Abstract
Human norovirus is the main cause of viral gastroenteritis, resulting annually in ∼ 700 million infections and 200,000 deaths, of whom most are children <5 years. Mouse norovirus-infected macrophages are the most widely used
Introduction
Human noroviruses (HuNoVs) infections are mostly acute and self-limiting, although chronic norovirus infections occur in patients with an immunodeficient state.1–3 Despite the yearly global impact of ∼700 million HuNoV infections, 219,000 deaths and 60 billion dollars in societal costs there is no specific antiviral therapies to treat (chronic) norovirus gastroenteritis. To assess the anti-norovirus effect of small molecules, we describe in detail the use of mouse norovirus (MNV.CW1)-infected mouse macrophages (RAW264.7 cells) as cell-based antiviral assays for the identification and characterization of anti-norovirus activity and mechanism of action
We here describe the assessment of the antiviral activity of the selected compounds by two different methods which are complementary. Besides that, the toxicity of the compounds needs to be assessed to determine the therapeutic window. Compounds to be tested could include all those with previously reported antiviral activity against other +ssRNA viruses, libraries of compounds available from commercial suppliers (e.g. FDA-approved drugs) or from academic partners and institutions. The concentrations to test initially are normally in the µM to mM range.
Another, more sensitive, assay such as a plaque reduction assay could also be used to determine antiviral activity. 6 However, this technique is more laborious and very time-consuming, with the extra difficulty of using semi-adherent macrophages in an assay in which strong adherence of cells is required. Moreover, using the CPE-based assay as described here, weaker antiviral compounds could be selected which give room for optimization and production of potentially more potent analogues.
Expertise needed to implement the protocol
The scientist needs to be trained to work with cell culture and pathogens at BSL-2 level.
Materials and reagents
Appropriate personal protection to work in a biosafety level 2 (BSL-2) laboratory (gloves, lab coat, shoe covers, safety glasses)
Disinfectant: bleach (5,000 ppm) or Virkon S.
Culture flasks (150 cm2, TPP, catalog number: 90856)
Cell scrapers (Corning, Falcon®, catalog number: 353086)
Cryotubes (Thermo Fisher Scientific, Thermo Scientific, catalog number: 377224)
Pipet filter tips (10 µL, 100 µL, 1,000 µL)
Disposable serological pipets (5 mL, 10 mL, 25 mL)
Disposable aspirator pipets (Falcon, catalog number: 357558)
Murine macrophage cells (RAW 264.7, ATCC, catalog number: TIB-71)
Murine norovirus (MNV-CW1, ATCC, catalog number: VR-1937)
Dulbecco’s phosphate buffered saline (PBS) (Thermo Fisher Scientific, catalog number: 14190094)
Dulbecco’s modified eagle’s medium (DMEM) (Thermo Fisher Scientific, GibcoTM, catalog number: 41965039)
Fetal calf serum (FCS) (Thermo Fisher Scientific, GibcoTM, catalog number: 10270106)
Sodium bicarbonate (Thermo Fisher Scientific, GibcoTM, catalog number: 25080060)
L-Glutamine (Thermo Fisher Scientific, GibcoTM, catalog number: 25030024)
HEPES (Thermo Fisher Scientific, GibcoTM, catalog number: 15630056)
Penicillin/streptomycin (P/S) (Thermo Fisher Scientific, GibcoTM, catalog number: 15140148)
Sodium pyruvate (Thermo Fisher Scientific, GibcoTM, catalog number: 11360039)
Minimal Essential Medium (MEM) without phenol red (Thermo Fisher Scientific, GibcoTM, catalog number: 51200046)
Eppendorf safe-lock tubes, 1.5 mL (Eppendorf, catalog number: 0030120086)
2′-
Falcon tubes 50 mL (Greiner Bio-one, catalog number: 227261)
CellTiter 96 AQueous MTS Reagent Powder (MTS, Promega, catalog number: G1111)
96-well plate (Falcon, catalog number: 353072)
Phenazine methosulfate (PMS, Sigma, catalog number: P9625)
Filter 0.22 µm (Thermo Scientific Nalgene catalog number: 161–0020)
Trypan Blue Stain, 0.4% (Logos Biosystems; Catalog number: T13001)
MNV forward primer: 5′-CAC GCC ACC GAT CTG TTC TG -3′ (Integrated DNA Technologies) 7
MNV reverse primer: 5′-GCG CTG CGC CAT CAC TC-3′ (Integrated DNA Technologies) 7
MNV RT-qPCR standard: 5′- tagaaatggtagtccc acgccaccgatctgttctgcg ctgggtgcgctttggaacaatgga tgctgagaccccgcaggaacgctcagcagtctttgtgaatgaggatgagtgatggcgcagcgccaaaagccaatggct -3′ (Integrated DNA Technologies) 4
MNV probe: 6-FAM–MGB 5′-CGC TTT GGA ACA ATG-3′ (Thermo Fisher Scientific) 4
RT-qPCR kit (iTaq Universal Probes One-Step Kit Catalog number: 1725141)
Equipment
Biosafety hood in a BSL-2 laboratory
Incubator (37°C, 5% CO2, humidified)
Pipet set (P10, P100, P1000)
Pipetboy (Integra Biosciences, catalog number: 155016)
Multipette® M4 (Eppendorf, catalog number: 4982000012)
Multichannel pipette (Eppendorf, catalog number: 3122000035)
−80°C freezer
PCR Workstation
Vortex
Centrifuge with a rotor suitable for 1.5 mL tubes
Centrifuge with a rotor suitable for 50 mL tubes
Inverted light microscope
Cell counter (Logos Biosystems Luna-II automated Cell counter, catalog number L400002)
Aspirator (Integra BioSciences Vacusafe catalog number: 158320)
Plate reader (SPARK® Multimode Microplate Reader)
Stirring plate
pH meter
Procedures
Cultivation of RAW 264.7 cells
Thaw RAW264.7 cells and seed these in 10% culture media (see recipes).
Grow the RAW264.7 cells in a CO2 incubator at 5% CO2 and 37°C.
Maintain the RAW264.7 cells by splitting of the cells once a week at a 1:10 ratio in a T-150 flask.
Expansion and quantification of Murine Norovirus.CW1 virus stock
When the RAW264.7 cells are ∼90% confluent infect the monolayer with MNV.CW1. When full cytopathic effect (CPE) is observed (∼2–3 days post infection, see Figure 1) perform two freeze-thaw cycles (−80°C vs. 37°C), collect the supernatants containing the virus after centrifugation (10 min, 1 500
Determine the viral titer by endpoint titration as previously described in Reed
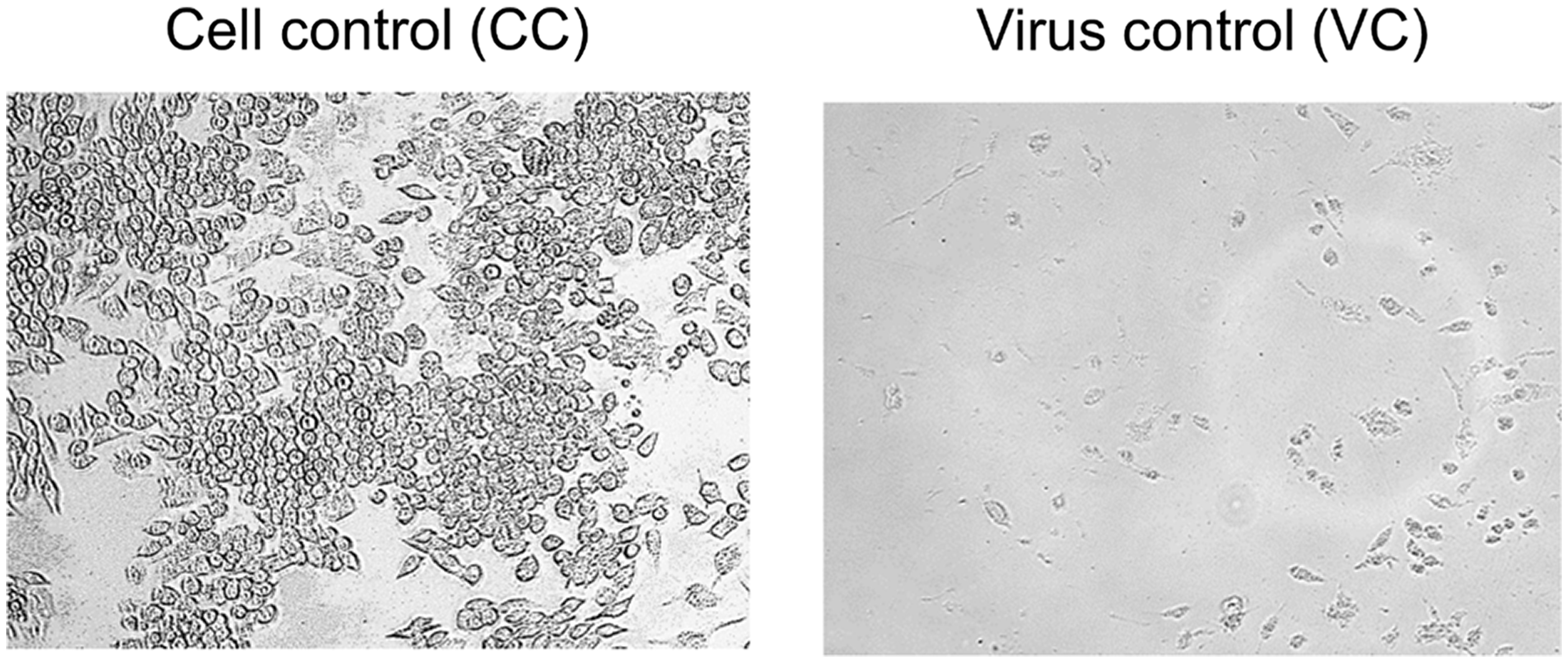
Macroscopic view of virus induced CPE. Left: uninfected RAW264.7 cells after 3 days, Right: MNV-infected RAW264.7 cells after 3 days.
Assessment of anti-norovirus activity
Add 100 µL of 2% culture media in all wells of a 96-well plate (see Figure 2).
Add 25 µL of 2% culture media in the first column.
Add the appropriate volume of compound to 1B-1G. It is recommended to include a reference compound such as 2CMC 5 at least once in a set of test plates. In each plate, rows A and H, columns 1 and 12 are not used for the assay, but simply to buffer the test wells, avoiding evaporation effects.
Mix the compound and medium with a multichannel pipette by pipetting up and down at least three times.
Take 100 µL of the compound dilution and transfer into the next column (e.g. 2B-2G).
Repeat steps 3–4 until reaching column 9 of the 96 well plate. Changing tips between rows is not required but might yield better results with some compounds (e.g. more viscous compounds).
Mix the compound and medium in column 9 and discard 100 µL.
Scrape the RAW 264.7 cells from a tissue culture flask using 5 mL of fresh assay media, homogenize the suspension by pipetting vigorously up and down and then determine the density of the cell suspension (cells/mL).
Prepare a 10,000 cells/50 µL in 2% culture media, by diluting appropriately the cell suspension.
Prepare a virus dilution (which is 4 times more concentrated then the desired final concentration, MOI of 0.1) in 2% culture media.
Add 50 µL of virus suspension to each well of rows 2–10.
Add 50 µL of the cell suspension to the inner 60 wells of the 96-well plate (rows 2–11, columns B-G).
Incubate the 96-well plate for 72 h in a CO2 incubator at 5% CO2 and 37°C.
Assess the antiviral effect of the compounds via an MTS assay and virus yield assay.
Calculate the EC50 value by logarithmic intrapolation.
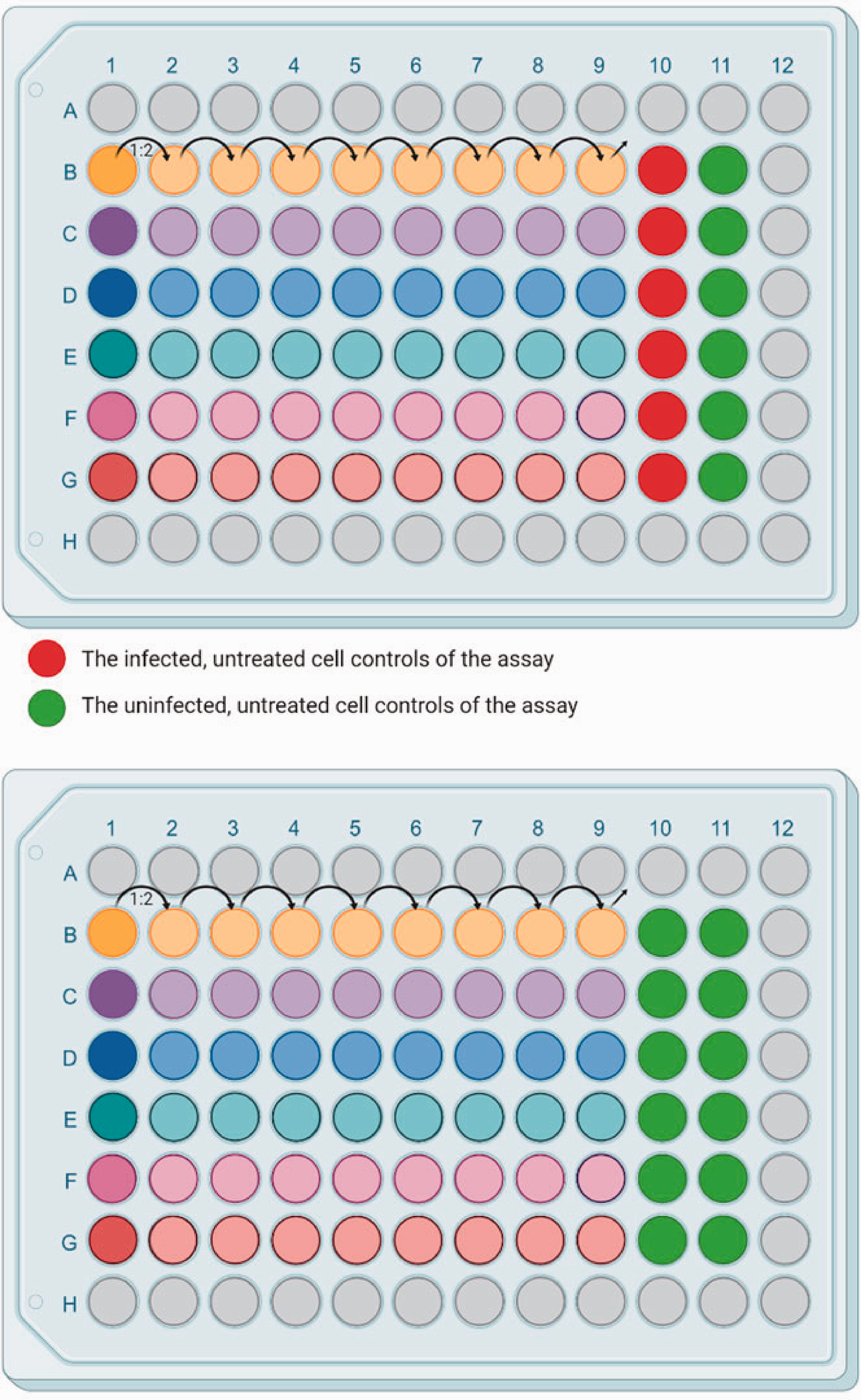
Layout of the dilution series of selected compounds on RAW 264.7 cells (1 x 104 cells/well). Antiviral assay (top) and cellular toxicity assay (bottom) in a 96-well plate. Created with BioRender.com.
Assessment of cellular toxicity
Add 100 µL of 2% culture media in all wells of a 96-well plate (see Figure 2).
Add 25 µL of 2% culture media in the first column.
Add the appropriate volume of compound to 1B-1G. In each plate, rows A and H, columns 1 and 12 are not used for the assay, but simply to buffer the test wells, avoiding evaporation effects.
Mix the compound and medium with a multichannel pipette by pipetting up and down at least three times.
Take 50 µL of the compound dilution and transfer into the next column. Tip change is an option; proceed here the same way than for the antiviral assay.
Repeat steps 5–6 until reaching column 9 of the 96 well plate.
Homogenize the compound and medium in row 9 and discard 50 µL.
Count the RAW 264.7 cells/mL of cell suspension and prepare a 10,000 cells/100 µL in 2% culture media.
Add 100 µL of the cell suspension to the inner 60 wells of the 96-well plate.
Incubate the 96-well plate for 72 h in a CO2 incubator at 5% CO2 and 37°C.
Determine the cytotoxicity of the compounds via an MTS assay.
MTS colometric assay
Prepare MTS solution (see recipe).
Make a 1:10 MTS dilution prepared in MEM without phenol red.
Collect the supernatant from the wells and store in a plate or individual tubes at −20°C for virus yield assay or discard.
Add 75 µL of MTS dilution to the inner 60 wells and incubate the 96-well plate for 2 h in a CO2 incubator at 5% CO2 and 37°C.
Measure the optical density (OD) at 498 nm in a plate reader.
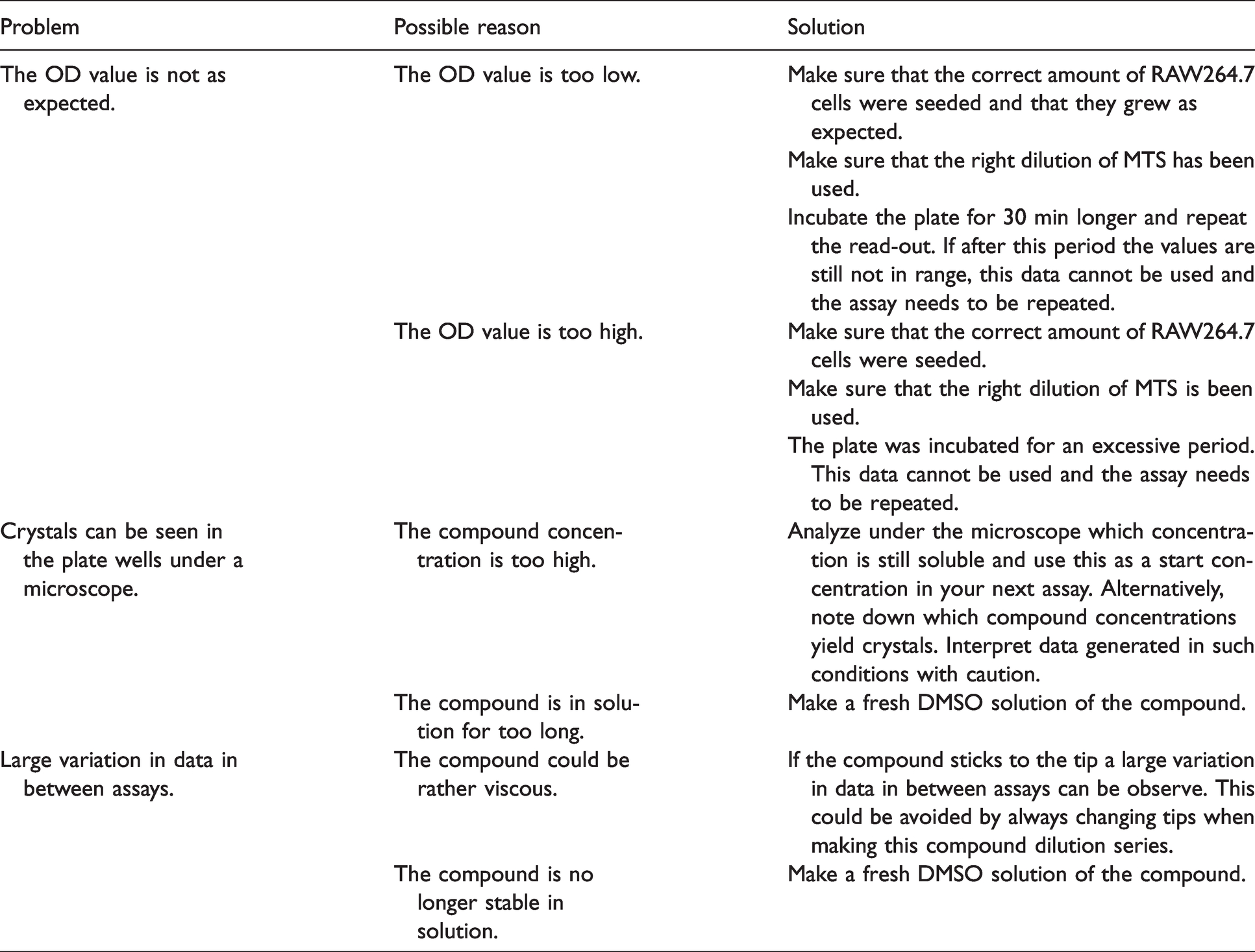
To validate the assay, perform quality control as follows: the cell control value should be between 0.6–1 and the virus control below 0.2.
A Z′ score can be calculated to assess assay quality. Calculate as follows:
When a compound results in a protective effect, perform visual scoring of the wells with “active” concentrations to confirm the presence of viable cells and absence of CPE.
Calculate the % of CPE reduction as follows: [ODtreated x ODVC]/[ODCC x ODVC] x 100, where ODCC represents the OD of the uninfected untreated cells, whereas ODVC represent the OD of infected untreated cells. The 50% effective concentration (EC50) is defined as the compound concentration that protected 50% of the cells from virus-induced CPE. Calculate the EC50 value by logarithmic intrapolation.
Calculate the % cell viability as follows:(ODtreated/ODCC)x100 where ODtreated represent the OD of treated cells, whereas ODCC represents the OD of the untreated cells. The 50% cytotoxicity concentration (CC50) is defined as the compound concentration that reduces the number of viable cells by 50%. Calculate the CC50 value by logarithmic intrapolation.
Troubleshooting advice.
Virus yield assay
Collect the appropriate volume of supernatant (according to the manufacturer’s protocol) of the treated and untreated infected cells in separate and adequately labelled Eppendorf tubes.
Extract the viral RNA using an appropriate kit, according to the manufacturer’s protocol. We standardly use the NucleoSpin RNA Virus Kit (Macherey–Nagel).
Store the RNA extracts at −20°C for short time storage and at −80°C for long time storage.
Perform an RT-qPCR to quantify the RNA levels. Cycling conditions are: reverse transcription at 50°C for 10 min, initial denaturation at 95°C for 3 min, followed by 40 cycles of amplification (95°C for 15 s, 60°C for 30 s). Use 900 nM of primers and 200 nM of probe.
Calculate the % of virus yield reduction is calculated as [(number of RNA copies VC)/(number of RNA copies treated cells)]x100. The 50% effective concentration (EC50) is defined as the compound concentration that reduced 50% of the viral RNA in the supernatant.
Expected results
Upon determination of the antiviral effect of a compound, here we give an example of the results to be expected for an active compound. We present a dose-response generated by using the reference compound 2CMC which activity we have described earlier. 5 The antiviral effect has been assessed as described in this protocol; the data was plotted as % of control as shown in Figure 3 using software such as Graphpad Prism. Compounds with EC50 values in the low µM range or lower are considered as having a good antiviral activity. Additionally, the CC50 values should be significantly higher to obtain a good therapeutic window.
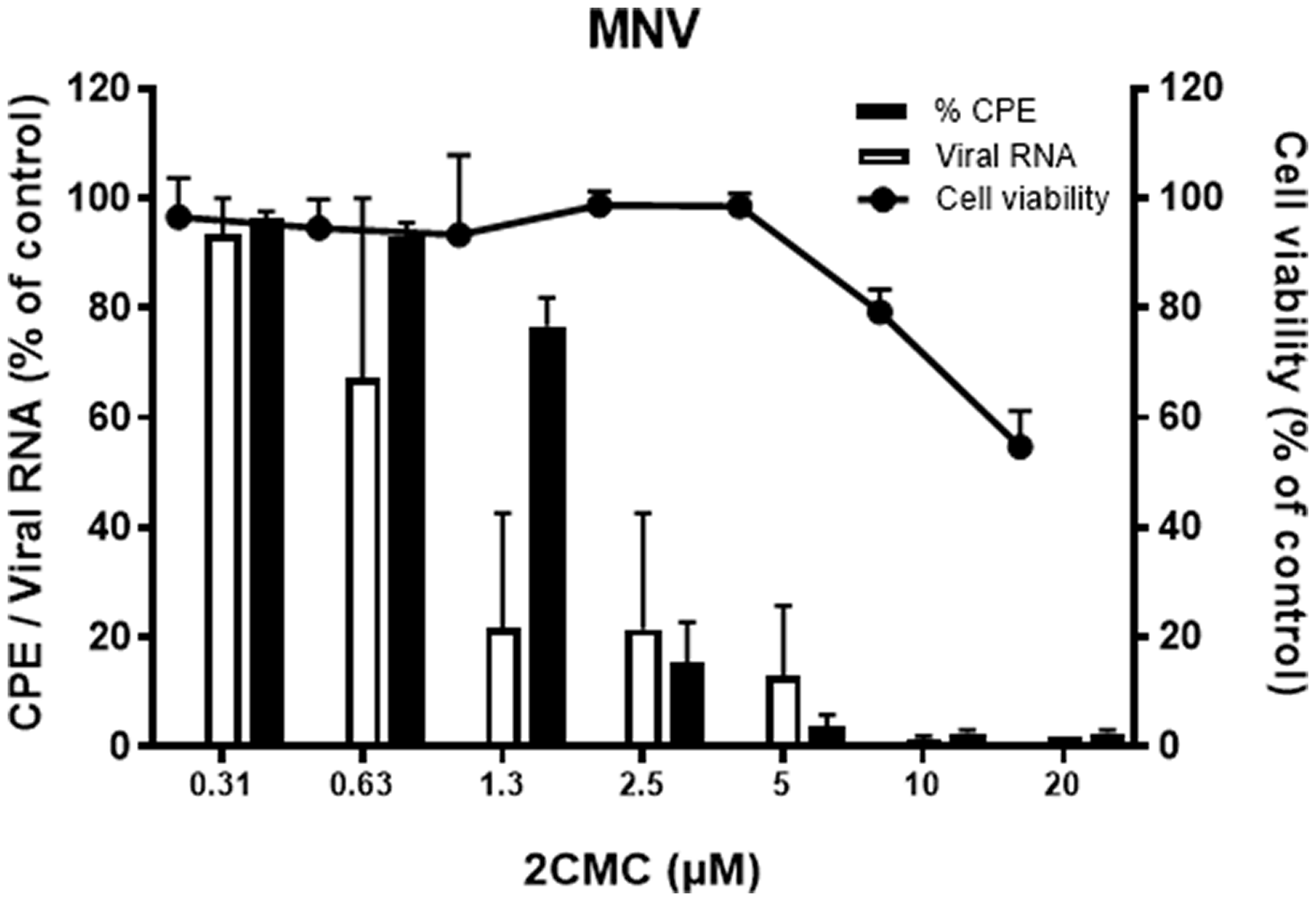
Cellular toxicity and
Conclusion
This easy and high-throughput assay allows for a good assessment of a high number of compounds for their potential antiviral activity against MNV. Antiviral compounds with a good
Recipes
Culture media for RAW264.7 cells
MTS preparation
! MTS is a light-sensible reagent, all recipients should be wrapped in aluminum foil and all handlings should be carried out in an as dark as possible environment.
! Freeze the MTS solution in aliquots of 2 mL
2 g of MTS Dissolve in 1 L sterile PBS Stir for 15 min Add 46 mg PMS Measure pH and adjust to a range of 6–6.5 Filter the suspension (0.22 µm) Freeze at −20°C in aliquots of 2–3 mL
Trouble shooting
See Table 1.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
