Abstract
Pleomorphic hyalinizing angiectatic tumor (PHAT) is a rare tumor of the soft tissue, usually located in lower extremities. There are rarely tumors reported in other anatomic locations. Herein, we report retroperitoneal PHAT in a male patient. A 41-year-old man was referred to our clinic due to an incidentally found retroperitoneal mass. Computed tomography (CT) scan showed a solid hypoechoic lesion containing fat component and calcified elements measuring about 80*72*45 mm in the right lower quadrant (RLQ) of the abdomen. Magnetic resonance imaging (MRI) showed circumscribe lesion measuring about 60 x 48 mm with partial enhancement and fat component. In pelvic exploration, a large mass was found that had encased the right external iliac artery and vein. Therefore, the mass and its surrounding iliac vessels were excised and removed en block. Then, the external iliac vessels were reconstructed with Gortex graft. No recurrence was found in 1 month and 3 months post-operation follow up. We report a pelvic retroperitoneal PHAT as a rare location of this tumor. It seems that PHAT must be considered in differential diagnosis in patients with soft tissue tumors in the pelvic cavity.
Introduction
Pleomorphic hyalinizing angiectatic tumor (PHAT) is a rare tumor that was first described in 1996 by Smith et al. 1 PHAT predominantly originates from the soft tissue of lower extremities. The tumor has a slight predilection for female gender and older ages; it usually presents as a lesion larger than 5 cm. 2 Herein, there is a report of an extremely rare location of this rare tumor. In this case, we found pathologically confirmed retroperitoneal PHAT in a male patient.
description
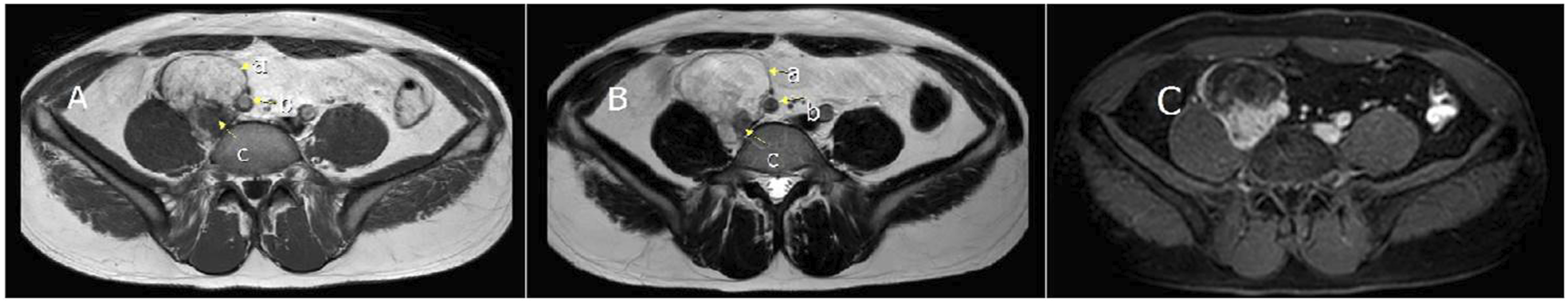
This research followed the tenets of the Declaration of Helsinki. Written and signed informed consent for publication was obtained from the patient. A 41-year-old man was referred to our clinic due to an incidentally found retroperitoneal mass. He had no important positive medical, family, and social history. His vital signs were stable. A mass was detected in the right lower quadrant (RLQ) during deep palpation in abdominal examination. Laboratory parameters were in the normal range. Abdomen and pelvic sonography showed an 8 cm mass in the retroperitoneum. Abdomen and pelvic computed tomography (CT) scan showed a solid fat containing lesion with soft tissue density component and calcifications measuring about 80*72*45 mm in RLQ (Figure 1). Abdomen and pelvic Magnetic resonance imaging (MRI) showed a well-circumscribed lesion with partial enhancement and fatty component. It was located adjacent to the right common iliac vessels, causing obliteration of the common iliac vein and encasement of the iliac artery (Figure 2). There was no diffusion restriction. Axial image of the CT scan demonstrated a fat-containing lesion with soft tissue component and calcification. Involvement of the right common iliac vessels is noted. A- Axial T1W image of the abdomen showed a fat-containing mass (a) with partial encasement of the right common iliac artery (b) and invasion to the right common iliac vein (c). B- Axial T2W image of the abdomen showed a fat-containing mass (a) with partial encasement of the right common iliac artery (b) and invasion to the right common iliac vein (c). C- Enhancement of the soft tissue component of the lesion is seen with involvement of common iliac vessels.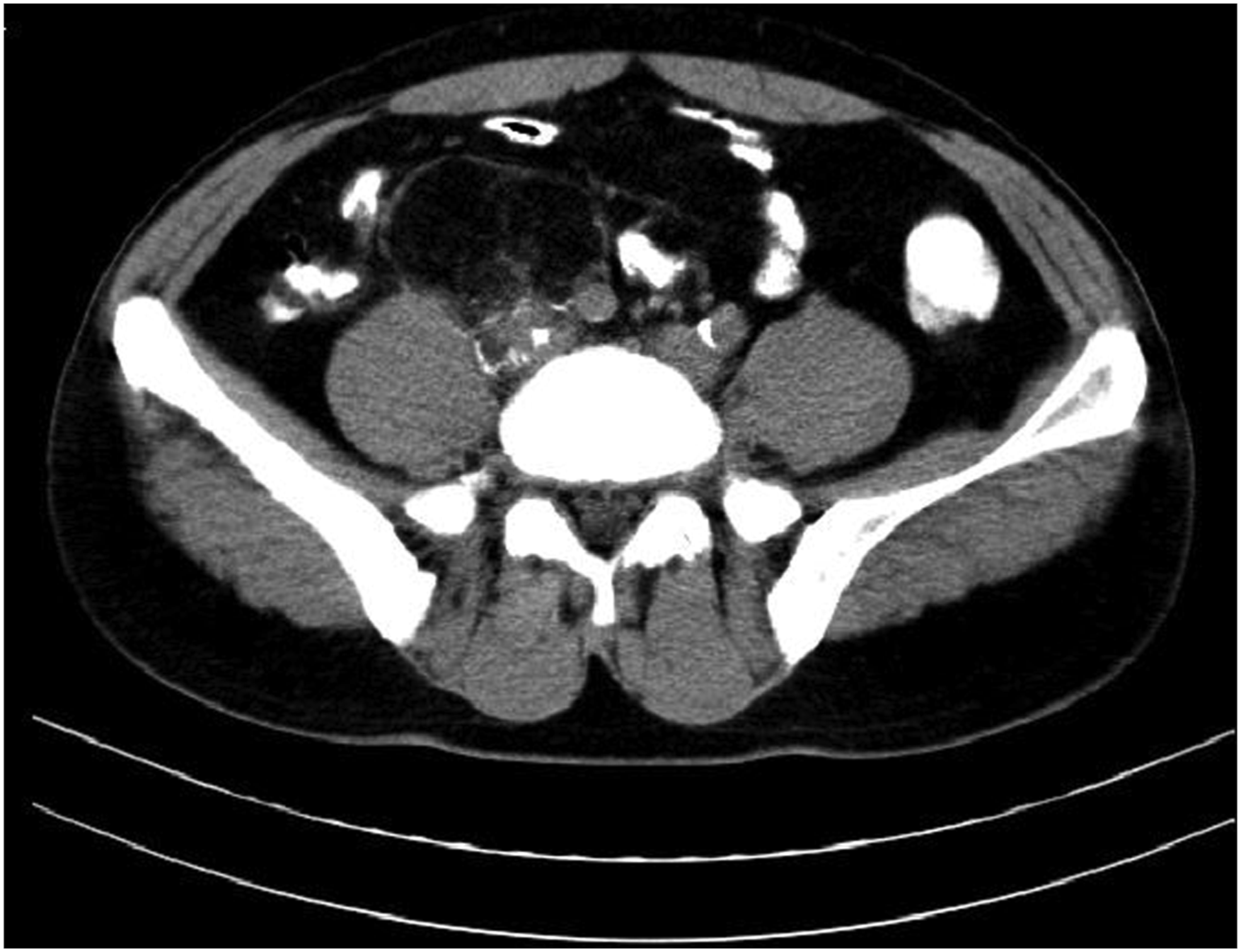
Operation
After prep and drape, under general anesthesia, through a midline incision, the right iliac fossa was entered. A large mass was found that had encased the right external iliac artery and vein. Therefore, after controlling the proximal and distal part of the external iliac vessels, the mass and its surrounding iliac vessels were excised and removed en block. Then, the external iliac vessels were reconstructed with Gortex graft. The specimen was sent for pathology review.
The patient tolerated the operation and had an uneventful post-op course. He was discharged 3 days after the operation.
Pathology
Gross examination of the retroperitoneal mass showed a relatively well-defined oval shaped mass measuring 7 × 7 × 7 cm with a smooth surface. The sections which were cut showed a yellow-white surface with hemorrhagic areas and foci of calcification. Microscopic examination showed a mixture of thin-walled ectatic blood vessels, spindle and pleomorphic tumoral cells, and lipomatous tissue with hyalinized collagenous areas (Figure 3(a)). Most blood vessels showed glomeruloid and papillary endothelial proliferation (Figure 3(b)). The intervening tumoral cells were spindle-shaped with marked pleomorphism; some of them had hemosiderin pigments with intracytoplasmic inclusions and micro-thrombi formation (Figures 3(c)-(d)). Rare mitotic figures were identified. No necrosis was detected. Small foci of ossification and calcification were seen (Figures 3(e)-(f)). Mast cells were present (Figure 3(g)). An immunohistochemical study was performed with positive CD34 in the tumoral cells (Figure3(h)). Smooth muscle actin was focally positive in the spindle cells. Cytokeratin, S100, BCL2, HMB45, and STAT6 were negative. CISH study for MDM2 gene amplification was negative. Hence, the diagnosis of PHAT was considered for this tumor. A- Microscopic section shows clusters of thin-walled angiectatic vessels admixed with the adipose tissue and pleomorphic spindle cells (Hematoxylin and Eosin, 40×). B- Blood vessels with glomeruloid and papillary endothelial proliferation (Hematoxylin and Eosin, 200×). C- Pleomorphic tumoral cells with intracytoplasmic vacuoles (arrow head) and microthrombi (arrow) (Hematoxylin and Eosin, 400×). D-Hemosiderin pigment (Hematoxylin and Eosin, 400×). E− Calcification (Hematoxylin and Eosin, 400×). F-Ossification (Hematoxylin and Eosin, 100×). G-Mast cell infiltration (Hematoxylin and Eosin, 400×). H-Immunohistochemical stain for CD34 (200×).
Follow up
The patient was followed 1 month and 3 months after the operation. No sign of relapse was detected in the follow up sonographic evaluations. Follow up laboratory findings were normal.
Discussion and conclusion
PHAT of the soft parts is a rare, yet distinctive, tumor with locally aggressive behavior; it was initially described in 1996 in a series of 14 cases by Smith et al. 1 It is found that PHAT is more prevalent among female gender and older ages 2 ; this patient was a young-to-mid-aged man. The mean age of literature reported cases of PHAT was 54.5 ± 17.1 and 61% of them were female. 3 Subcutaneous soft tissues of lower extremities were the most reported site of origination of PHAT.2,4 Our patient presented with deep pelvic retroperitoneal PHAT.
A report similar to our case was a 26-year-old female with an about 9 cm mass located in front of the sacrum and behind the uterus and rectum. 5 Braiki et al. 6 reported a 40-year-old woman with a 10*7*5 cm mass located in the pelvic retroperitoneum just near the right ureter and right iliac vessels. Another case of retroperitoneal involvement was a 71-year-old white woman with a 4.2 × 6 cm mass in the right renal hilum . 7 Balasubiramaniyan et al. 8 reported a 30-year-old female with complaint of dysmenorrhea and a 7.5 × 6 × 4.5 cm mass in the left presacral area abutting the left lateral wall of the rectum. Other unusual sites for PHAT such as the breast, 9 vulva, 10 and oral cavity 11 have been reported in the literature.
Microscopically, the most striking feature at low magnification is the presence of clusters of thin-walled ectatic blood vessels, ranging in size from small to macroscopic and distributed throughout the lesion 1 Because of the paucity of reports, the lineage of this tumor has yet to be elucidated. It has been suggested that deposition of perivascular hyaline material leads to progressive vascular obliteration and tumoral hypoxia, which, in turn, promotes VEGF production by the neoplastic cells, resulting in active angiogenesis . 12 In the series reported by Folpe and Weiss , 13 the authors identified a pattern at the periphery of some tumors, which they termed early PHAT. These areas are characterized by low to moderate cellularity composed of bland, hemosiderin-laden spindled cells with wavy nuclei arranged in fascicles which infiltrate the peripheral adipocytes. The morphologic features of early PHAT are identical to those of hemosiderotic fibrolipomatous tumor. In our case, also, there were areas which resembled early PHAT. By immunohistochemistry, the cells of PHAT express CD34 and vimentin but are negative for actins, desmin, cytokeratin, EMA, von Willebrand factor, and CD31.8,14 However, these findings are not specific for its diagnosis.
In view of its histologic features, PHAT bears similarities with other soft tissue tumors and could be easily misdiagnosed as any of them. Examples of such tumors include low grade malignant fibrous histiocytoma, ancient schwannoma, cellular angiofibroma, solitary fibrous tumor, gastrointestinal stromal tumor, undifferentiated pleomorphic sarcoma, and hemosiderotic fibrolipomatous tumor.14,15 For differentiation of these tumors, combinations of pathologic examination and immunohistochemical stains are used .5,16,17
Imaging features of PHAT are not focused in the literature, and there are few reports about this issue. It is mostly described with soft tissue lesions without calcification 4,13,14; however, CT imaging of our patient illustrated some calcified components in the mass.
A systematic review on PHAT illustrated that the overall local recurrence rate of this tumor was 28.6%, and the estimated mean time for recurrence was 43.87 months [95% confidence interval: 24.52–63.22; and standard error: 7.59]. 2 We followed the patients with ultra-sonography 1 month and 3 months after the operation that was tumor free. He was advised for follow up every 3 months.
In conclusion, PHAT is a rare soft tissue tumor of pelvic retroperitoneum that can be differentiated from other entities with respect to histopathological and immunohistochemical features.
Footnotes
Acknowledgements
The authors would like to thank Shiraz University of Medical Sciences, Shiraz, Iran and also Center for Development of Clinical Research of Nemazee Hospital and Dr. Nasrin Shokrpour for editorial assistance.
Author contributions
MS, AZ, and AA were involved in the patient’s surgery and manuscript drafting. MA and BG performed the histological examination of the mass and contributed to writing the pathology section. HJ reviewed the imaging modalities. All authors read and approved the final draft of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the ethics committee, and the patient gave his consent.
Informed consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor of this journal.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Appendix
List of abbreviations
