Abstract
Osteosarcomas of the ribs are rarely reported but have a high potential for pulmonary metastases. The therapeutic strategies for this disease are not well defined. The primary treatment recommendations include wide resection with clear surgical margins and chest wall reconstruction if needed. We present a case of costal osteosarcoma with pleural and lung extension successfully treated by en-bloc thoracic resection with free surgical margins and chest wall reconstruction with rib titanium bars, polypropylene mesh and a rectus abdominis free flap with microvascular anastomoses. This case demonstrates the importance of this therapeutic strategy and highlights the need of early intervention in managing this disease.
Keywords
Introduction
Osteosarcoma is the most common primary bone tumor in children and adolescents, with a higher incidence in the second decade of life and more common in men,1,2 arising from bone-forming mesenchymal cells. 3 The fibroblastic type is related to poor survival. 4
Osteosarcomas rarely occur in flat bones, so rib sarcomas are rarely reported. 5 Hu et al. 2 described one case and found about 17 other cases of primary rib osteosarcomas described in the English literature up to September 2023.
This tumor has a high potential for metastases, with the lungs being the most common site of spread in approximately 90% of cases. It is important to note that pleural metastases are rarely reported.6,7 The 5-year survival rate is less than 20% after the development of metastases. 8
Excisional biopsy and needle biopsy are essential methods used to confirm the diagnosis of bone and soft tissue tumors. 9
It must be considered that primary rib osteosarcoma has not been extensively investigated, with therapeutic strategies not defined, being surgery with wide margins the preferred local treatment of the primary tumors, and whenever feasible, with radiotherapy on patients in whom such margins could not be accomplished, so early preoperative diagnosis and definitive local control remain major challenges and should contribute to improved outcomes. 10
In this manuscript, we present a patient with costal osteosarcoma with pleural and lung extension that was successfully resolved through en-bloc chest resection and thoracic wall reconstruction followed by chemotherapy and radiotherapy.
Case description
We present the case of a 61-year-old male patient with a history of well controlled hypothyroidism with levothyroxine, grade I obesity, and laparoscopic cholecystectomy who was seen by other physicians with sudden, moderate pain in the left thoracic region for 3 months attributed to physical exertion, without modifiers that resolve spontaneously and with recurrence at a lower intensity level, but without improvement. Furthermore, enlargement of the left supra-mammary region of the upper outer quadrant with an indurated and fixed mass of approximately 6 × 7 cm in diameter, with defined borders and mild pain on palpation, no changes in skin color, no ulcerations, no discharge, and no weight loss, therefore, paraclinical studies performed showed on chest CT a tumor mass of the chest wall in the anterior arch. of the fifth left rib with a significant lytic lesion with heterogeneous areas suggestive of central necrotic changes, with a diameter of 5.45 × 6.0 × 6.3 cm with a wide base of pleural invasion projecting toward the middle anterior third of the left hemithorax with infiltrative effect on the soft tissues of the chest wall (Images 1(A) and (B)). A tru-cut needle biopsy of the lesion was done, and histopathologic examination revealed a malignant fusocellular osteoid-forming malignant neoplasm compatible with osteosarcoma, for which a multidisciplinary surgical solution was chosen during the tumor board meeting in our hospital after the final pathology results from the biopsy were reviewed, due to the feasibility of complete en-bloc resection, and considering that in this case not even neo-adjuvant therapy would improve global surviving. Chest CT with evidence of the tumor (white arrows).
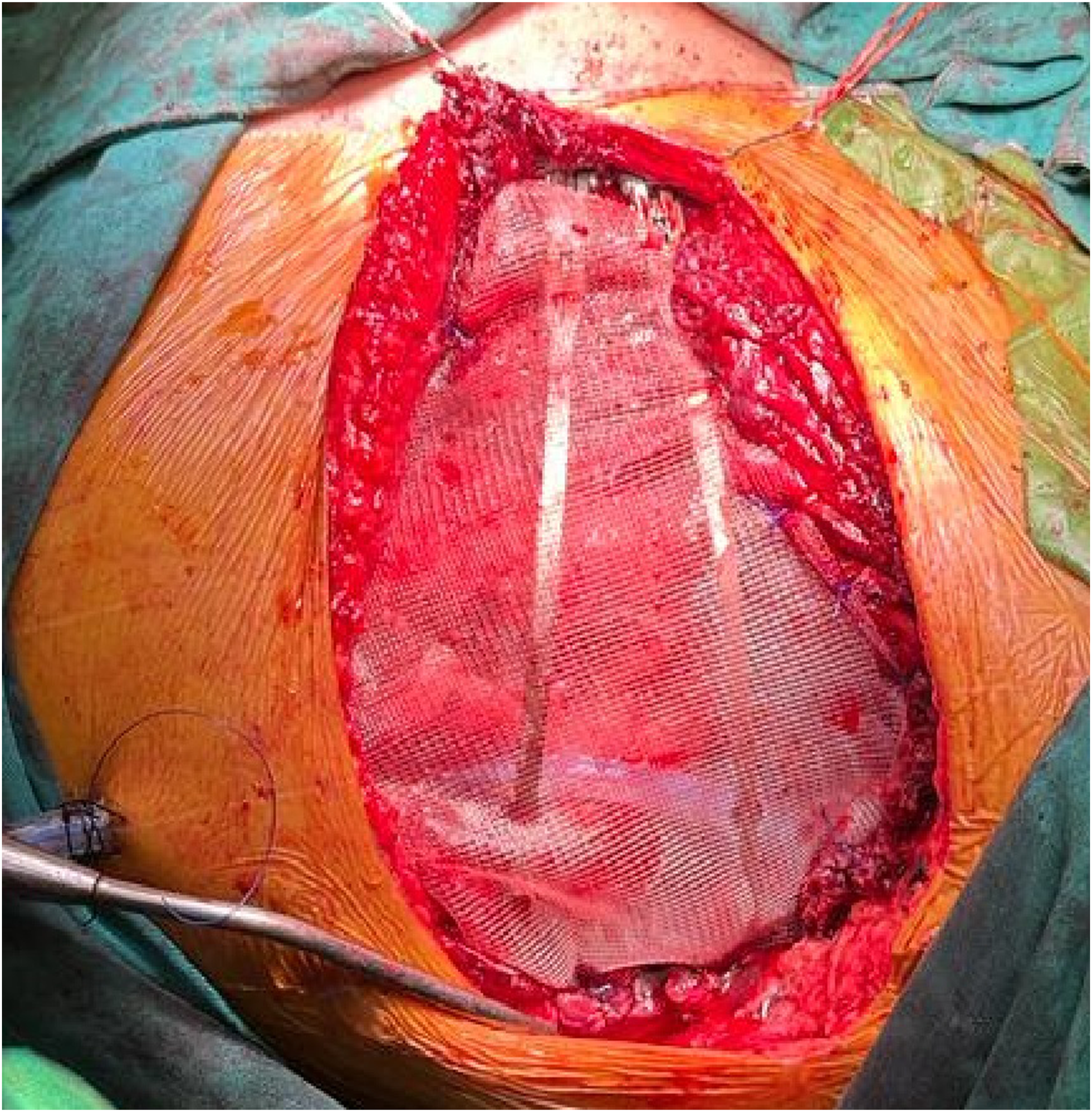
The cardiothoracic surgery team performed a bronchoscopy procedure with findings of the left bronchial tree without evidence of macroscopic lesions, with a cytology report of left alveolar bronchial lavage with reactive bronchial hyperplasia, negative for malignancy, and a left anterior thoracotomy with findings of a tumor lesion in the anterior aspect of the fifth costal arch. of hard consistency, non-mobile, with invasion to the parietal pleura and lung parenchyma of segment 4 of the left upper lobe. Therefore, en-bloc resection of the chest wall including costal arches of the fourth, fifth, and sixth ribs with lung resection was done, as well as the skin, adipose tissue, pectoral muscle, and lung parenchyma resection with auto-sutures, with a pathological sample of about 19 × 11 × 5 cm of diameter and a trans-operative frozen study showing tumor-free surgical margins (Image 2(A) and (B)). This was followed by chest wall reconstruction with two costal bars and a polypropylene mesh and placement of a chest drainage tube (Images 3 and 4). The plastic surgery team then completed the operation with a microsurgical reconstruction of the chest wall defect with a rectus abdominis myocutaneous free flap, with microvascular anastomoses, using the left internal mammary artery and mammary veins as the recipient vessels, carefully dissected by the previous team without complications (Image 5). (A) 3D printing of the tumor and rib cage. (B) En-bloc resection of the chest wall tumor. Chest wall reconstruction with 2 costal bars and polypropylene mesh placement. (A) Post-surgical chest X-ray imaging. (B) Follow up chest x-ray. Microsurgical reconstruction and rectus abdominis myocutaneous free flap conformation.


Immediately after surgery, the patient was transferred to the intensive care unit for observation and initial treatment, followed by hourly Doppler checks with adequate biphasic flow at marked areas of the flap which remained vital and had good-capillary filling.
The patient progressed positively and was discharged from the hospital on seventh day after surgery.
The final histopathological report confirmed the presence of a grade 2 osteosarcoma of conventional, fibroblastic, chondroblastic, and telangiectatic type in the fifth-rib arch. measuring 7 × 6.5 × 5.5 cm, tumor stage pT2-pNx-pM1, with presence of invasion in the upper lobe of the left lung, the resected lesion had free margins (Image 6). (A) Histopathological images. HE 4x: conventional osteosarcoma with telangiectatic areas. (B) Histopathological images. HE 20x: fibroblastic component. (C) Histopathological images. HE 10x: chondroblastic component.
Patient was treated with chemotherapy of Doxorubicin 75 mg/m2 plus Ifosfamide 2500 mg/m2 for 6 cycles after his hospital discharge, and then with radiotherapy 60 Gy in bed, divided into 45 days after completing chemotherapy, these last two treatments were done in a different medical institution as per patient`s relatives request due to financial issues.
Follow-up of the patient during this first post-surgical year after completing neo-adjuvant therapy was made with physical exam, total body tomography every 3 months to look for local recurrences, lung metastases. During this follow-up, the patient presented grade 2 hematological toxicity due to chemotherapy characterized by neutropenia requiring growth factors of neutropenic colonies, accompanied by alopecia, asthenia, and nausea that was controlled with antiemetics.
Discussion and conclusions
According to current medical literature, the primary treatment for rib osteosarcoma involves a broad resection, including superior and inferior ribs, which must be resected en-bloc and with clear surgical margins, followed by chest wall reconstruction if required. An autologous tissue substitute, GORE-TEX® implants, and meshes (e-PTFE; Gore-Tex, Newark, DE) can be used for this procedure. 11
Nevertheless, it may be difficult to close the chest wall defect following resection, so it must be considered that the indications for reconstruction are any chest wall defect larger than 5 cm; however, on the posterior side of the chest wall, defects up to 10 cm might not impair function due to the presence of the scapula.12,13
There is currently no “gold standard” recommended procedure for reconstructing the chest wall; instead, the surgical approach should be customized based on the clinical condition of the patient, any underlying diseases, and the location and size of the chest wall defect. The use of prosthetic materials ought to be determined by the surgeon’s experience, local availability, and cost. Thus, reconstruction of the chest wall depends on pathology, location, and age. It needs to shield the intrathoracic structures, maintain long-term cardiac, respiratory, and cosmetic functions, allow for future growth and development, prevent upper extremity instability, and not interfere with adjuvant radiation therapy. In cases of cancer, the goal of the procedure is also to obtain the widest margins possible, free of malignant disease or for palliative purposes.13,14
Two broad categories can be used to categorize chest wall reconstruction techniques: non-rigid fixation and rigid fixation. Combinations of synthetic meshes, bioprosthetic materials, steel or titanium constructs, autografts, homografts, and bovine xenografts can be used for relatively simple or complex bone reconstruction, while direct closure, skin grafts, local advancement flaps, pedicled or free myocutaneous or osteomyocutaneous flaps, or a combination of these, can be used to cover soft tissue.12,14
Dai et al. 15 recommend choosing mesh and titanium bars as reconstruction methods for being these choices more reasonable and safer, while the latissimus dorsi flap can be a good technique for repairing soft tissue defects. However, the choice of technique and material is more dependent on the surgeon’s experience, the location and size of the chest defect, the cost, and the availability of materials locally. 13
As we mentioned before, our patient was diagnosed with costal osteosarcoma invading soft tissues, pleural and pulmonary tissues, which led to a very unusual presentation of this type of neoplasia, so based on our previous experience with the multidisciplinary management of chest tumors, such as in a case published in 2023
16
where our cardiothoracic team performed a left thoracotomy, en-bloc resection of a chest wall tumor with diaphragm resection, left lung lobectomy and posterior reconstruction of the chest wall and diaphragm; and based on the medical literature recommendations for the management of this type of tumor, such as the tumor location in the costal wall, the size bigger than 5 cm, and the structures involved around it as in example the lung parenchyma, we made the 3D reconstruction and after analyzing the images, based on our past experience in similar cases, we decided the interdisciplinary surgical treatment, proceeding to extract the mass with en-bloc resection of the chest wall tumor including the skin, adipose tissue, pectoral muscle, fourth, fifth, and sixth costal arches, and lung parenchyma with auto sutures and with free surgical margins confirmed with trans-operative frozen biopsy study, and then a reconstruction of the chest wall with two titanium costal bars and a polypropylene mesh followed by a rectus abdominis myocutaneous free flap with microvascular anastomoses, after which the patient showed an adequate evolution and recovered according to plan (Image 7). Patient condition and flap result during follow up.
Finally, we have to mention that as potential limitations in our approach, there may be to obtain surgical margins due to the complexity of this osteosarcoma which involved soft tissues, pleura, and lung, leading a challenge to obtain optimal wide margins, and the inherit potential complications of the reconstruction with a myocutaneous free flap such as injury to the anastomosed pedicle of the flap, for which in our case an adequate space was created in the mesh to keep it in place without damaging the vascular anastomoses.
There is also a discussion about neoadjuvant therapy because conventional osteosarcomas typically respond well to chemotherapy, but other subtypes like fibroblastic osteosarcoma may be more resistant, and in this case, the tumor was a mix of fibroblastic, chondroblastic, and telangiectatic components, which may present variable responses to chemotherapy, so there was a high risk of inadequate response, and postoperative complications like infections or delayed wound healing.
Also, obesity was a mild limitation during the surgical approach due to the excess of fat tissue which conditioned to a more extensive dissection, and during the immediate post-surgical management for the patient’s mobilization, however, obesity grade I was not a contraindication to continue with surgery.
As final recommendations we have to emphasize the importance of multidisciplinary approach to obtain the best results with this patients, the importance of intraoperative frozen study for margins to confirm an R0 resection, the use of advanced imaging techniques, such as 3D reconstructions, to help visualize the tumor and surrounding anatomy which can significantly improve the surgical planning, and finally, we recommend selecting prosthetic materials based on patient anatomy, tumor location, and availability, as well as ensuring that the reconstruction materials do not interfere with subsequent radiation therapy.
Footnotes
Acknowledgements
A special acknowledgement to the staff of this illustrious journal for the opportunity to publish this research in this prestigious journal, and to contribute to the scientific and surgical community through it.
Author contributions
-Santiago A. Endara, MD: conceptualization, methodology, validation, formal analysis, review and editing, visualization. -Gerardo A. Davalos, MD: validation, formal analysis, visualization. -Armando Serrano, MD: validation, formal analysis, review and editing, visualization. -Patricia Ponton, MD: validation, formal analysis, visualization. -Santiago Muñoz-Palomeque, MD: methodology, formal analysis, investigation, data curation, original draft preparation, visualization. -Cynthia D. Lopez, MD: validation, formal analysis, visualization. -G. Ariel Diaz, MD: investigation, data curation, visualization
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data availability statement
The data that support the findings of this study are available from the corresponding author, Santiago A. Endara, MD, upon reasonable request.

