Abstract
De novo renal transplant carcinoma, especially in the context of bilateral renal carcinoma, is rare and often presents as small, low-grade papillary renal cell carcinoma (RCC). There is currently no consensus or effective treatment for advanced metastatic RCC after kidney transplantation. A 40-year-old man developed de novo renal transplant carcinoma with venous thrombus and lung metastases 20 years after transplantation. The patient underwent cytoreductive nephrectomy followed by sequential treatment with tyrosine kinase inhibitors (TKI) and anti-PD-1 monoclonal antibodies. After 2 years, the patient showed excellent graft function with no evidence of cancer progression. Despite subsequent graft failure, the disease remained controlled for more than 2 years and the patient survived for more than 3 years, which was significantly longer than the typical survival of 10 to 20 months in patients with advanced kidney cancer. The results suggest that combining cytoreductive nephrectomy with TKI and anti-PD-1 therapy may significantly prolong survival in patients with renal allograft carcinoma.
Keywords
Background
The incidence of renal cell carcinoma (RCC) among kidney transplant recipients has increased as a result of long-term immunosuppression therapy. Solid organ tumors, lymphomas, and skin non-melanoma were reported in 10.8% of patients who received a renal transplant, with renal and lung cancers being the most common solid malignancies. 1 Most RCCs detected in transplant recipients are de novo neoplasms in the native kidney; however, RCCs can also emerge in the donor kidney as recessive tumors present in the donor organ prior to transplantation.2,3 A lack of relevant studies means that the systemic treatment of metastatic RCC after renal transplantation poses a challenge, and objective treatment evaluations are necessary to address this issue. The current case report outlines the various therapeutic options that have been demonstrated to yield favorable short-term outcomes in renal transplant recipients diagnosed with metastatic RCC.
Case presentation
A 20-year-old man with end-stage renal disease underwent allograft kidney transplantation in 2000, 3 months after commencing hemodialysis, with one kidney placed in the left lumbar region. The patient was maintained on a combination of prednisone, cyclosporine, and mycophenolate after transplantation. No imaging examinations were conducted during subsequent routine follow-up. The patient presented in September 2020 with a history of recurrent episodes of hematuria (Figure 1). Contrast-enhanced computed tomography (CT) demonstrated bilateral malignant renal masses (left 8.5 cm × 4.3 cm; right 3.8 cm × 3.5 cm) accompanied by tumor thrombi in the bilateral renal veins and inferior vena cava (Figure 2(a)). Further positron emission tomography-CT suggested involvement of the adrenal gland and vertebra (L4), ischial metastases, and abdominal lymphadenopathy (Figure 2(b)). Chest CT revealed multiple nodules in both lungs, indicative of metastatic disease (Figure 3). The imaging results provided compelling evidence of malignancy. A percutaneous biopsy was performed under ultrasound guidance, and subsequent histological assessment disclosed a poorly differentiated clear cell RCC. Laboratory investigations, including blood and blood biochemistry tests, revealed no abnormalities. The patient’s Eastern Cooperative Oncology Group score was 1, which led to a diagnosis of advanced RCC, grade cT3N1M1, based on the above results.

Timeline of case. OS, overall survival; SD, stable disease; PR, partial response.
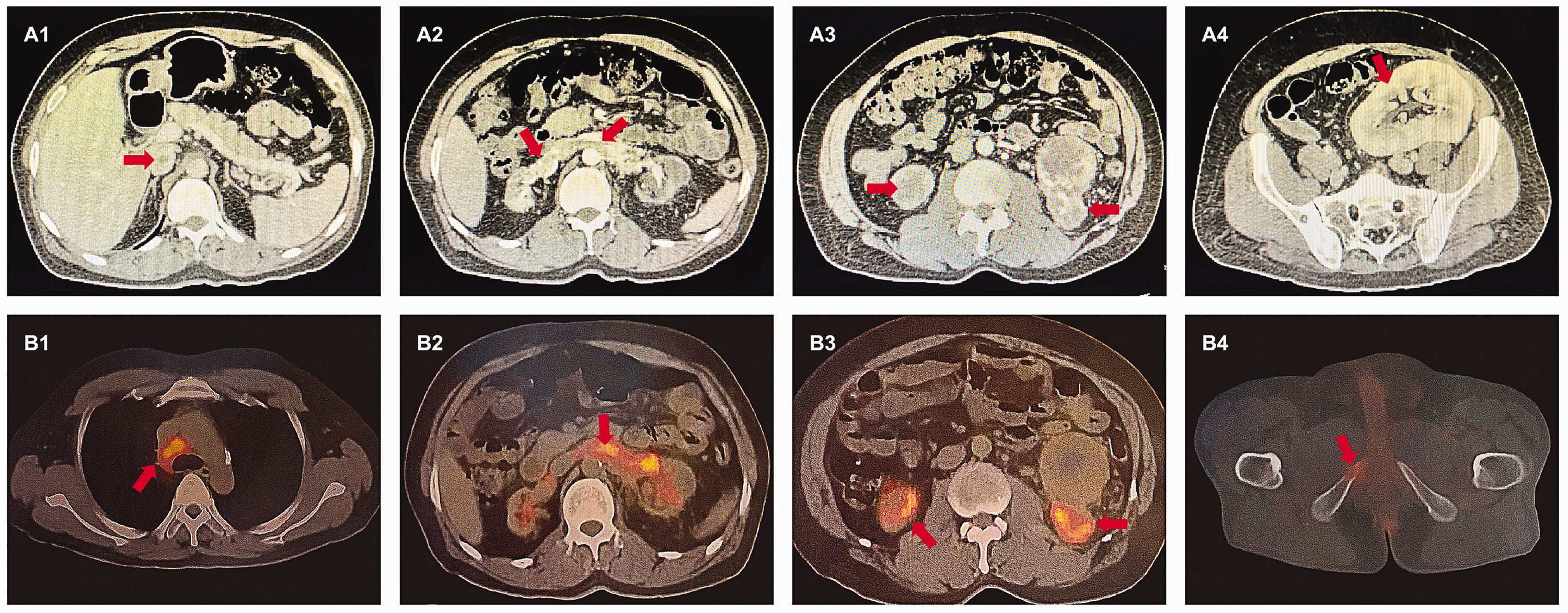
Preoperative abdominal computed tomography (CT) and positron emission tomography (PET)-CT images. (a1 and a2) Abdominal CT-enhanced portal phase scan showing filling defects in the inferior hepatic segment of the inferior vena cava and the lumen of both renal veins, suggesting cancerous emboli. (a3) Abdominal CT-enhanced parenchymal phase scan showing bilateral autologous kidney atrophy, visible as a significantly enhanced mass shadow, partially protruding outside the kidney contour, with uneven density within it and the presence of liquefied necrosis. (a4) CT-enhanced parenchymal phase scan showing transplanted kidney in the left iliac fossa, with compensatory enlargement and clear corticomedullary demarcation. (b1) PET/CT fusion showing enlarged lymph nodes with increased fluorodeoxyglucose (FDG) uptake in the mediastinal 4 R region, SUVmax 8.4. (b2) PET/CT fusion showing widened renal veins on both sides with increased FDG uptake in the luminal strips and nodules, SUVmax 7.3. (b3) PET/CT fusion showing bilateral autologous renal atrophy with increased FDG uptake in the mass shadow, SUVmax 9.3. (b4) PET/CT fusion showing osteolytic destruction of the right sciatic bone with increased FDG uptake, SUVmax 3.7.

Chest computed tomography images. (a1-4) 8 October 2020: multiple nodular metastases in the lung, the larger one located in the anterior segment of the upper lobe of the left lung. (b1-4) 10 October 2020: CT was repeated after surgery showing multiple metastases in the lung, larger than before. (c1-4) 10 February 2021. (d1-4) 31 March 2021: CT showing multiple metastases in the lung, fewer and smaller than before. (e1-4) 10 October 2022: CT 16 months after pazopanib showing multiple metastases in the lung larger than before. (f1-4) 10 February 2023: CT after the patient stopped pazopanib and switched to acitinib, showing metastases in the lung larger than before. (g1-4) 2 March 2023.
The predictive risk, according to the standards set by the International Database Consortium for Metastatic Renal Cell Carcinoma (IMDC), was identified as low and surgery was therefore recommended, comprising cytoreductive nephrectomy and removal of the tumor thrombi in the renal vein and inferior vena cava. The patient provided consent for treatment. Pathological examination confirmed the diagnosis of clear cell RCC, and immunohistochemical staining demonstrated programmed death-ligand 1 (PD-L1) expression (Figure 4).

Patient’s specimens and pathology. (a) Resected kidney and renal vein and inferior vena cava cancer thrombi. (b) Kidney section. (c) Hematoxylin–eosin staining pathology and (d) immunohistochemistry was positive for programmed cell death 1 ligand 1.
Genetic profiling was carried out using a custom panel of 539 genes, to assess the presence of potentially relevant somatic and pathogenic germline variants (Table 1). Sequencing analysis indicated no evidence of pathogenic or likely pathogenic germline alterations in the examined specimen. Although some somatic alterations of uncertain clinical significance were identified, no findings suggested a potential response to any current Food and Drug Administration-approved therapies.
Genetic profiling.

In view of the increased risk of graft rejection linked to immune checkpoint inhibitors, we proposed a targeted-agent approach. Anti-rejection drug therapy was maintained following surgery, and the patient had full knowledge of the labeled use of the medication. Sunitinib monotherapy was administered orally at a dose of 50 mg/day for 4 weeks, with a 6-week cycle. After the initial follow-up cycle, chest CT scans demonstrated a notable reduction in metastatic lesions, indicative of a partial response. The patient, however, developed a rash on their hands and feet, which persisted over the course of 3 months and became increasingly bothersome for the patient. In light of the patient’s prior response to the targeted agent, we recommended the alternative targeted drug, pazopanib (800 mg/day), as a possible alternative treatment option, given its reduced risk of rash-related adverse effects. The patient’s symptoms gradually improved, culminating in complete resolution after four cycles of treatment. A chest CT scan demonstrated that the lung metastasis had not progressed further (Figure 4) and the patient was evaluated with stable disease.
The patient’s hemoptysis had worsened by August 2022, accompanied by the onset of pain and discomfort throughout his body. A chest CT scan demonstrated the presence of multiple metastases in both lungs, which were markedly larger than those in previous scans, indicative of progressive disease. Skeletal CT also revealed the presence of multiple bone metastases in the lumbar spine, iliac bone, and pubic bone. The patient was subsequently administered acitinib (5 mg twice daily) for a period of 6 months, during which time larger lesions in the lungs were identified. The efficacy was evaluated as progressive disease. At this time, the use of a tyrosine kinase inhibitor (TKI) was discontinued and the PD-1 inhibitor tirellizumab was administered, in accordance with the patient’s genetic analysis. Unfortunately, the patient’s renal function declined, necessitating definitive dialysis after two cycles (6 weeks) of treatment. The patient died on 17 October 2023, having survived for a total of 37 months. The patient had survived with the tumor for 3 years following the combination treatment.
Written informed consent to publish the data was obtained from the patient and their family. The reporting of this study conforms to CARE guidelines. 4
Discussion
Advancements in organ transplantation have greatly reduced the early-mortality rate and prolonged the survival period for kidney transplant recipients. Malignant tumors represent an important long-term complication after kidney transplantation, and the increased prevalence of malignant tumors following renal transplantation can be attributed to the interaction of multiple pathological factors, including the immunosuppressed status of renal transplant recipients. The long-term use of immunosuppressive medication is a requisite component of anti-rejection therapy following kidney transplantation; however, this results in the development of long-lasting immunosuppression, which renders patients susceptible to the development of RCC. Farrugia et al. found a greater probability of malignancy-related fatality among 19,103 kidney transplant patients compared with the general population, with renal malignancy being the third most common cause of malignancy-related deaths, accounting for 9.8% of such deaths among kidney transplant recipients. 5 Despite some reports of autologous RCC after renal transplantation, most have involved accounts of cases or small series. Acquired cystic kidney disease and long-term dialysis have been identified as significant risk factors6,7; however, neither of these predisposing factors was evident in the current patient. The processes responsible for the development of RCC in these patients thus remain unclear, and guidelines fail to provide targeted advice for these patients.
Numerous surgeons have expressed reservations about the safety of surgical procedures in patients with immunosuppression; however, patients with autogenous native renal carcinoma following transplantation were shown to have a better prognosis if they received timely surgical intervention,8,9 with a 5-year survival rate exceeding 80% following nephrectomy. 10 Klatte and colleagues suggested that sunitinib, sorafenib, tecataxor, and everolimus were effective options for the treatment of primary renal malignancies in patients unsuitable for surgical resection or with distant metastases.
Cytoreductive nephrectomy is a pivotal aspect of the comprehensive therapeutic regimen for metastatic RCC; however, the debate over its necessity persists. The selection and scheduling of cytoreductive nephrectomy require multidisciplinary and comprehensive analysis from various perspectives. In clinical practice, risk models such as the Memorial Sloan Kettering Cancer Center and IMDC scores are used for patient stratification and treatment, and it is essential to consider patient-specific factors to make the most appropriate treatment decisions.11,12 Several studies have demonstrated improved survival rates following reductive nephrectomy, particularly in patients with a low-risk IMDC score.13–15 Notably, the randomized, controlled CARMENA study was based on data from patients at medium and high risk, and its conclusions may thus not be applicable to patients with a low risk of developing the condition in question. Subgroup analysis of the CARMENA data demonstrated that only high-risk patients met the criteria for the non-inferiority test, resulting in median overall survival. 16 The current literature indicates that radical nephrectomy is an effective treatment for localized RCC in kidney transplant patients; however, studies in patients with distant metastases are limited.3,17 There is presently no evidence to suggest that these scoring systems are unsuitable for application in patients who have undergone kidney transplantation, and it can therefore be concluded that tumor-reducing surgery remains the preferred treatment for this specific population.
Immune checkpoint inhibitors, including those targeting PD-1 inhibitors, have recently emerged as a pivotal area in cancer immunotherapy. Nevertheless, it remains uncertain if PD-1 inhibitors should be administered in organ transplant recipients with tumors, given that their activated T cells can exert effects on allogeneic antigens from the donor, rather than solely on malignant cells. The use of immunosuppressants has been shown to not only elevate the occurrence of malignant neoplasms post-transplantation, but also to reduce tolerance to cancer therapy, ultimately impacting treatment efficiency. 18 Conversely, immune checkpoint inhibitors may cause irreversible acute renal failure, and most patients required hemodialysis to survive, despite high-dose hormone therapy.19–21 Consideration should thus be given to the interplay between immunosuppressants and cancer treatment when devising therapeutic plans.
We present a case highlighting the effects of cytoreductive nephrectomy in post-transplant patients with bilateral native RCC. The patient exhibited a sustained response following cytoreductive nephrectomy in conjunction with sequential TKI and anti-PD-1 monoclonal antibody therapy. This is the first study to report the use of this combined treatment approach in kidney transplant recipients with metastatic RCC. Further studies are needed to reassess the application of surgical and pharmacological interventions in this specific demographic.
