Abstract
Diffuse large B-cell lymphoma (DLBCL) represents the most common type of malignant lymphoma. In the last few years, significant progress has been achieved in the understanding of the molecular pathogenesis of this entity. Gene expression profiling has identified three molecular DLBCL subtypes, termed germinal-center B-cell-like (GCB) DLBCL, activated B-cell-like (ABC) DLBCL, and primary mediastinal B-cell lymphoma (PMBL). In this review, we summarize our current understanding of the biology of these DLBCL subtypes with a special emphasis on novel diagnostic and therapeutic approaches.
Introduction
Diffuse large B-cell lymphoma (DLBCL), the most common subtype of malignant lymphoma, is characterized by heterogeneity with respect to clinical presentation, morphology, and molecular pathogenesis [Coiffier, 2001]. The introduction of gene expression profiling revealed the existence of at least three distinct molecular subtypes of DLBCL that differ in the expression of thousands of genes and that seem to arise from B cells at different stages of differentiation [Rosenwald et al. 2003, 2002; Savage et al. 2003; Wright et al. 2003; Alizadeh et al. 2000]. According to their gene expression profiles these subtypes were termed germinal-center B-cell-like (GCB) DLBCL, activated B-cell-like (ABC) DLBCL, and primary mediastinal B-cell lymphoma (PMBL). These subtypes do not only differ with respect to their gene expression profiles, but they depend on different oncogenic pathways and are characterized by significant differences in overall survival following standard treatment [Lenz et al. 2008b, 2008c; Monti et al. 2005; Rosenwald et al. 2002]. Whereas patients with GCB DLBCL respond favorably to a combined approach of the anti-CD20 antibody rituximab and chemotherapy, more than 50% of ABC DLBCL patients will succumb to their disease [Lenz et al. 2008b]. This implies that novel therapeutic strategies are critically warranted to improve prognosis of affected patients. However, this can only be achieved by a better understanding of the underlying molecular mechanisms that drive lymphomagenesis.
Molecular pathogenesis of GCB DLBCL
GCB DLBCLs seem to originate from germinal-center B-cells (Figure 1) [Rosenwald et al. 2002; Alizadeh et al. 2000]. Accordingly, these lymphomas characteristically express many genes such as

Germinal-center B-cell-like (GCB) diffuse large B-cell lymphoma (DLBCL) derived from normal germinal-center centroblasts. BCL2 is deregulated by t(14;18) translocations.
Various oncogenic pathways are deregulated in GCB DLBCL that contribute to the molecular pathogenesis of this entity (Figure 1). In approximately 45% of GCB DLBCL patient samples, a t(14;18) translocation juxtaposing the
Another hallmark of GCB DLBCL biology is the deregulation of the phosphatase and tensin homologue (PTEN)– phosphatidylinositol 3-kinase (PI3K) signaling pathway (Figure 1) [Lenz et al. 2008c]. The PI3K signaling cascade is initiated with the phosphorylation of phosphatidylinositol 4,5-bisphosphate (PIP2) to phosphatidylinositol 3,4,5-trisphosphate (PIP3), resulting in cellular processes such as proliferation, cell survival, and cell growth [Chalhoub and Baker, 2009; Salmena et al. 2008]. The conversion of PIP2 into PIP3 is tightly regulated by the opposing activities of the lipid phosphatase PTEN and class I PI3K family members. Whereas the PI3Ks catalyze the conversion from PIP2 to PIP3, PTEN hydrolyzes the 3-phosphate to generate PIP2 [Salmena et al. 2008]. Upon PTEN loss, PIP3 accumulates and AKT and mTOR are activated, further promoting cell survival, proliferation, and growth [Salmena et al. 2008]. Roughly 15% of GCB DLBCLs harbor heterozygous or homozygous deletions of
GCB DLBCLs are furthermore characterized by gain or amplification of
Molecular pathogenesis of ABC DLBCL
The gene expression profiles of ABC DLBCL patient samples suggest that this subtype is derived from B cells that are in the process of differentiating into plasma cells (Figure 2) [Rosenwald et al. 2002]. In contrast to GCB DLBCL, most genes characteristically expressed by normal germinal-center B-cells are downregulated in ABC DLBCL. However, these samples exhibit an upregulation of many genes normally expressed in plasma cells [Wright et al. 2003]. Specifically, ABC DLBCLs express XBP-1, a key regulator of the secretory phenotype of plasma cells [Shaffer et al. 2004; Wright et al. 2003]. However, full plasmacytic differentiation is blocked by different genetic abnormalities (Figure 2). Approximately 25% of ABC DLBCL patient samples harbor inactivating mutations of

Activated B-cell-like (ABC) diffuse large B-cell lymphomas (DLBCLs) are derived from plasmablasts that are characterized by a block in differentiation. ABC DLBCLs show constitutive activation of the oncogenic nuclear factor kappa B (NF-κB) signaling pathway as well as deregulation of the anti-apoptotic BCL2 protein. The molecular mechanisms by which NF-κB is activated will determine responsiveness to specific inhibitors.
ABC DLBCL is characterized by additional genetic abnormalities that play an important role in its pathogenesis (Figure 2). The
Another pathogenetic feature of ABC DLBCLs is the constitutive activation of the NF-κB signaling pathway that promotes cell survival and proliferation and inhibits apoptosis (Figure 2) [Davis et al. 2001]. NF-κB is a family of inducible transcription factors consisting of five members, RelA (p65), RelB, c-Rel, NF-κB1 (p50 and its precursor p105), and NF-κB2 (p52 and its precursor p100) [Jost and Ruland, 2007]. These proteins form different homodimers and heterodimers that are kept inactive by binding to inhibitory proteins of the IκB family. Activation of NF-κB through various stimuli leads to phosphorylation and proteosomal degradation of the IκB proteins by the multiprotein IκB kinase (IKK) resulting in nuclear translocation of the NF-κB subunits and transactivation of their target genes [Jost and Ruland, 2007]. ABC DLBCLs are characterized by constitutive activation of the NF-κB pathway. Inhibition of this pathway using either a dominant active form of IκBα or a specific IKK inhibitor is toxic to ABC but not to GCB DLBCL cell lines indicating the importance of NF-κB signaling in ABC DLBCL biology [Lam et al. 2005; Davis et al. 2001].
Insights which upstream molecular mechanisms lead to the constitutive activity of NF-κB in ABC DLBCL were obtained by a small hairpin RNA (shRNA) screen that revealed toxicity of shRNAs directed against CARD11, BCL10, and MALT1 in ABC DLBCL cell lines [Ngo et al. 2006]. In normal B and T cells these three proteins form a transient signaling complex (the CBM complex) following stimulation that activates IKK [Rawlings et al. 2006]. In contrast, in ABC DLBCL the CBM complex is constitutively activated due to different mechanisms. Resequencing revealed that approximately 10% of ABC DLBCL patients harbor activating
Additional mechanisms for the constitutive activation of NF-κB have been reported. Approximately one third of ABC DLBCL patients carry biallelic inactivation of the negative NF-κB regulator A20 [Compagno et al. 2009; Kato et al. 2009]. Reintroduction of A20 into A20-deficient cell lines resulted in inhibition of NF-κB signaling and induced apoptosis and cell growth arrest [Compagno et al. 2009]. Alternatively, a recent study showed that activating mutations affecting the adaptor protein
Molecular pathogenesis of PMBL
PMBL has been identified as a distinct entity based on clinical and morphological features. PMBL normally affects young women with a median age of 30–35 years with the mediastinum being the predominant site of lymphoma manifestation. Morphologically PMBL tumors are frequently sclerotic and are characterized by diffuse proliferation of medium-sized to large B-cells with a heterogeneous morphology [van Besien et al. 2001]. However, PMBL cannot be reliably diagnosed solely by morphologic and clinical parameters. A recent gene expression profiling study showed that approximately one quarter of cases diagnosed as PMBL by conventional criteria did not exhibit the characteristic gene expression signature of PMBL and thus represented other molecular DLBCL subtypes with mediastinal involvement [Rosenwald et al. 2003]. Interestingly, PMBLs share many clinical and molecular features with nodular sclerosis classical Hodgkin lymphoma (cHL) [Rosenwald et al. 2003; Savage et al. 2003]. Similar to PMBL, nodular sclerosis cHL often affects young women with a mediastinal mass that is sclerotic in the histological examination. With respect to gene expression profiling, more than one third of the genes overexpressed in PMBL are also highly expressed in cHL cell lines [Rosenwald et al. 2003]. Furthermore, PMBL and nodular sclerosis cHL utilize the same oncogenic pathways such as NF-κB [Rosenwald et al. 2003; Savage et al. 2003]. However, despite these similarities PMBL and nodular sclerosis cHL can reliably be distinguished by histology and gene expression profiling.
PMBLs are characterized by gain or amplification of the chromosomal band 9p24 [Lenz et al. 2008c]. Roughly 50% of PMBL cases harbor this abnormality. One target gene of this amplicon is JAK2, a tyrosine kinase which regulates cytokine signaling through STAT transcription factors. Interestingly,
A recent shRNA library screen revealed that additional genes within the 9p24 amplicon might be involved in the biology of PMBL [Rui et al. 2010]. The histone demethylase JMJD2C seems to cooperate with JAK2 to modify the PMBL epigenome and thereby promotes proliferation and survival in PMBL cell lines [Rui et al. 2010]. Thus, inhibition of both JAK2 and JMJD2C might represent a novel therapeutic approach for PMBL. PD-L1 and PD-L2 that are ligands for the PD receptor on T-cells are also upregulated by this amplification in PMBL [Rosenwald et al. 2003]. Engagement of the PD receptor by its ligands inhibits signaling through the T-cell receptor, suggesting that amplification of these genes modulates the interaction between PMBL cells and the surrounding T cells [Rosenwald et al. 2003]. Finally, PMBLs are characterized by constitutive activation of the NF-κB signaling pathway [Rosenwald et al. 2003; Savage et al. 2003]. Inhibition of NF-κB using a specific IKK inhibitor was toxic to PMBL cell lines, indicating that constitutive activation of this pathway is important for PMBL biology [Lam et al. 2005]. The molecular mechanisms leading to this constitutive activation are poorly understood. In more than 30% of PMBL cases, the negative NF-κB regulator A20 is inactivated [Schmitz et al. 2009].
Clinical implications
The DLBCL subtypes are characterized by different overall survival rates following standard anthracycline-containing chemotherapy [Monti et al. 2005; Rosenwald et al. 2002]. GCB DLBCL and PMBL patients have relatively favorable 5-year overall survival rates. In contrast, the majority of ABC DLBCL patients will succumb to their disease. The introduction of the anti-CD20 antibody rituximab significantly improved the outcome for DLBCL patients [Coiffier et al. 2002]. This benefit is observed in both ABC and GCB DLBCL patients. However, GCB DLBCL patients still do significantly better compared with patients diagnosed with ABC DLBCL (Figure 3A) [Lenz et al. 2008b].
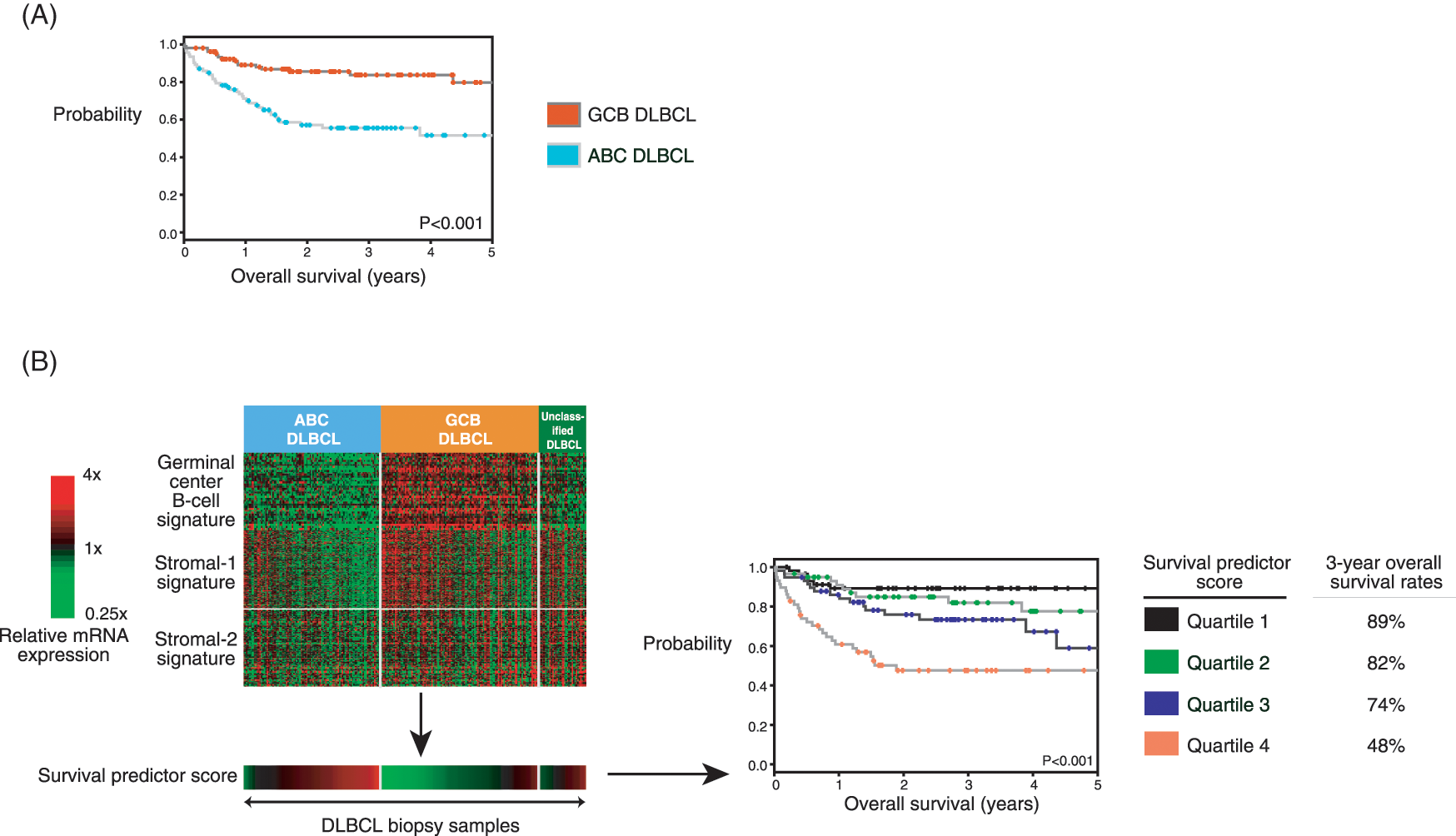
(A) Activated B-cell-like (ABC) and germinal-center B-cell-like (GCB) diffuse large B-cell lymphomas (DLBCLs) are characterized by significant differences in overall survival following a combination of rituximab and chemotherapy. (B) A multivariate gene expression-based survival model can be used to predict survival upfront at diagnosis in DLBCL patients treated with a combination of rituximab and chemotherapy. For every patient biopsy, a survival predictor score can be calculated. These scores are used to divide DLBCL patients into quartiles of significantly different 3-year overall survival rates of 89%, 82%, 74%, and 48%.
Gene expression profiling can provide additional information besides the distinction of ABC and GCB DLBCL. A multivariate gene expression-based survival predictor model that is independent of the International Prognostic Index (IPI) can be used to predict survival upfront at diagnosis in DLBCL patients treated with a combination of rituximab and anthracycline-containing chemotherapy [Lenz et al. 2008b]. Three gene expression signatures termed ‘germinal-center B-cell’, ‘stromal-1’ and ‘stromal-2’ are associated with survival (Figure 3B). The prognostically favorable ‘germinal-center B-cell’ signature parallels the distinction of ABC and GCB DLBCL and consists of genes characteristically expressed in germinal-center B-cells. In contrast, the ‘stromal-1’ and ‘stromal-2’ signatures reflect the composition of nonmalignant cells, i.e. the tumor microenvironment [Lenz et al. 2008b]. The favorable ‘stromal-1’ signature reflects extracellular matrix deposition and histiocytic infiltration. In contrast, the prognostically unfavorable ‘stromal-2’ signature reflects tumor blood-vessel density. A multivariate model for survival was created from these three gene expression signatures. For every patient biopsy, a survival predictor score was calculated. These scores were used to divide the patients into quartiles of 3-year overall survival rates of 89%, 82%, 74%, and 48% and 3-year progression-free survival rates of 84%, 69%, 61%, and 33% (Figure 3B) [Lenz et al. 2008b].
A different approach to utilize changes in gene expression to predict outcome in DLBCL patients was taken by Lossos and colleagues [Lossos et al. 2004]. A predictor of survival measuring only six genes (
In addition to its value in predicting patient outcome, gene expression profiling using DNA microarrays can additionally be applied to distinguish DLBCL from Burkitt’s lymphoma (BL). Two recent studies convincingly showed that BL is characterized by a unique gene expression profile [Dave et al. 2006; Hummel et al. 2006]. Intriguingly, in these analyses a substantial number of cases that were diagnosed as DLBCL by conventional methods showed the gene expression profile of BL, suggesting that they represent BL cases that are difficult to diagnose by current methods. This distinction however is of major clinical importance, as BL requires more intensive treatment strategies.
Future developments
Despite the recognition and acknowledgement of different molecular DLBCL subtypes more than a decade ago, the translation into clinical routine is only slowly progressing and microarray-based technologies are just emerging in the clinical application. Several efforts to use immunohistochemistry to distinguish the molecular subtypes have been undertaken [Choi et al. 2009; Hans et al. 2004]. A recent report by Meyer and colleagues introduced a novel algorithm (Tally) that seems to be able to distinguish GCB and non-GCB DLBCL subtypes analyzing five immunohistochemical makers (CD10, GCET1, MUM1, FOXP1, and LMO2) [Meyer et al. 2011]. This algorithm showed a high concordance with gene expression profiling and the two identified groups of patients had different overall survival rates. However, this algorithm needs to be confirmed by other groups, especially as previous immunohistochemical approaches to determine the molecular DLBCL subtypes could not always be confirmed in additional analyses. This might be due to the limitations of immunohistochemistry, as staining and scoring procedures are variable even between experienced laboratories as shown by a recent study of the Lunenburg Lymphoma Biomarker Consortium. Therefore, technical variations between laboratories should be harmonized, before these algorithms can be reliably implemented into routine diagnostics [de Jong et al. 2009].
Recently, a quantitative nuclease protection assay was applied on FFPE tissue to distinguish DLBCL patient samples into ABC and GCB DLBCL [Rimsza et al. 2011]. Classification into the molecular subtypes using this assay was compared with results obtained by gene expression profiling applying DNA microarrays. 73% of GCB DLBCLs and 79% of ABC DLBCLs were categorized correctly using this assay (confidence cutoff >0.9) [Rimsza et al. 2011]. As the classification into molecular subtypes will be indispensable in the future for the rational design of clinical studies and the application of new pharmacologic agents, the use of this quantitative nuclease protection assay might hold promise due to its applicability on FFPE patient material. However, gene expression profiling remains the gold standard to distinguish the molecular DLBCL subtypes especially as there has been significant progress in the development of microarray protocols that can be performed on FFPE tissue [Williams et al. 2010].
In the last couple of years significant progress has been made in the development of novel compounds that specifically target oncogenic pathways (summarized in Table 1). Several of these strategies are currently being evaluated in clinical trials. However, these efforts will only be successful if the molecular target or signaling pathway is indeed expressed or utilized by the malignant cells. Thus, it is important to employ our molecular understanding of these diseases and incorporate techniques such as gene expression profiling that can be used to predict if a specific tumor is addicted to a certain oncogenic pathway.
Specific inhibitors targeting pathways that play an important role in DLBCL pathogenesis.

DLBCL, diffuse large B-cell lymphoma; ABC, activated B-cell-like; GCB, germinal-center B-cell-like; IKK, IκBα kinase; PMBL, primary mediastinal B-cell lymphoma.
Due to their dependency on NF-κB signaling, components of the NF-κB and/or BCR pathways might represent a promising therapeutic target for ABC DLBCL patients (Table 1). Specific inhibitors targeting PKC and MALT1 showed promising results in preclinical models [Naylor et al. 2011; Ferch et al. 2009; Hailfinger et al. 2009]. It is conceivable that these therapeutic approaches can be translated into the clinic. This was underscored by a recent study in which ABC and GCB DLBCL patients were treated with a combined approach of chemotherapy and the proteasome inhibitor bortezomib that can inhibit NF-κB signaling [Dunleavy et al. 2009]. Strikingly, only ABC DLBCL patients showed benefit by the addition of bortezomib. GCB DLBCL might preferentially respond to PI3K or mTOR inhibitors due to their dependency on the PTEN–PI3K pathway. Finally, PMBL might benefit from the addition of JAK2 inhibitors (Table 1). These efforts will eventually lead to more specific and effective and less-toxic treatment strategies of DLBCL patients.
Footnotes
This work was supported by research grants to GL from the German Research Foundation (DFG), the Deutsche Krebshilfe, the Berliner Krebsgesellschaft e.V., and the Else Kröner-Fresenius-Stiftung.
The authors declare no conflicts of interest in preparing this article.
