Abstract
Ameloblastoma is a rare tumor arising from odontogenic cells that is benign, yet locally aggressive. Metastasizing ameloblastoma (METAM) is an ultra-rare ameloblastoma variant in which both primary and secondary tumors have histological features of benign ameloblastoma. This is a case report of a patient who presented with a jaw mass and subsequent lung metastases, later diagnosed as METAM. Initial treatments, including carboplatin, etoposide, and taxane-based chemotherapy, were ineffective. Molecular profiling revealed mutations including PIK3CA H1047R and BRAF V600E. The patient was enrolled in a tumor-agnostic trial and began treatment with copanlisib, a PI3K inhibitor, which resulted in a partial response and durable disease regression. After 76 cycles, she continues to tolerate therapy well with minimal adverse events. This case highlights the potential of targeted therapies such as copanlisib for treating METAM, providing a promising therapeutic option for patients with PIK3CA mutations.
Keywords
Introduction
Ameloblastoma is a rare tumor arising from epithelial rests of Malassez, quiescent epithelial cell remnants involved in the formation of dental lamina that remain in periodontal tissues throughout adult life. 1 They are the most common odontogenic neoplasm, yet only represent 1% of all oral tumors. While some literature has labelled these tumors as “benign” and they do often have benign appearing histological features, they can also invade locally adjacent vital structures. These tumors are often locally aggressive and have a high recurrence rate.
Metastasizing ameloblastoma (METAM) is an ultra-rare ameloblastoma variant. Notably, both primary and secondary tumors have histological features of benign ameloblastoma and METAM is a diagnosis made only in retrospect. This is in contrast to ameloblastic carcinoma which exhibits malignant histological features. Currently, there are no specific histological features to predict METAM. A recent review of 42 published cases within the past decade (2010–2019) showed a mean age of occurrence of 43 years (range 8–74 years) with a slight male predominance. 2 The most common site of primary lesion for METAM is the mandible, likely owing to the overall higher incidence of solid multi-cystic ameloblastoma in the mandible compared with other gnathic bones. The mean latent period between initial diagnosis and metastatic disease was 12 years with a range of 0–35 years (Four cases had metastatic disease on initial presentation). The most common site of metastasis is the lungs (80% of cases), followed by the lymph nodes. Patients diagnosed with metastatic ameloblastoma to the lung have a median survival of 3 years with a 5-year survival rate of 37%. 3 Given the rarity of these tumors, there is currently no established treatment protocol.
The growing availability of comprehensive molecular profiling platforms has led to a large increase in predictive biomarkers and novel oncogenic targets, which in turn has spurred a shift towards tumor-agnostic therapy. Tumor-agnostic therapy is a treatment strategy that utilizes genomic information to identify potential novel therapies regardless of histological origin. The NCI sponsored MATCH trial was designed to evaluate whether patients whose tumors harbor specific molecular alterations benefit from matching targeted therapies in independent sub-protocols regardless of histology. 4
Consent
Written informed consent was obtained from the patient for publication.
Patient case
The patient initially presented in October of 1996 with a jaw mass. She was then noted to have synchronous lung nodules incidentally found on a chest X-ray ordered for a positive Purified Protein Derivative (PPD) test. This was followed by a lung biopsy which revealed ameloblastoma (Figure 1). She was found to have METAM with the primary site involving her jaw and with involvement of lungs bilaterally. (Note: histopathology of the primary tumor was not available in the Electronic Medical Record). She underwent treatment with three cycles of Carboplatin (750 mg/m2 day 1) and etoposide (200 mg/m2 days 1 + 2) until 1997 and then received taxane-based chemotherapy in early 2002 with no response to the latter regimen. Following completion of these therapies, she underwent close monitoring with CT scans every 3 to 6 months. Lung biopsy specimen. Histologic assessment of the lung biopsy shows a trabecular and nested arrangement of tumor cells with basal palisading and central stellate reticulum, diagnostic of metastatic ameloblastoma. Some areas with squamous, follicular, and granular cell changes are also seen. (H&E, 100x).
She was subsequently found to have progressive disease first noted in February 2012 on a CT scan showing progression of her lung lesions. Treatment options were discussed, including chemotherapy with cisplatin/adriamycin. As the patient was asymptomatic, a recommendation was made to hold off on chemotherapy and continue serial imaging. The patient’s disease remained stable on follow up CT imaging until February of 2018, when her course was complicated by new onset hypoxia. CTA imaging was done to rule out a pulmonary embolism which showed stable innumerable metastatic pulmonary nodules and masses. However, repeat CT imaging 5 months later revealed an increasing disease burden with multiple lesions showing an interval increase of 2 mm. She underwent repeat lung biopsy in August of 2018, complicated by pneumothorax treated with chest tube placement. Histology of the lung biopsy showed islands of basaloid cells in a palisading arrangement with high nuclear to cytoplasmic ratios, resembling ameloblast like cells. Tempus molecular profile of the biopsied tissue showed the following mutations: BRAF V600E, PIK3CA H1047R, SMARCB1 R377H, and MED12 G44V.
She was then enrolled in the MATCH clinical study, subprotocol Z1F: Phase II Study of Copanlisib in Patients with Tumors with PIK3CA Mutations (PTEN Loss Allowed). She has been receiving copanlisib 60 mg IV on days 1, 8 and 15 of each 28-day treatment cycle. She has tolerated therapy well with mainly grade 1 adverse reactions and two Grade 2 adverse reactions. The most common grade 1 adverse reactions attributed to copanlisib were headache and rash. The patient experienced Grade 2 left upper extremity bursitis attributed to copanlisib and Grade 2 rash in her arms, chest and neck attributed to topical body oil. Since beginning therapy with copanlisib, she has experienced notable disease regression with a partial response per the Response Evaluation Criteria in Solid Tumors (RECIST) (v1.1) Criteria (Figure 2). Imaging completed after cycle 65 showed a 38.9% decrease in the target lesion from 11.3 cm to 6.9 cm. The patient has now completed cycle 76 and she continues to tolerate therapy well. Metastatic lung disease regression from prior to starting copanilisib (left image) to the most recent imaging from June 2024 (right image).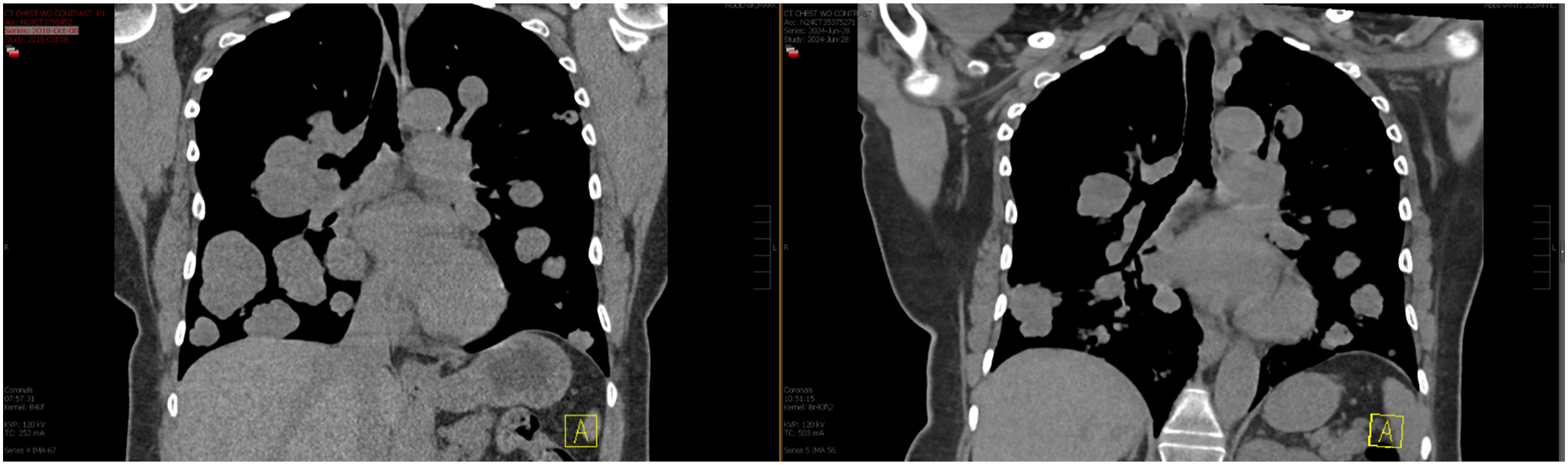
Discussion
PIK3CA is one of the most commonly mutated oncogenes and has been observed in many tumor types including colon, breast and ovarian. 5 Consequently, targeting PIK3CA has gained major clinical interest. Unfortunately, initial efforts to target PIK3CA mutations has yielded mixed results due to dose-limiting toxicities and lack of effective isoform-specific inhibitors. Copanlisib is a class I phosphoinositide 3-kinase inhibitor targeting the α (encoded by PIK3CA) and δ (encoded by PIK3CD) isoforms.
The NCI-MATCH is a multi-arm tumor-agnostic trial for patients with refractory cancers selected on the basis of tumor genomic alterations to receive matching targeted therapies. MATCH Subprotocol Z1F evaluated the effectiveness of copanlisib in tumors with PIK3CA mutations. Adult patients with any solid tumors or myeloma with an activating mutation in PIK3CA in their tumor and who had either progressed on standard treatment or for whom no curative treatment existed were eligible for enrollment on this arm.
Data for this phase II trial was reported in early 2022 with a time of data cutoff of January 6, 2021. Twenty-five patients were included in the primary efficacy analysis and the overall response rate (ORR) was 16% (90% CI: 6%-33%). Clinical benefit rate (defined as complete response, partial response, or stable disease for at least 6 months) was 36% (9 out of 25 patients). The Z1F study met its primary end point with copanlisib, showing promising clinical activity in select tumors with PIK3CA mutation in the late-line, refractory setting. In addition to the clinical case detailed above, partial responses were observed in endometrial carcinoma, low-grade myxoid liposarcoma and clear cell carcinoma of the anterior abdominal wall. Out of nine patients with H1047R PIK3CA variants, two experienced partial responses and two maintained stable disease.
Out of 30 patients included in the toxicity analysis, there were 16 grade 3 toxicities and one grade 4 toxicity (hyperglycemia). Of the grade 3 toxicities, the most common were hypertension (n = 9, 30%), hyperglycemia (n = 7, 23%), maculopapular rash (n = 2, 7%), generalized muscle weakness (n = 2, 7%), and dehydration (n = 2, 7%). Grade 4 hyperglycemia was experienced by one patient (3%). Three patients discontinued the study drug due to toxicity. 6
In the past decade there have only been 18 reported cases of METAM worldwide. 3 While some patients may be treated successfully with resection of the metastatic tumor,7,8 treatment options for patients with unresectable metastases remain limited. Evidence supporting the role of chemotherapy in these instances is mixed, with some studies showing lack of objective improvement9,10 while others report partial response with the combination of cisplatin and cyclophosphamide 11 as well as tumor regression with agents including Vinblastine, Bleomycin, Paclitaxel and Carboplatin.12–15 Li et al. reported partial remission after six cycles of mesna, adriamycin, ifosfamide and dacarbazine with a progression-free survival of more than 9 months.
As our insights into the driver mutations of METAM evolve, targeted therapies may allow for improved effectiveness and fewer systemic adverse effects than standard chemotherapy. For example, a complete response was seen in a 26-year-old female with METAM revealing a BRAF V600E mutation treated with the combination of BRAF-inhibitors dabrafenib and trametinib. 16 Similar case reports have shown durable clinical responses with BRAF inhibitors.17,18 Among ameloblastomas of the maxilla, mutations in BRAF are most commonly seen (∼50%) followed by mutations in SMO (40%) with mutations in PIK3CA seen in <3% of tumors.19,20 The patient featured in this case has a co-existent BRAF-V600 E alteration, a stronger driver, yet has still shown prolonged response to copanlisib monotherapy.
The durable clinical response with low associated toxicity seen in our patient along with the encouraging evolving data on tumor-agnostic targeted therapies provides promising new treatment pathways for patients with METAM.
Footnotes
Author contributions
Meghan Lynch wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
