Abstract
Objectives
To examine the efficacy of deferasirox (DFX) by comparison with deferoxamine (DFO) in managing iron overload in patients with sickle cell anaemia (SCA).
Methods
Online databases were systematically searched for studies published from January 2007 to July 2022 that had investigated the efficacy of DFX compared with DFO in managing iron overload in patients with SCA.
Results
Of the 316 articles identified, three randomized clinical trials met the inclusion criteria. Meta-analysis of liver tissue iron concentration (LIC) showed that iron overload was not significantly higher in the DFX group compared with DFO group (WMD, −1.61 mg Fe/g dw (95% CI −4.42 to 1.21). However, iron overload as measured by serum ferritin was significantly lower in DFO compared with DFX group (WMD, 278.13 µg/l (95% CI 36.69 to 519.57). Although meta-analysis was not performed on myocardial iron concentration due to incomplete data, the original report found no significant difference between DFX and DFO.
Conclusion
While limited by the number of studies included in this meta-analysis, overall, the results tend to show that DFX was as effective as DFO in managing iron overload in patients with SCA.
Keywords
Introduction
Sickle Cell Anaemia (SCA) is the most common monogenic inheritable blood disorder and can result in multiple life-threatening complications such as end-organ damage, kidney disease, increased stroke risk, increased susceptibility to infections and pulmonary problems.1–3 The most common type of sickle cell disease is the result of the inheritance of two alleles (haemoglobin [Hb] SS) which causes the production of an abnormal form of beta (β)-globulin.4,5 Blood transfusion is one of the key practices in the management of patients with SCA. Despite its value, blood transfusion is also associated with iron overload which is a cause of significant morbidity in these patients.6,7
Over the past 40 years, deferoxamine (DFO) has been the treatment of choice for iron overload, 8 and its efficacy is well established in patients with sickle cell disease.9–11 However, the need for overnight infusion and issues such as infection at the injection site, have been identified as limitations that can lead to low compliance. Deferasirox (DFX) is an orally absorbed iron chelator that has been shown to be effective in reducing iron overload in patients with SCA.4,12 While previous systematic reviews have compared the efficacy and safety of DFX with DFO, the chelation regimens have focused on patients with thalassemia.13–16 An area that requires additional research is the comparative effectiveness of DFX and DFO in managing iron overload in patients with SCA.
Therefore, we conducted a systematic review and meta-analysis according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidance 17 to compare the iron overload reduction capacity of DFX relative to DFO in patients with SCA.
Methods
The electronic databases and libraries of PubMed Central (PMC), Google Scholar, Embase, Medline, and Cochrane Library were used to search and extract articles that were published from January 2007 to July 2022 that had compared the efficacy of DFX with DFO in reducing iron overload in patients with SCA. Emphasis was put on Random Control Trials (RCTs), cross-sectional research, and experimental research. Articles that could not be retrieved from the databases were extracted from other online libraries and archives using the title name or DOI numbers. MeSH-related English words such as “sickle cell anaemia”, “iron overload”, “deferasirox”, and “deferoxamine” were used in the search. In addition to the database search, we also reviewed reference lists of retrieved studies to identify other studies that met the inclusion criteria (i.e., ‘snowballing approach’).
For a published report to be included in the meta-analysis, it had to have measured iron concentration in liver tissues, serum ferritin, and/or myocardial iron concentration. Studies that focused on outcomes other than iron overload and those that focused on patients with thalassemia were excluded from the analysis. Only English-language publications were included.
Two independent reviewers extracted the data from the identified reports after first performing a risk of bias analysis as recommended by the Cochrane Handbook Version 5.1.0. 18 The following items were extracted from the articles: first author; study type; publication year; efficacy outcomes; subgroup analysis. The two reviewers selected the articles and a third researcher resolved any differences of opinion. The study was registered on the PROSPERO online system (ID CRD42022350535).
Statistical Analyses
The meta-analysis was performed using Review Manager software version 5.4 (RevMan 5.4). The data were pooled, and intervention efficacy was performed for the DFX and DFO groups. Mean difference, standard deviation and 95% confidence intervals (CI) were used to describe the data. The level of evidence quality of each study was estimated according to the guidelines of Grading of Recommendations, Assessment, Development, and Evaluation (GRADE). 18
The I2 statistic was used to assess statistical heterogeneity and the effect of pooling each study. If I2 statistic ≥50% and P < 0.05, a random effects model was applied to the data. If no heterogeneity was observed, a fixed effect model was to be used. Forest plots were used to summarise the pooled studies. A P-value <0.05 was considered to indicate statistical significance.
Results
Of the 316 articles identified, 167 were excluded due to duplication (Figure 1). A further 68 articles were eliminated for being non-English or animal studies, 24 were excluded due to being non-RCTs, non-relevance to SCA, lack of intervention measures, and failing to meet other inclusion criteria and for seven studies the data were not retrievable. Therefore, three studies were included in the systematic review (Table 1).19–21 All included studies were RCTs, multicentre and were published between 2007 and 2020. The number of participants in each included study varied from 195 to 393.

Flow diagram of study selection.
Characteristics of the three studies included in the meta-analysis.
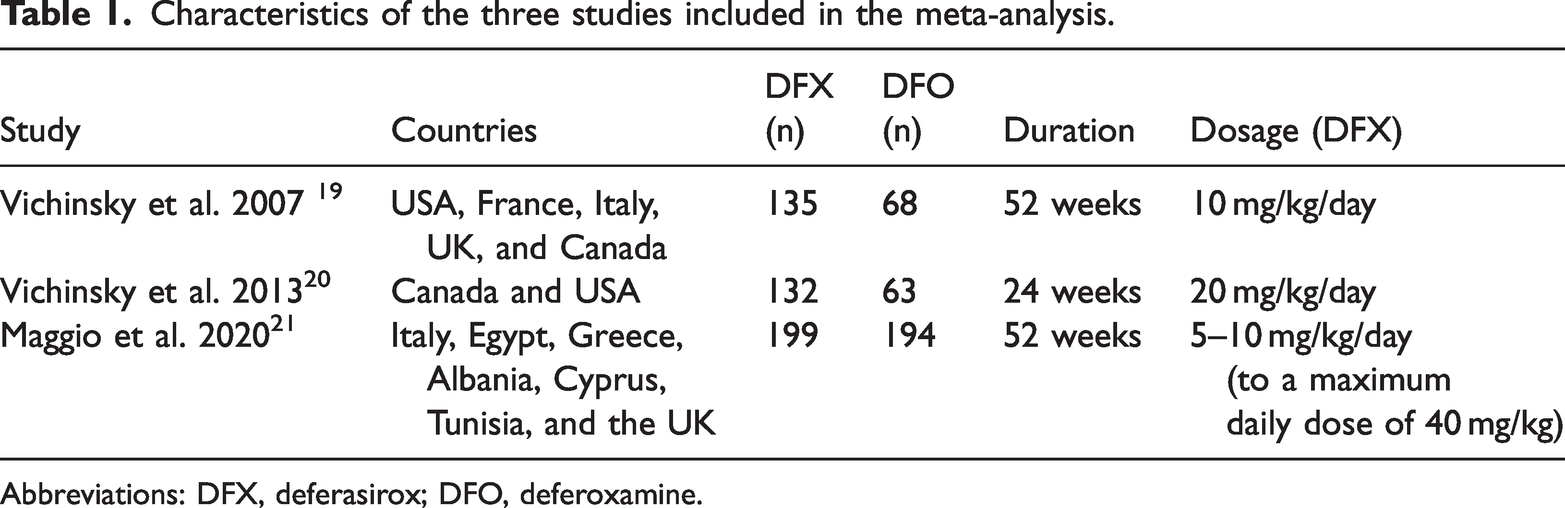
Abbreviations: DFX, deferasirox; DFO, deferoxamine.
The RCTs were assessed to determine their value in the systematic review and meta-analysis. Risk of bias was assessed according to seven categories outlined in assessment of risk of bias 18 and according to the risk of bias table (Figure 2) methodological quality of the included studies was moderate
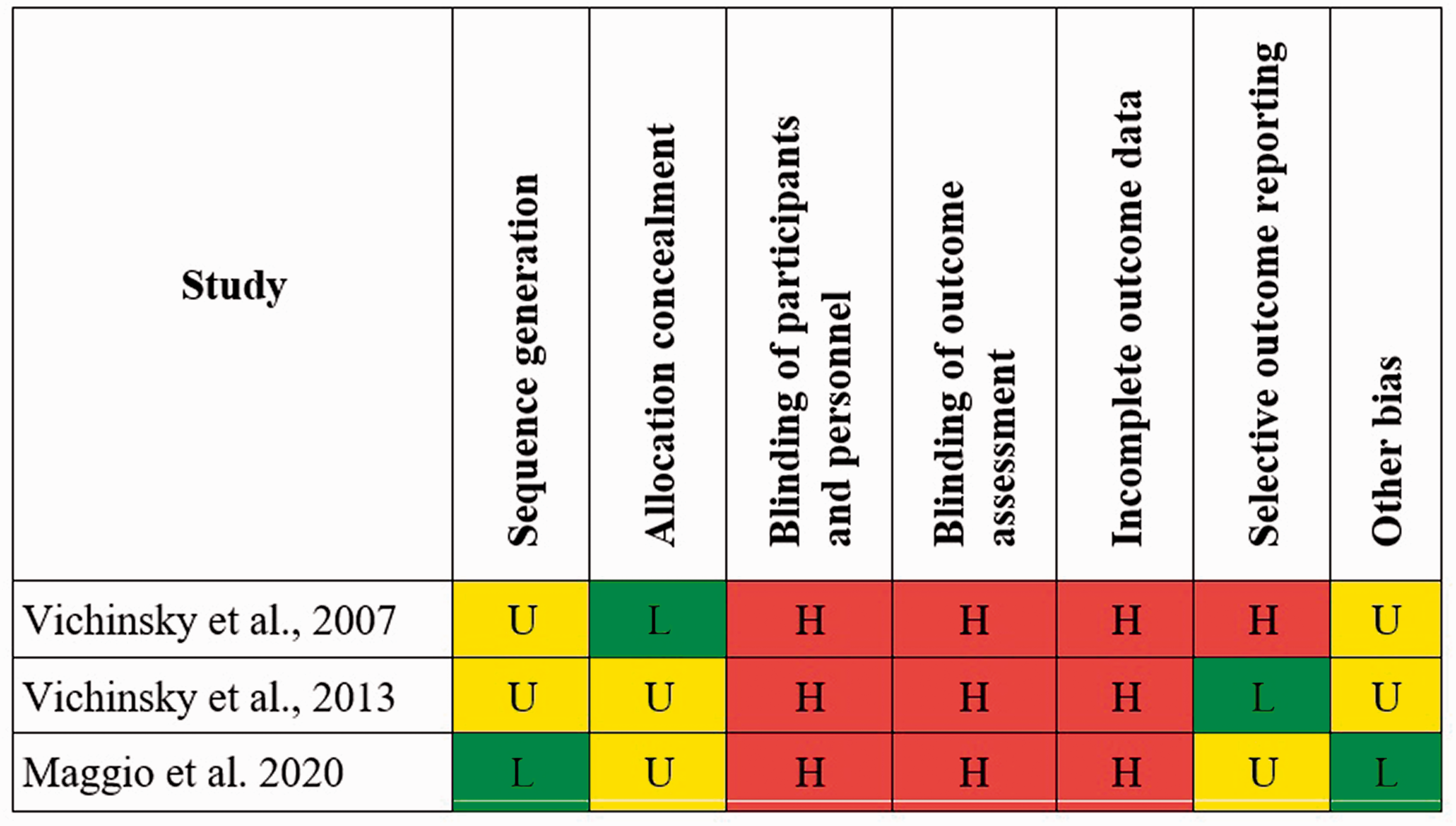
Risk of Bias (RoB) graph as recommended by the Cochrane Handbook Version 5.1.0. 18
Iron overload was measured as serum ferritin in all three studies,19–21 liver tissue iron concentration in two studies19,21 and myocardial iron concentration in one study (Table 2).21 Other outcomes that were assessed included, adverse events, compliance and pharmacokinetic evaluations but are not included in this meta-analysis. Period of follow-up ranged from 24 to 52 weeks. Based on iron concentration outcomes, there were no large or unexplained findings indicating that the inconsistency quality of the GRADE approach was rated up. 22
Iron concentration outcome in the three studies included in the meta-analysis.

Abbreviations: DFX, deferasirox; DFO, deferoxamine; LIC, liver iron concentration; IC, iron concentration; SD, standard deviation.
Of the two studies that assessed liver tissue iron concentration only one study was included in the analysis 19 because of missing SD data in the other study. Overall, iron overload was not significantly greater in the DFX group compared to the DFO group (WMD, −1.61 mg Fe/g dw (95% CI −4.42 to 1.21 mg Fe/g dw; P = 0.26) (Figure 3). Heterogeneity was significant (I2 = 72%, P = 0.03). Further inspection of the data showed that liver iron concentration showed no statistical difference for the overall group of patients, weighted mean difference (WMD) −0.20 mg Fe/g dw (95% CI −2.98 to 2.58 mg Fe/g dw) nor for the sub-group of patients receiving simple transfusions (WMD, −0.20 mg Fe/g dw (95% CI −1.97 to 1.57 mg Fe/g dw). However, for the sub-group of patients receiving exchange transfusions, the DFO-treated patients showed a higher, statistically significant reduction in liver tissue iron concentration (WMD −5.2 mg Fe/g dw (95% CI −8.56 to −1.84 mg Fe/g dw).

Forest plot – Weighted mean difference of liver iron concentration (LIC) values in the deferasirox (DFX) and deferoxamine (DFO) groups.
Of the three studies that assessed serum ferritin reduction, only two were included in the statistical pooling,19,20 because of incomplete data in the other study. Overall, the iron overload of the participants in the DFO group, as measured by serum ferritin, was significantly lower than those in the DFX group (WMD, 278.13 µg/l (95% CI 36.69 to 519.57 µg/l; P = 0.02) (Figure 4). No heterogeneity was observed (I2 = 0%, P = 0.02).

Forest plot – Weighted mean difference of iron overload (serum ferritin) values in the deferasirox (DFX) and deferoxamine (DFO) groups.
Myocardial iron concentration was only recorded in one study. 22 Meta-analysis was not performed on this measure because of insufficient data for our analysis but the authors had reported no significant difference between DFX and DFO in myocardial iron.
Discussion
Regularly transfused patients with SCA are exposed to transfusion-related iron overload. DFO has been the treatment of choice for iron overload for the past 40 years. 8 However, compliance concerns and adverse complications associated with DFO have increased interest in alternative therapies. Orally administered DFX offers an opportunity to improve outcomes for patients through improved adherence.23,24 Previous systematic reviews have evaluated DFX by comparison with DFO in their capacity to reduce iron overload but have focused on patients with thalassemia13–16 and so there is a need to evaluate these iron chelating agents in patients with SCA.
Although limited by the number of studies included in the review, the results of this present meta-analysis showed that liver iron concentration showed no difference between DFX and DFO. A significant difference in serum ferritin was observed between groups in favour of DFO, and although meta-analysis was not performed on myocardial iron concentration due to incomplete data, the original report found no significant difference between DFX and DFO. Therefore, overall, the results tend to show that DFX was as effective as DFO in managing iron overload in patients with SCA. These results are in agreement with previous findings in thalassemia studies that showed noninferiority of DFX compared with DFO.25,26
The current study had several limitations. For example, only three studies were included in the meta-analysis, thus reducing the pooling effect. However, large numbers of patients were involved in these multicentre studies. In addition, the meta-analysis focused on efficacy measures (iron overload) and so data on long-term safety of these iron chelating agents needs to be evaluated. Furthermore, the three studies were heterogenous in their different dosing regimens and lengths of assessment periods.
Chronic iron overload is a complication that affects patients with SCA and other haemoglobinopathies. Additional large, multicentre, investigator sponsored studies that focus on DFX by comparison with DFO and other iron chelators are necessary for the understanding of the efficacy and safety of DFX in people with SCA. In addition, studies that examine different dosing schedules and/or in sickle cell disease as a whole, are also required.
Research Data
Research Data for Deferasirox versus deferoxamine in managing iron overload in patients with Sickle Cell Anaemia: a systematic review and meta-analysis
Research Data for Deferasirox versus deferoxamine in managing iron overload in patients with Sickle Cell Anaemia: a systematic review and meta-analysis by Talal Qadah in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
