected patients. Combination therapies of either epigenetic therapies with conventional chemotherapy, different forms of epigenetic therapies, or epigenetic therapies with immunotherapy promise even higher success rates even in elderly and RR-AML patients as they target cell proliferation at different levels. However, further research is needed to identify biomarkers that predict response to therapy and guide drug selection for individual patients.
Footnotes
Acknowledgements
Amer Zeidan is a Leukemia and Lymphoma Society Scholar in Clinical Research and is also supported by a NCI’s Cancer Clinical Investigator Team Leadership Award (CCITLA).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
A.M.Z. had a consultancy with and received honoraria from AbbVie, Otsuka, Pfizer, Celgene, Ariad, Agios, Novartis, Acceleron, Astellas, Daiichi Sankyo and Takeda; and received honoraria from and was a speaker for Takeda. The remaining authors declare no competing financial interests.
