Abstract
The transformation of a benign phyllodes tumor (PT) into a malignant PT and/or carcinoma is extremely uncommon. We present a case of a 66-year-old female with a huge mass on the left breast which was successfully removed by surgical resection. The pathological diagnosis was infiltrating lobular carcinoma with pure rhabdoid features and the malignant transformation of a benign phyllodes tumor. The first time this rare case was reported, it is demonstrated a special phenomenon through the synchronous transformation of PT grades and the carcinomatous transformation of PT.
Introduction
The transformation of a benign phyllodes tumor (PT) into a malignant PT is rare, particularly the synchronous progression from benign, to borderline and malignant. To date, this phenomenon is not yet reported in the medical literature. Only some previous reports showed a malignant transformation with the individual history of the resection of a benign PT (metachronous transformation).1–6 Simultaneously, development of mammary carcinoma within a PT, especially the synchronous phenomenon, is exceedingly rare. Only few reports of metachronous transformation and synchronous phenomenon (the development of a carcinoma within a PT) have been described in the literature.7–12
Rhabdoid-like cells are characterized by an abundant eosinophilic cytoplasm with eccentric vesicular nucleus and prominent acidophilic nucleolus. Malignant rhabdoid tumor is a very aggressive form of tumor first described as a variant of Wilms’ tumor of the kidney in 1978. 13 Rhabdoid tumors of the extrarenal settings were later reported in many tissues including the liver, soft tissue, the central nervous system, the gastrointestinal tract, thyroid, and lung.14–17 In breast cancer, rhabdoid-like morphology has been reported in the myoepithelial component of epithelial myoepithelial carcinoma. 18 In 2001, Mogotlane et al. reported the occurrence of rhabdoid cells in two cases of infiltrating breast duct carcinomas of no specific type. 19 Herein, we report the clinicopathological characteristics of the first case of infiltrating breast lobular carcinoma of LUMB HER2 + subtype with rhabdoid feature coexisting with the concurrent malignant transformation of phyllodes tumor in a Vietnamese woman.
Case Presentation
This is a 66-year-old postmenopausal, non-smoking female patient, who was admitted to the Vietnam National Cancer Hospital in December 2022 with a huge mass on the left breast. Ten months before presentation, she discovered a small lump by self-palpation, and it was progressively increasing in size. Clinical examination revealed a painless, mobile mass occupying most of the whole left breast, with absent bloody nipple discharge and no abnormal enlarged left axillary lymph node. Ultrasound scan and mammography of the left breast was done which demonstrated a defined margin, and a strongly irregular hypoechoic mass that was 66x31x100 mm in size (Figure 1(a) and (b)). Microcalcification was not found. An ultrasound diagnosis of BIRADS IVb was made. The ultrasound results didn’t show any abnormal left axillary lymph nodes. An ultrasound guided core needle biopsy was done and taken to pathology laboratory for histopathological diagnosis. A diagnosis of phyllodes tumor grade I was made. Left mammography pictures show that has a mass with increased density, lobulated edges, well – demarcated margins, and rough calcifications, without infiltrating signs. (a) Left Mediolateral Oblique (LMLO) View; and (b) Left Craniocaudal (LCC) view.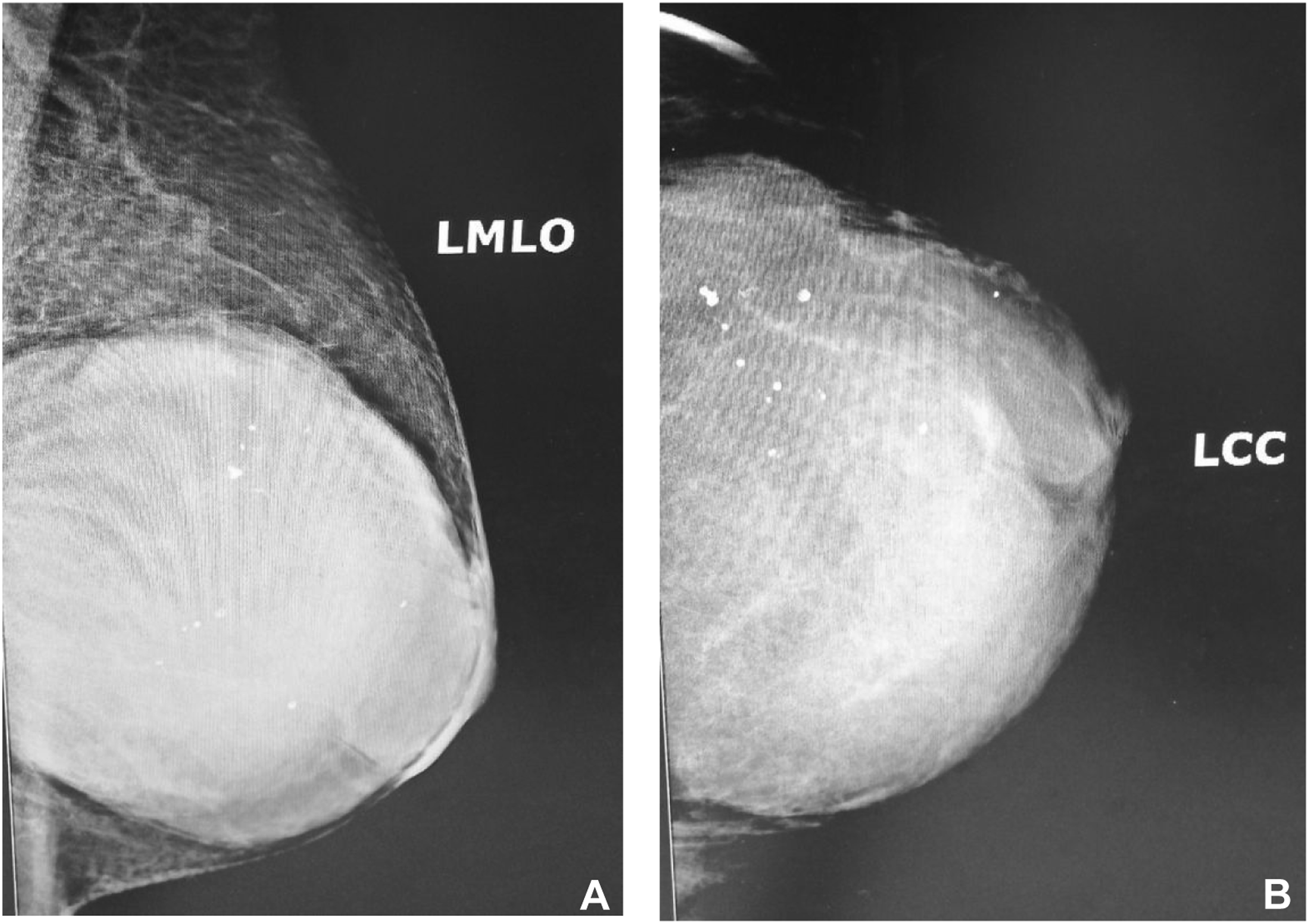
Pathology and Immunohistochemistry
Following the diagnosis of benign phyllodes tumour, a left mastectomy was done and taken to the pathology laboratory. A left breast was received and transection showed a well-demarcated solid grey-white firm mass.
Microscopically, two distinct tumor components - carcinoma and PT with overlapping regions were seen (Figure 2(a)). Carcinomatous cells had a rhabdoid appearance. The tumoral cells were in sheets, nests and sometimes singly disposed but clusters and tubules with occasional foci of few residual ducts were also seen (Figure 2(b) and (c)). Areas with malignant PT features showed bland spindle cells with mild nuclear atypia, pale eosinophilic cytoplasm and brisk mitosis with up to 8 mitoses per 1 high field power (Figure 2(d)). Photomicrographs of H&E stained slides show (a) Three grades of PT and, spectrum of carcinomatous transformation from benign (1st star) to borderline (2nd star) to malignant (3rd star), malignant transformation of one duct (Discontinuous lines and 4th star) and invasive carcinoma (5th star) (Magnification X5). (b) Magnification of the 4th star of Figure 1(a): Carcinoma cells (3rd arrow and star) tended to be located between the outer myoepithelial layer (1st arrow) and the inner luminal epithelial cells (2nd arrow) (Magnification X10). (c) Invasive lobular carcinoma with rhabdoid feature show cells are characterized by abundant eosinophilic cytoplasm with globular paranuclear eosinophilic inclusions and with an eccentric vesicular nucleus (Magnification X20). (d) Malignant spindle cell sheets of phyllodes tumor (Magnification X40).
A diagnosis of carcinoma was considered with myoepithelial carcinoma as a differential. Immunohistochemitry was done which showed diffuse expression of pan cytokeratin and CK7, weak positivity for CD117, ER, PR and HER2 positivity but was negative for CK20 and CD56. Ki67 index was shown to be 10%. The malignant epithelial cells had a IHC HER2 score of 2 plus was tested by Dual-ISH to identify amplification of the HER2 gene. The nuclei of invasive cancer cells show the HER2 gene amplification. Patient was classified in molecular subgroup as luminal B HER2+ (LUMB HER2+), according to St Gallen 2013. E-cadherin shows the scattered and weak activity. The rhabdoid – like malignant epithelial cells were moderately expressed for myogenin. CD117 was also positive for benign ductal epithelium in normal breast tissue and for benign mammary ducts entrapping in carcinomatous tissue, as should be concise.
Meanwhile, the benign myoepithelial cells are only immunoreactivity for p63 (show in Figure 3(d)). For PT tissues almost were negative for all of the above markers. Ki67 marker was expressed in less 1.0 and 5.0%, and 60.0% of benign, borderline and malignant phyllodes tumors (Figure 3(c)), respectively. The rhabdoid cells moderately expressed myogenin. Similarly, benign phyllodes tumor is negative for CD56 but this marker is expressed in moderate or strong levels of borderline or malignant phyllodes tumors, respectively (Figure 3(b)). Areas demonstrating PT components were for all the aforementioned immunomarkers. Photomicrographs of IHC stained slides show (a) The pancytokeratin positive cancer cells (1st arrows) and negativity of malignant PT (1st star) (Magnification X20). (b) CD56 staining showed the various expression of three grades of PT: negativity of begnin PT (1st star), moderate expression of borderline areas (2nd star) and strong positive malignant PT (3rd white stars) (Magnification X10). (v) Ki67 index shows the difference of malignant PT and invasive carcinoma (separated by red dotted line) (Magnification X20). (d) p63 was expressed for the begnin myoepithelial cells and negativity of in situ carcinoma (red stars) and malignant PT (black star) (Magnification X10).
Based on the histopathological characteristics and the combined IHC results, a diagnosis of a synchronous tumor consisting of an infiltrating globular carcinoma with pure rhabdoid features (stage1A; pT1bN0) and a malignant phyllodes tumor (stageII; pT1N0, G2) was made.
Treatment and follow up
Patient was only operated onto removing the whole left breast with tumor. The patient declined additional treatment but is on regular follow-up. She is doing well with no recurrence.
Discussion
The transformation of a benign PT into a malignant one is very rare, particularly the transition from benign to borderline and malignant at the same time. To date, this phenomenon is not reported in the medical literature. Only Pornchai et al. reported a malignant transformation of a benign PT 36 months after resection, the similar reports have been recorded previously.1–6 Most studies had described the mechanism of the malignant transformation. It had been suggested that mutation of residual benign PT cells caused the malignant transformation. 6 Most studies to date focused on stromal alterations and showed recurrent copy number gains and losses at +1q, −13q, −6q, +5, and −10p.20–22 It is believed that the growth of tumor comes from the epithelial–stromal interaction process including Wnt-APC-Beta-catenin pathway and estrogen receptor pathway. 23 Genetic aberrations have been consistently demonstrated in PTs with increasing frequency from benign to borderline to the malignant PTs. Molecular studies have shown that the increase in the expression of Beta-catenin or estrogen receptor is associated with benign PT, but if there is a decrease in the expression, it is associated with malignant PT.24,25 In malignant PT, the growth depends on the stromal component, which is molecular profile, and gene mutation like other types of cancer includes p53, Ki67, CD117 (c-kit), EGFR, and CD34. 1 Moreover, the molecular studies have shown that the increase in the expression of Bcl2, Beta-catenin or estrogen receptor is associated with benign PT, but if there is a decrease in the expression, it is associated with malignant ones.24,25 In this case, no molecular studies were carried out.
Malignant transformation of epithelial elements of PT is very rare with only 39 cases reported in the literature. 7 However, this malignant transformation must be displayed within the PT, not the adjacent breast tissue. While, to date, the phenomenon of the synchronous transition from benign, to borderline and malignant has not been reported in the medical literature. Breast carcinoma was noted in the adjacent breast tissue or far from the PT, indicating a synchronous relationship between the carcinoma and PT. In our case, the patient’s tumor was a synchronous carcinomatous transformation of PT, with the carcinomatous components showing a closed relationship to the epithelial component of the PT.
These rhabdoid cells are characterized by abundant eosinophilic cytoplasm with globular paranuclear eosinophilic inclusions, an eccentric vesicular nucleus, and a prominent acidophilic nucleolus. They were first described as a variant of Wilms’ tumor of the kidney in 1978. 13 Histogenetically, these cells display both mesenchymal (vimentin) and epithelial (cytokeratin) marker positivity, 19 and they stain positively for myogenin and desmin. 18 Our findings of positive myogenin are consistent with these histopathology and IHC findings. In 2001, Mogotlane et al. reported the occurrence of rhabdoid cells belong to two infiltrating breast duct carcinomas of no specific type. 19 In 2020, Grenier et al. reported the first case of breast epithelial-myoepithelial carcinoma with rhabdoid-like characteristics. 18 In the breast, tumors with rhabdoid morphology include primary breast rhabdomyosarcoma, phyllodes tumor with heterologous differentiation, and metaplastic carcinoma with mesenchymal differentiation, one epithelial-myoepithelial carcinoma of Grenier et al.’s report, two carcinomas of no specific type,18,19 while tumors with rhabdoid-like morphology only included invasive lobular carcinoma up to now.
A common finding among the many various types of tumors displaying a significant (greater than 10%) population of rhabdoid cells seem to be the adverse clinical outcome. 19 Mogotlane et al.’s report showed a patient with approximately 60% of rhabdoid cells had aggressive disease wherein 7 months postmastectomy with clear margins of excision, there was tumor recurrence and lung metastases. 19 Therefore, besides identifying tumor cells with rhabdoid features, it is also very important to account the percentage of this cancer cell component.
Considering the origin of these tumor cells, there could also exist a multipotential stem cell capable of both epithelial and mesenchymal differentiation accounting for this phenotype. This hypothesis was suggested by Wick et al. in their study of the origin of carcinosarcoma. 26 The histopathology of this tumor showed tumor cells located between the luminal epithelium and the myoepithelial cells of the mammary ducts. Immunohistochemical staining with p63 (positive for myoepithelial cells) and CD117 (positivity of benign luminal cell) also demonstrated the location of tumor cells. CD117 showed scattered positivity of carcinomatous cells and histologically, it is possible that these are reproductive cells (breast cancer stem cells). However, further investigations are studied, especially molecular biology researches, to confirm whether rhabdoid tumor cells are exactly breast cancer stem cells.
Conclusion
This exceedingly rare case revealed some distinctive features notably carcinoma with the purely rhabdoid feature which should be considered as a separate histopathological type; A distinction should be made between the concurrent carcinomatous component in the breast adjacent to PT, carcinomatous transformation of PT and synchronous transformation of PT grades. It is very important to be aware of this heterogeneous cancer type in order to provide the accurate information for making an appropriate treatment decision and prognosis.
Footnotes
Author Contributions
Tu Anh Do participated directly in diagnosis, treatment, and examination of the patient, performed literature review, and assisted in drafting of the components of the case reports, and formatting the presented material. He should be considered the first author. Chu Van Nguyen wrote part of the diagnostic consultant of the HE stains and immunohistochemical staining, literature review, and assisted in drafting of the components of the case report, and assisted in formatting the presented material. Nhung Thi Mai and Duong Ngoc Nguyen took part in the pathological techniques of the patient, and assisted in literature review. Khoa Hong Pham and Hung Van Pham took part in the diagnostic and treatment consultant and, assisted in literature review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability statement
All data analysed during this case report are included in this article. Further enquiries can be directed to the corresponding author.
