Abstract
Urachal cancer is a rare genitourinary malignancy that arises from the embryologic remnant of the urachus. The malignancy is considered to be aggressive, with no clear consensus on appropriate management for advanced disease. Although traditionally considered to be related to bladder cancer given its embryologic origin, several next generation sequencing studies have revealed the genomic profile of this genitourinary malignancy most closely resembles colorectal cancer. Moreover, these studies have identified potentially actionable mutations including EGFR, KRAS and MET. In addition, recent data suggests that immunotherapy may benefit some patients with advanced urachal cancer. Nonetheless, continued research is warranted to better understand how to treat this rare genitourinary cancer.
Introduction
Urachal cancer is a rare genitourinary malignancy which arises from the fibrous remnant between the fetal bladder and allantois. 1 Obliteration and involution of the urachus occurs at the end of gestation or the first few days of birth. An autopsy series has suggested that in nearly one third of individuals, the remnant of the urachus may persist. 2 Failure of involution of the urachus can lead to a multitude of potential pathologic diseases which were first described by R. Campbell Begg in 1930. 3 Individuals with urachal cancer may present with gross or microscopic hematuria, dysuria, or abdominal pain, whereas others may present with non-specific symptoms such as fatigue and unintentional weight loss. 4 Given the rarity of this genitourinary cancer, there is a lack of prospective clinical data on the appropriate management of localized and advanced urachal cancer. However, several recent studies evaluating the genomic profile of urachal cancers have identified potential newer therapeutic approaches in managing advanced urachal cancer.5–8 In this review, we will discuss current treatment approaches for localized and advanced urachal cancer, genomic profiling data on urachal cancers, and the use of immunotherapy and targeted therapies in managing urachal cancer.
Epidemiology
Urachal cancer has historically been categorized as a malignancy of the bladder, and accounts for less than 1% of bladder cancers. 9 Several population-based studies have demonstrated a higher incidence of urachal cancer in men and during the fifth decade of life. 10 Based off data from the Surveillance, Epidemiology and End Results (SEER) database, the most common histology is adenocarcinoma which represented over 80% of cases, followed by less frequent histologies such as squamous cell carcinoma and sarcoma. 11 The same cohort of 420 patients found that nearly 60% of patients presented with locally advanced or metastatic disease (stage III and stage IV disease, respectively) per American Joint Committee on Cancer (AJCC) staging. The study also found that the median overall survival (OS) was 57 months and median cancer-specific survival (CSS) was 105 months. A retrospective, population-based study by the California Cancer Registry including 315 patients demonstrated that individuals with localized disease lived longer (178 months) than those with locally advanced (83 months) or metastatic disease (19 months), and found no differences in survival based off sex, ethnicity, or neighborhood socioeconomic status. 10
Although there is no standard accepted staging system, the two most commonly used staging systems including the Sheldon staging system (stages I–IV) and Mayo staging system (stages I–IV). 12 Per the Sheldon staging system, stage I disease is defined as cancer confined to the urachal mucosa, stage II disease is defined as invasion confined to the urachus itself, stage III disease (A–D) includes local extension, and stage IV disease is metastatic to lymph nodes (A) or distant sites (B). Similarly, the Mayo staging system defines stage I disease as being confined to the urachus and/or bladder, stage II disease extending beyond the muscular layer of the urachus and/or bladder, stage III disease infiltrating regional lymph nodes, and stage IV disease infiltrating non-regional lymph nodes or distant sites.
Current treatment approaches
Given a lack of large prospective, randomized controlled trials, there is no clear consensus on the management of locally advanced or metastatic urachal cancer. In fact, the National Comprehensive Cancer Network (NCCN) and European Association of Urology (EAU) do not have specific recommendations on treatment approaches for urachal cancer. However, the Canadian Urological Association (CUA) and Genitourinary Medical Oncologists of Canada released a consensus statement on managing urachal cancer. 13
Case studies from single centers have suggested that surgical resection with partial or radical cystectomy with en bloc resection of the umbilicus and urachal remnant are preferred in the setting of localized disease.2,12,14,15 Some single center and multi-institutional studies have also suggested the use of regional lymphadenectomy for individuals who have locally advanced disease. 16 However, retrospective multi-center data including 163 patients from Ke et al. did not show any correlation between lymphadenopathy with risk of recurrence or prognosis. 17 Based off level 4 evidence (case-control studies or case series), a consensus statement from Canadian Urological Association (CUA) and Genitourinary Medical Oncologists of Canada recommended en block surgical resection of the umbilicus, urachal ligament and partial cystectomy with pelvic lymphadenopathy as the preferred treatment approach. In addition, the group recommended radical cystectomy with en bloc resection for umbilicus and urachal ligament in cases where partial cystectomy was not feasible. Although there are case reports of use of neoadjuvant chemotherapy with or without adjuvant chemotherapy, there is no clear consensus on the role of either for localized disease. 18
There is currently no consensus on the role of radiotherapy in treating urachal cancer. Urachal tumors are generally considered to not be radiosensitive. Per the consensus statement from CUA and Genitourinary Medical Oncologists of Canada, radiation therapy is recommended for consideration after surgery in the setting of positive surgical margins and second-look surgery is not feasible. 13 In addition, the consensus statement which is based off expert opinion (evidence level 5), radiotherapy is a consideration as palliative treatment in individuals with advanced disease.
The appropriate chemotherapy regimen for treating metastatic urachal cancer remains unclear. Based off case series and institutional studies, the most commonly employed chemotherapy regimens include either cisplatin and/or 5-fluorouracil (5-FU). A case series by Siefker-Radtke et al. reported a response rate of 33% among 20 patients treated with either cisplatin or 5-FU based chemotherapy regimens. In a prospective trial by Galsky et al. evaluating 20 patients with non-transitional cell carcinoma of the urothelial, 6 patients had urachal cancer and were treated with cisplatin, paclitaxel, and ifosfamide. The study reported a response rate of 35% with median survival being 24.8 months. 19 While commonly used in treating metastatic colorectal cancer, the combination regimen 5-FU, leucovorin, and oxaliplatin (FOLFOX) has also been studied and used in treating metastatic urachal cancer. 20 In a case series involving 5 patients, 40% (2 of 5) showed an objective response including 1 with complete response and 1 with partial response. 21 A meta-analysis of 1010 cases suggested that 5-FU containing chemotherapy regimens were more efficacious compared with cisplatin-based regimens in terms of radiographic response rates; however, combination regimens including both therapies appeared to have the lowest rates of progression. 4 Given the modest response rates seen with these aforementioned chemotherapy regimens, there has been interest in evaluating the genomic profile of urachal cancers to better understand which regimens may be most effective in the metastatic setting and to identify any possible oncogenic driver mutations that correlate with targeted therapies.
Genomic profiling of urachal cancers
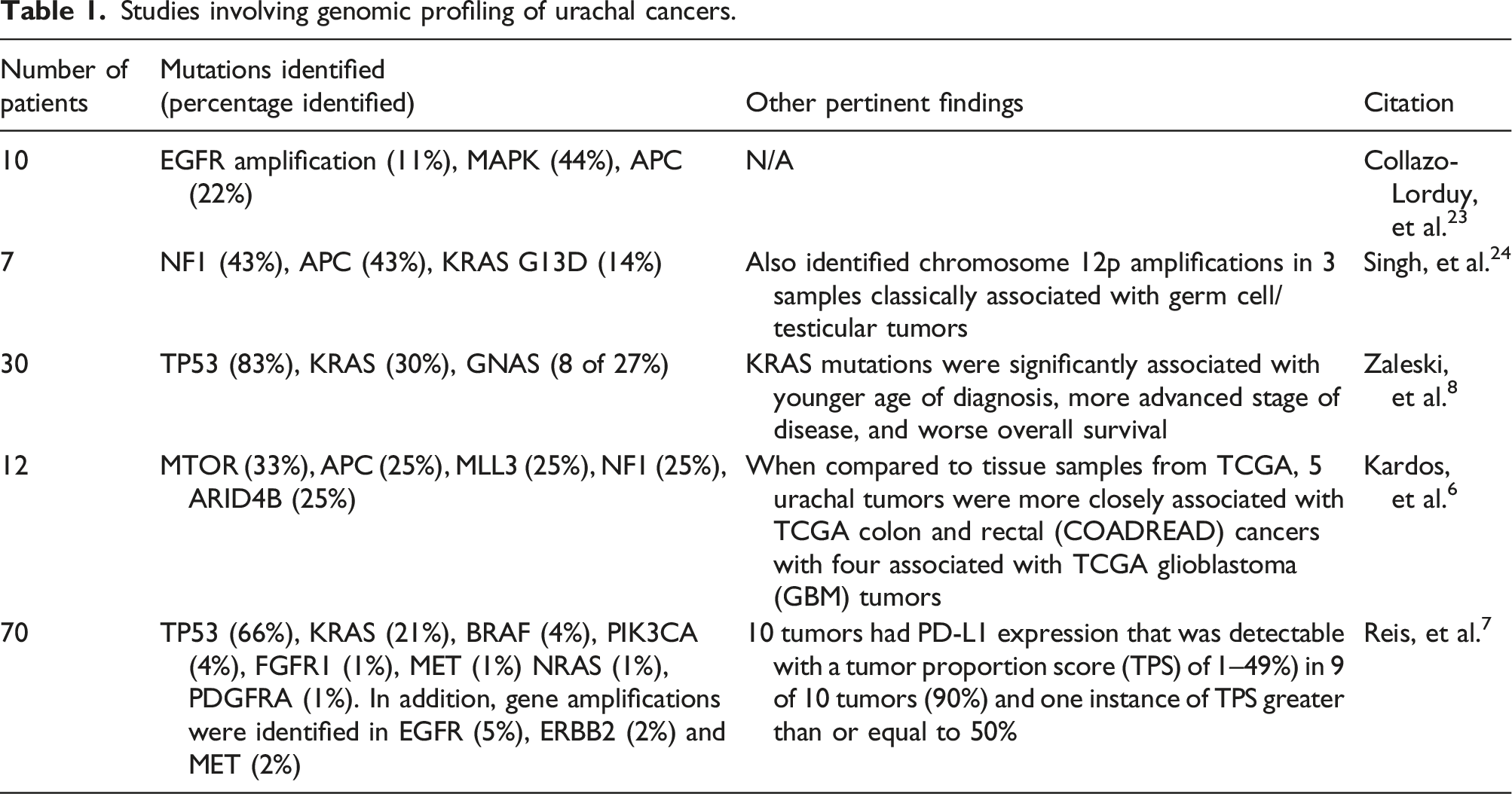
With the advent of next generation sequencing readily available in the clinical setting in the late 2010s, more individuals have undergone evaluation for possible targetable mutations. 22 In 2016, Collazo-Lorduy et al. reported on genomic sequencing for 10 patients with metastatic urachal cancer with mutations including EGFR amplification found in 1 patient, mutations in the mitogen-activated protein kinase (MAPK) pathway in 4 of 9 samples, and mutations in the adenomatous polypsosi coli (APC) gene in 2 of 9 samples. 23 A case series by Singh et al. of 7 patients with metastatic urachal cancer identified neurofibromin 1 (NF1) mutations as part of the MAPK pathway in 3 patients, APC mutations in 3 patients, and KRAS G13D mutation in 1 patient. The study also identified chromosome 12p amplifications in three tumor samples which is a finding classically associated with germ cell/testicular tumors (Table 1). 24
A single center study by Zaleski et al. found that among 30 cases of urachal cancer, next generation sequencing revealed mutations in all samples with TP53 (25/30) being the most commonly mutated gene followed by KRAS (9/30) and GNAS (8/30). 8 The study also found that KRAS mutations were significantly associated with younger age of diagnosis, more advanced stage of cancer, and poorer overall survival.
While next generation sequencing has identified potentially actionable mutations, a study by Kardos et al. of 12 urachal tumors suggested that the molecular profile of urachal cancer resembles colorectal cancer as opposed to bladder cancer. 6 In their first-of-its kind study, whole transcriptome profiling by RNA sequencing of 12 samples was conducted. The study revealed nearly all tumors had TP53 inactivation, with several other gene mutations at high frequencies including MTOR (33%), APC (25%), MLL3 (25%), NF1 (25%), ARID4B (25%). The study reported less frequency of mutations traditionally associated with bladder cancer such as fibroblast growth factor receptor 3 (FGFR3), ARID1A, KDM6A, and CDKN1A. In addition, the study identified 3 tumors (25%) with inactivating mutations associated with microsatellite instability including MSH2 and MSH6, which was associated with higher mutation burdens and insertion/deletion rates. Finally, the study compared the genomic profiling of these 12 tumors with transcription analysis of tumors from 12 different tissues of origin as part of The Cancer Genome Atlas (TCGA). Five urachal tumors were more closely associated with TCGA colon and rectal (COADREAD) cancers with four associated with TCGA glioblastoma (GBM) tumors. Embryologic development of the cloaca dividing into anterior and posterior portions, with the anterior portion becoming the urogenital sinus (which leads to the allantois and development of the urachus) and the posterior portion becoming the rectum may explain these findings.6,25
The largest known study evaluating targetable mutations in urachal cancer involved 70 patients as reported by Reis et al. 7 The specimens underwent next generation sequencing, as well as immunohistochemical analyses for DNA mismatch repair proteins as well as PD-L1 status. The study detected a total of 73 mutations and 4 gene amplifications among the 70 analyzed specimens with 55 (79%) of samples harboring at least one genomic alteration. The most common mutations in order of frequency were as follows: TP53 (66%), KRAS (21%), BRAF (4%), PIK3CA (4%), FGFR1 (1%), MET (1%) NRAS (1%), PDGFRA (1%). In addition, gene amplifications were identified in EGFR (5%), ERBB2 (2%) and MET (2%). No microsatellite instability-high (MSI-high) tumors were detected. With regards to PD-L1 testing, 10 tumors had PD-L1 expression that was detectable with a tumor proportion score (TPS) of 1–49%) in 9 of 10 tumors (90%) and one instance of TPS greater than or equal to 50%. Similar to the study by Kardos et al, the authors concluded that the genomic profile of these urachal cancers most closely resembled the profile of colorectal cancers. This finding may explain why urachal cancer is responsive to 5-FU based chemotherapy regimens, such as FOLFOX, which are used for treating colorectal cancers.
Immunotherapy
Immunotherapy has drastically altered the treatment landscape of several genitourinary cancers including urothelial carcinoma and renal cell carcinoma. 26 Several case series and case reports have discussed the use of immunotherapy in treating advanced urachal cancer. In the case series by Kardos et al. identifying microsatellite instability due to mutations in MSH2 and MSH6, one patient was treated with anti-programmed death-ligand 1 (anti-PD-L1) antibody atezolizumab. On initial imaging, the patient appeared to have disease progression that was later attributed to tumor flare from immunotherapy. On repeat imaging at the second time interval on treatment, the patient was noted to have regression of two metastatic sites (two lung nodules) but increase in the size of a left hilar node with associated necrosis.
A case report by Zheng et al discussed the case of a patient who developed pulmonary metastases while on chemotherapy for recurrent localized disease. 27 The patient was treated with 3 cycles of gemcitabine plus nedaplatin, and due to no reduction in tumor size, an anti-PD-L antibody was added although the immunotherapy drug name is not specified in the report. The patient was found to have stable disease on repeat imaging after 3 cycles of combination chemotherapy plus immunotherapy, but ultimately developed disease progression after 5 cycles of treatment.
Targeted therapies
Given the identification of potentially actionable driver mutations, several institutions have reported case reports of patients with metastatic urachal cancer received treatment with targeted therapy. In the previously discussed case series by Collazo-Lorduy, one patient’s tumor was identified as harboring EGFR amplification and wild-type KRAS. This patient was ultimately treated with the EGFR monoclonal antibody cetuximab as monotherapy, and experienced a partial response that lasted for at least 8 months. 23 Retrospective, multi-institutional data from France demonstrated median overall survival of 19 months (95% CI, 7 – N/A) in 6 patients who were treated with anti-EGFR monoclonal antibodies in either the first- or second-line treatment settings. 16
A phase I trial in Japan evaluated the efficacy of tepotinib, an inhibitor of mesenchymal-epithelial transition (MET) factor. 28 In the study, a total of 12 patients were treated including 1 patient with urachal cancer who had stable disease of at least 12 or greater weeks in duration. There are no other known studies in the literature reporting the use of targeted therapies in treating advanced urachal cancer.
Future directions
Given the modest responses seen with chemotherapy, there is an urgent need to develop systemic therapies with improved outcomes for individuals with recurrent or metastatic urachal cancer. A review of ongoing clinical trials reveals only one ongoing prospective phase II study (NCT04611724) evaluating modified FOLFIRINOX (5-FU, leucovorin, irinotecan, and oxaliplatin) that is being conducted at single center in South Korea as of November 2020.
Studies involving genomic profiling of urachal cancers.
Conclusion
Urachal cancer has classically been described as an aggressive genitourinary malignancy. Although recent larger datasets suggest otherwise, individuals with metastatic disease have limited treatment options which historically has been chemotherapy. Several case reports as discussed have demonstrated partial responses or stable disease with the use of targeted therapies including EGFR antibodies, MET inhibitor, and immunotherapy with immune checkpoint inhibitors. Although there are no known ongoing studies specifically evaluating targeted therapies or immunotherapy in treatment of urachal cancer likely owing to the rarity of this malignancy, urachal cancer may possibly be studied in basket trials depending on mutational status found on next generation sequencing. Continued research into improved therapeutics in this rare malignancy are warranted.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ARK declares the following interests: Stock and Other Ownership Interests: ECOM Medical; Consulting or Advisory Role: Exelixis, AstraZeneca, Bayer, Pfizer, Novartis, Genentech, Bristol Myers Squibb, EMD Serono, Immunomedics, and Gilead Sciences; Speakers’ Bureau: Janssen, Astellas Medivation, Pfizer, Novartis, Sanofi, Genentech/Roche, Eisai, AstraZeneca, Bristol Myers Squibb, Amgen, Exelixis, EMD Serono, Merck, Seattle Genetics/Astellas, Myovant Sciences, Gilead Sciences, and AVEO; Research Funding: Genentech, Exelixis, Janssen, AstraZeneca, Bayer, Bristol Myers Squibb, Eisai, Macrogenics, Astellas Pharma, BeyondSpring Pharmaceuticals, BioClin Therapeutics, Clovis Oncology, Bavarian Nordic, Seattle Genetics, Immunomedics, and Epizyme; Travel, Accommodations, Expenses: Genentech, Prometheus, Astellas Medivation, Janssen, Eisai, Bayer, Pfizer, Novartis, Exelixis, and AstraZeneca. DJB has received consulting fees from Seagen.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Contributorship
DJB – Conceptualization, Investigation, Writing – Original Draft, Writing – Review & Editing. ARK – Conceptualization, Writing – Original Draft, Writing – Review & Editing.
