Abstract
Medical decisions in hemophilia care are primarily related to the type of factor replacement and treatment regimen. With the growing number of treatment options for patients with hemophilia, decision making is more complex and requires careful consideration of benefits, risks, and patient goals. Shared decision making and decision-aid tools facilitate patient and healthcare provider communication. In this review, the overall role of shared decision making in medicine is outlined, with special emphasis on models for practical implementation. Examples of shared decision making in hemophilia are outlined, and application to new therapeutics is discussed through a case-based approach.
Keywords
Introduction
The current era of hemophilia treatment is one of rapid and disruptive therapeutic advances including extended half-life (EHL) clotting factor replacement, nonfactor replacement products, and gene therapy (Table 1).1–3
Treatment options for patients with hemophilia.
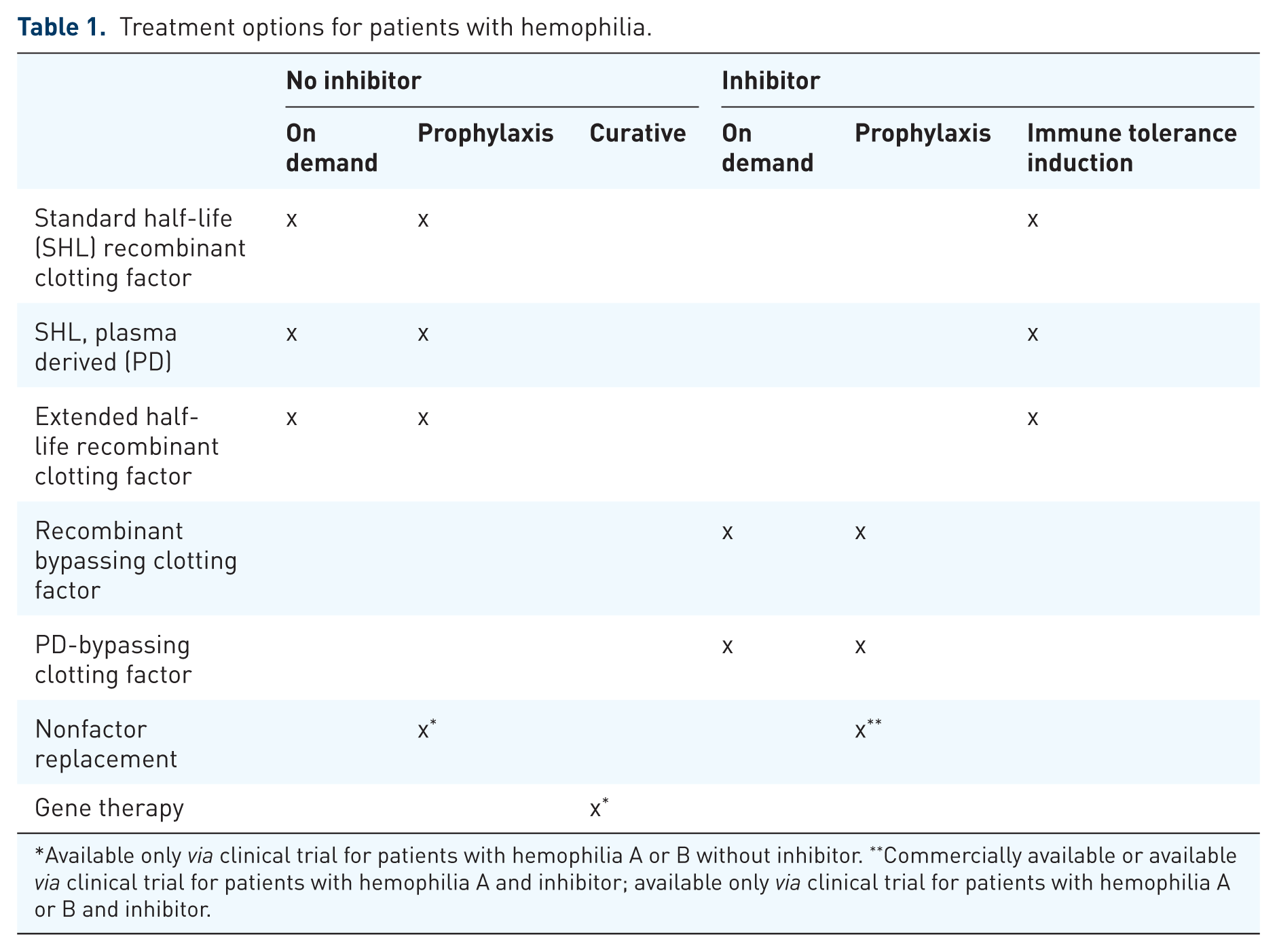
Available only via clinical trial for patients with hemophilia A or B without inhibitor. **Commercially available or available via clinical trial for patients with hemophilia A and inhibitor; available only via clinical trial for patients with hemophilia A or B and inhibitor.
Therapeutic advances are impacting disease management and goals of therapy, and are complicating healthcare decision making. This review focuses on the roles of patients and healthcare professionals (HCPs) in shared decision making (SDM) in this new era of hemophilia care.
Key roles of the patient and HCP revolve around medical decision making. The respective decision-making roles of patients and HCPs have evolved as the practice of modern medicine has developed over the past century. Over the past 20 years, there has been a shift in medical decision making, from a clinician-based biomedical model, where physicians make decisions on behalf of the patient, to a patient-centered biopsychosocial model. 4 Patient- and family-centered care may be facilitated through SDM, an intervention in which the patient and HCP collaborate in making healthcare decisions based on the best available data about treatment benefits and side effects, while ensuring that the patient’s values, preferences and goals are integrated into this process. 5
SDM depends upon the clinician’s ability to fully inform the patient, while ensuring the patient’s active involvement in managing their illness, and maintaining a personal patient–provider interaction. This approach reduces the asymmetry in information exchange and redistributes the decision-making power equally between the provider and the patient. 6 Balance is achieved by providing patients with the capacity to act independently, while ensuring that they are not abandoned and receive the needed support throughout this complex process. 5 SDM is recognized as a strategic process in the management of chronic disease 7 and has been implemented in multiple chronic disease management programs worldwide, 8 including comprehensive hemophilia care. 9 Studies have shown that patients want to actively participate in their care 10 and are interested in learning about treatment options. 7 By stimulating productive patient–provider interactions, SDM has resulted in improved treatment adherence, 11 patient satisfaction, 12 and higher overall longitudinal healthcare quality. 8
Implementation of shared decision making
Even though SDM, as a conceptual framework, constitutes an essential part of patient- and family-centered care, 13 its practical implementation in day-to-day practice is challenging. Barriers to implementation include providers’ gap in knowledge about the process of SDM. In order to effectively implement SDM, providers must understand what the process entails, that it can be efficiently incorporated into clinical care, and that patients are truly interested and able to be active participants in their healthcare. To overcome these challenges, there are several proposed models that aim to simplify the implementation of SDM.
The ‘three-talk model’ was initially proposed by Elwyn and colleagues in 2012 and was further refined after incorporating feedback from relevant stakeholders and clinicians from different specialties. 6 It consists of three simple conversational steps: (a) team talk, where the provider introduces the concept of choice and why it is important to talk about choices; (b) option talk, where the provider describes the different options available in a structured and understandable fashion, with emphasis on the similarities and differences between options; and (c) decision talk, where the provider helps the patient explore preferences, determine goals and make decisions through deliberation and collaboration. The overall goal of this model is making a decision based on informed preferences (Figure 1).

Three-talk model of shared decision making.
The ‘deliberation dialogue model’ proposed by Lamiani and colleagues emphasizes finding common ground through the exploration of the patient’s illness experience and utilizes the patient’s interpretations, feelings, preferences, values and social context to reach a collective goal of care. 14 This model consists of three stages: (a) opening stage, where either participant brings up a topic describing it as a problem that needs to be solved; (b) argumentation stage, where participants put forward solutions, with arguments for or against their proposed solutions; and (c) closing stage, where the patient and HCP agree on a course of action as the final outcome of the discussion. Lamiani and colleagues found that 80% of consultations in hemophilia contained a deliberation dialogue (Figure 2). However, when the authors analyzed the interactions between patients and their providers to assess the completeness of the dialog, 49% of deliberations were incomplete due to completely or partially lacking an argumentation or a closing stage.

The structure of deliberation dialogues.
The SHARE approach is a five-step process for SDM developed by the Agency for Healthcare Research and Quality (AHRQ).
15
It consists of five steps: (a)
In implementing SDM, there are several additional important components that ensure its success. The presence of an established trusting relationship between the patients and their provider is essential and should not be compromised. When appropriate, the HCP should also utilize community resources, including patient volunteers or support groups, who are willing to share their experiences as real-life examples. In addition, improving patient–provider communication must be accomplished through appropriate provision of information, which allows the patient to be self-efficacious (e.g. able to cope with their illness). The capability of a provider to communicate effectively requires formal training and has been positively associated with patients’ treatment adherence. 11 Communication training should become an integral part of early medical training, as outlined by Dr Calvin Chou at the University of California, San Francisco’s Academy of Communication in Healthcare. 16 In combination with the HCP’s attentive listening, effective communication results in the patient having a sense of self-efficacy, which leads to patient proactivity, as they start to feel comfortable to request further information, share uncertainties, and become more confident in their ability to take part in determining and completing their treatment plans.
Furthermore, the conversation must comply with concepts of closed-loop communication, which includes the presence of a conclusion statement summarizing the decision and future steps. SDM must also be adapted to the patients’ developmental stage (i.e. childhood, adolescence, adulthood), plus race, ethnicity and culture, in order to provide culturally competent care. 17 Finally, regardless of which model is applied, an essential component of SDM is the presence of appropriately designed decision support tools. These tools summarize available evidence to guide the clinician during the discussion and serve as an educational resource throughout the decision-making process.
Shared decision making in current hemophilia care
SDM in hemophilia care first originated in the aftermath of the treatment-related human immunodeficiency virus (HIV) epidemic that devastated the hemophilia community in the 1980s and 1990s. 18 Overall, factors which influence medical decision making in hemophilia care include proven and perceived risks, particularly infection and inhibitor development; proven and perceived benefits, particularly reduction in bleeding and improvement in mobility; prior patient experience; family history; patient preference regarding clotting factor mixing and administration; physician recommendation; treatment availability; and insurance coverage.19–25
Scalone and colleagues evaluated patients’, pharmacists’ and physicians’ preferences in choosing a treatment regimen for patients with inhibitors. 26 This study highlighted different preferences among providers and patients and importance of acknowledging patient views to optimize treatment outcomes. Nilson and colleagues examined factors influencing treatment in patients with mild hemophilia and identified significant disconnect between patients and providers in regards to decision making. This disconnect was attributed to ineffective communication and resulted in suboptimal patient knowledge about their disease and options for treatment. 27 These concepts continue to be relevant as the hemophilia community identifies the optimal approach to incorporate novel hemophilia treatments in practice.
SDM is particularly important in regard to prophylaxis, which requires a commitment to regular treatment to prevent bleeding. Lane and colleagues qualitatively examined factors influencing the treatment decision making of young men with severe hemophilia, particularly related to use of prophylaxis, and concluded that identifying a common language to discuss treatment options is critical. 28 Gringeri and colleagues have advocated for personalized plans of care for patient-centric prophylaxis that incorporates patient-provided information about lifestyle, physical activity and risk taking, support network and financial resources; they advocate for an adaptive plan driven by individual patient outcomes and self-management. 29 The British Columbia Hemophilia Adult Team developed a strategy for optimizing prophylaxis regimens aligned with patient priorities using a standardized multidisciplinary approach involving nurses, social workers, psychologists and other HCPs, 30 and in a 12-month cohort study of 18 patients, patients had decreased annual bleed rate and improve quality of life. 9 The inclusion of social workers and psychologists as part of the team is particularly important for engaging adolescents and young adults with hemophilia who may not recognize the importance of self-care and prioritizing health (Figure 3).

Standardized approach to individualized treatment plan.
In order to facilitate SDM in the clinical care of hemophilia, Athale and colleagues developed two-sided decision tools (see http://www.decisionbox.ulaval.ca). 19 The tools include decision boxes for physicians and patient decision aids. The framework for these tools is shown in Table 2.
Framework for two-sided decision tools.

These hemophilia-specific tools focus on three key areas: choosing factor replacement product when beginning treatment in a previously untreated patient (PUP); choosing when to start prophylaxis and with what dose; and choosing when to start immune tolerance induction (ITI) and with what dose. The patient decision aids provide information about what the intervention is, who it is for, different options, and what is involved. The decision aids also include a matrix of risks and benefits to allow the patient to properly weigh these factors, and a list of goals specific to the intervention for the patient to rank. The physician decision box includes more detailed information on the studies and level of evidence to support each treatment option and key issues that may influence patient preferences. These decision aids will need to be updated to incorporate new treatment options since they do not include information on EHL clotting products, nonfactor products or gene therapy.
Shared decision making in the era of extended half-life factor products
EHL products are recombinant clotting factor products that are modified through strategies including Fc-fusion and pegylation. 31 These products have 1.4–1.7-fold and 3–5-fold higher half-life than standard half-life (SHL) factor VIII and IX products, respectively, with similar efficacy and safety.32–34 There have been no significant adverse events in any of the studies involving EHL, including no cases of inhibitor development in previously treated patients.
EHL have the potential to decrease the frequency of infusions in the treatment of hemophilia. This has been most evident in factor IX EHL products, allowing patients to infuse once every 7–14 days, while achieving higher trough levels when compared with traditional factor products. For patients with hemophilia A, EHL products may be given every 3–5 days, depending on their individual pharmacokinetics (PK). Patients with a more hectic and unpredictable lifestyle may value the lower infusion frequency, such as illustrated in our case. In addition, patients with limited peripheral access and those transitioning from central venous access to peripheral infusions would be good candidates for EHL products. Overall, decreasing infusion frequency may result in improved prophylaxis adherence among patients and fewer patients who discontinue prophylaxis. 35 Furthermore, improved PK profile of these products may allow patients to achieve higher trough levels if administered using the same schedule as SHL products. This option may be optimal for patients with target joints, who require higher factor levels to prevent breakthrough bleeding or those with an intense activity level that require higher factor peaks or troughs. 36 In the latter case, this benefit must be balanced with higher cost of therapy. 37
Due to the variability in individual PK, PK testing of SHL and EHL products can inform decisions about switching. Prior study shows that patients should be involved in the decision about implementation of PK testing. 38 Physicians may utilize services such as WAPPS-Hemo (https://www.wapps-hemo.org/) for PK profile calculations. PK testing results may then be shared with patients to facilitate decision making regarding whether or not to switch from a SHL to an EHL product.
Even though there is a large body of efficacy and safety data for EHL products, a recent survey highlighted the gap in delivery of this information to patients. The authors describe patient expectations of the EHL products compared with SHL products (i.e. half-life extension, improved efficacy, and similar side-effect profile), with 60% of patients willing to switch to these products if these criteria are met. 39 However, 70–80% of patients reported they did not receive sufficient information to make such a decision, emphasizing the lack of communication between providers and patients that can be addressed through SDM.
When discussing switching between products, the conversation should include patient-specific aspects (i.e. age, adherence and activity characteristics), disease-specific aspects (i.e. bleeding phenotype and joint status), factor-specific aspects (PK profile and frequency of infusion), and cost effectiveness. As real-work experiences of patients and providers mature, this information can also be shared with the hemophilia community.
Shared decision making in the era of nonfactor products
Emicizumab-kxwh, a humanized monoclonal antibody which binds factors IX and X to generate thrombin in the absence of factor VIII, was approved by the US Food and Drug Administration in 2017 for prevention of bleeding in patients with congenital hemophilia A and inhibitors. 40 Emicizumab-kxwh has demonstrated superior efficacy when compared with bypassing agents and once weekly subcutaneous administration significantly decreases burden compared with intravenous clotting factor for prophylaxis or ITI. When providing information about this therapy, HCPs must also discuss the potential risk of venous thrombosis and microangiopathic hemolytic anemia when used in combination with activated prothrombin complex concentrates at high doses (>100 IU/kg/day) for more than 24 h in the treatment of breakthrough bleeds and how to mitigate this risk by changing the treatment protocol for breakthrough bleeding. Even though further events have been avoided by implementing strict guidelines on treatment of breakthrough bleeding, these events highlight the importance of discussing safety concerns as part of SDM when implementing novel therapies.
Indications for nonfactor replacement products are expected to expand to patients without inhibitors as studies of emicizumab-kxwh in patients without inhibitors are completed and to patients with hemophilia B as additional nonfactor replacement products. 3 When discussing nonfactor products with patients under the umbrella of SDM, HCPs must be well informed and should explore the patient’s degree of risk tolerance, which usually depends on extent of disease burden and presence of treatment options. Even if risk tolerance is high, an HCP must provide balanced information about the associated risks and benefits without influencing the patient’s decision. Furthermore, obtaining more data on the safety of these products will allow patients with low risk tolerance, such as patients who are bleed free on clotting factor prophylaxis, to make an informed decision with more accurately calculated risk–benefit ratio.
Shared decision making in the era of gene therapy
As a single gene disorder, hemophilia is an optimal disease for implementation of gene therapy. The most promising form of gene therapy to date is in vivo gene transfer therapy, which uses a nonpathogenic viral vector to transduce the target gene into hepatocytes. There are an increasing number of gene therapy trials for adult patients with congenital hemophilia A and B without inhibitors, and more than 50 patients have already been enrolled.2,41 Phase I–II trials have demonstrated clinically significant efficacy, with the majority of patients stopping prophylactic treatment with minimal-to-no-breakthrough bleeding.42,43 Overall, patients treated with gene therapy have a substantial decrease in disease and treatment burden which should translate to improvement in quality of life and decreased lifetime healthcare costs. The optimal strategy which achieves highest sustained factor level is yet to be determined. The primary adverse events have been transaminitis treated with steroids. Other potential risks associated with gene transfer therapy include: (a) risk of gene integration into the DNA, which may occur but is considered to be rare as a result of the episomal nature of the transduced gene; (b) risk of inhibitor formation; (3) risk of thrombotic event if the sustained factor activity is above the normal range; and (4) potential for gene toxicity, which needs long-term follow up for it to be detected. 44
As phase III trials are completed and gene therapy is commercialized, the therapy will be more readily available to adults and, possibly, children. As HCPs prepare for this game changing therapy, certain unique practical topics need to be discussed. First, a clear definition of the therapeutic indications and target trough level is needed. Eligibility and treatment goals should be directed through a personalized risk–benefit analysis involving a patient’s input. Considering a patient’s clinical course, current treatment outcomes and treatment goals are particularly important in patients with mild and moderate hemophilia and in cases where other new therapies have already decreased the burden of disease (e.g. patients with hemophilia B on EHL factor IX products). Second, as pharmaceutical companies are deciding on the optimal financial approach to providing this novel therapy, the economic benefit of having such a curative treatment may be balanced by the possibility of a continuous long-term annual cost approach. If this is the case, some insurance companies may decide not to cover the cost of gene therapy, thus limiting its availability to a select group of patients. Third, many of the potential risks outlined may take many years to manifest and are not quantifiable with the current data. These risks may be more substantial, as this therapy is offered to young children with hemophilia. Since these unclear risks may have serious life-altering consequences on patients, the risk–benefit analysis at the core of SDM may be difficult to outline and this limitation should be frankly discussed by the HCP.
Shared decision making in developing countries
Standard prophylaxis regimens used in developed countries are cost prohibitive in developing countries, which have limited resources for diagnosis and management.45,46 Therefore, allocation of resources and goals of treatment are different than in developed countries. Currently the approach to prophylaxis in patients living in developing countries consists of low-dose factor replacement regimens (i.e. 5–10 IU/kg two to three times per week), with individualized modifications to achieve improved quality of life rather than a specific bleed rate.47–50 The availability of new therapies will be variable throughout the world, based on governmental drug approval, medical infrastructure, HCP expertise, and cost. As the World Federation of Hemophilia (WFH) works to alleviate treatment disparities through humanitarian aid (see https://www.wfh.org/en/humanitarian-aid-program) perhaps the novel therapeutics will be integrated into care even in resource-limited countries.
As the WFH works to alleviate treatment disparities in the developing world, the novel therapeutics will offer more effective tools that can be practically integrated into care in resource-limited countries. If available, nonfactor replacement products will provide patients with less frequent, subcutaneously administered treatments that can be given by local providers and will eliminate the need for intravenous access. However, gene therapy, as a one-time curative treatment option, may be more feasible in resource-poor countries than replacement therapeutics. 51 Ultimately, with appropriate diagnostic capabilities resulting in early recognition, these therapies may be given prior to onset of complications, thus limiting the resultant disabilities commonly seen in developing countries. 52
For any therapy, culturally adapted SDM may be implemented in developing countries to facilitate discussions about medical and surgical treatment. Both the burden of the treatment regimen and its expected goals should be discussed to allow the patient to make an appropriate decision, tailored to their resources and life situation.
Summary of patient and HCP roles in SDM
As the complexity of treatments options increases, the roles of the patients and HCPs in SDM are critical. The patient needs to educate oneself about the treatment options including risks and benefits, availability, and cost. This can be done proactively outside of medical visits and at scheduled visits. The patient must consider which risks and benefits really matter and decide what the goals of care are (e.g. life without bleeding, more active lifestyle, fewer worries about bleeding). The patient must decide if they are willing to participate in a clinical trial in order to access a new therapeutic agent. The patient must identify potential barriers to adhering to the therapy including cost, training to administer, time conflicts for follow up, etc. The patient must have open communication with the HCP about their preferences and take an active role in self-management of hemophilia. Finally, the patient should be accountable for their treatment. Mobile health applications may be used to track bleeding and bleed management, as well as adherence with prophylaxis with the goal of optimizing the effect of the prescribed treatment.
The HCP must educate oneself about the indications for treatment and available evidence regarding safety, efficacy, dosing, monitoring strategies and insurance coverage. This can be done through review of the medical literature, attending conferences, attending online webinars and through discussion with experts in the field. For EHL products, HCPs should discuss the utility of PK testing with patients, particularly those on prophylaxis. 38 The HCP must be able to communicate this information to the patient, using available decision aids and taking into consideration the patient’s developmental stage, health literacy and cultural background; listen to the patient’s preferences and goals, consider barriers to implementation, and then collaborate with the patient to design a treatment plan that aligns with the patient’s clinical status, and personal preferences and goals of care. This may require initial or additional training in SDM 53 and cultural competency. In this collaborative process, the HCP should be mindful not to appear the ‘physician as expert’ paradigm but to understand the gap between patient and provider goals 54 and focus on a customized plan that aligns with patient priorities. 30 Moreover, the HCP should work with the patient to identify and track patient-centered outcomes of importance, such as decreased pain, increased physical activity, or decrease in missed work or school.
Conclusion
Decision making in hemophilia is complicated when there are multiple effective therapies without a clear-cut treatment of choice. SDM and decision-aid tools should be incorporated into the comprehensive care model to optimize patient outcomes as defined by the patient. Together, patients and the HCPs can advocate for the hemophilia community as a whole to ensure ongoing development of novel therapies, and access to treatment and comprehensive care.
Footnotes
Acknowledgements
Dr Fadi Nossair received a 2017 HTRS/Novo Nordisk Clinical Fellowship Award in Hemophilia and Rare Bleeding Disorders from the Hemostasis and Thrombosis Research Society (HTRS), which was supported by Novo Nordisk Inc.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
