Abstract
Pleomorphic hyalinizing angiectatic tumor of soft tissue (PHAT) is a rare, low-grade neoplasm of uncertain lineage that predominantly affects the lower limbs of middle-aged adults. We report the case of a male in his early 20s who presented with a progressively enlarging, mildly tender mass above the right knee, present for 6 years. Imaging studies revealed a well-circumscribed lesion with obvious contrast enhancement on magnetic resonance imaging and marked internal vascularity on color Doppler ultrasound, located anterior to the distal quadriceps femoris. Extended excision was performed, and histopathologic examination confirmed the diagnosis of pleomorphic hyalinizing angiectatic tumor of soft tissue with negative surgical margins (R0 resection). The patient remained disease-free at the short-term follow-up (as of mid-2025). This report highlights the clinicopathologic features that differentiate pleomorphic hyalinizing angiectatic tumor of soft tissue from mimics such as myofibroma and intramuscular hemangioma, particularly in younger patients and at unusual anatomical locations.
Keywords
Introduction
Pleomorphic hyalinizing angiectatic tumors (PHATs) are rare, locally aggressive soft-tissue neoplasms first defined by Smith et al. in 1996 in a seminal study of six cases. 1 Histologically, PHATs are characterized by ectatic, hyalinized blood vessels surrounded by pleomorphic stromal cells, 2 a feature that often leads to diagnostic confusion with malignant tumors such as undifferentiated pleomorphic sarcoma (UPS). Although classified as low-grade neoplasms, PHATs exhibit a tendency for local recurrence, and distant metastasis is extremely rare. 3
PHAT is exceptionally rare, with fewer than 100 cases reported in the literature. 4 It primarily affects adults (median age, 50–60 years) and shows no significant sex-based predilection.5,6 The tumor predominantly arises in the subcutaneous tissues of the lower extremities 7 (e.g. ankle and foot); however, in rare cases, it involves the upper limbs, trunk, and visceral sites.8,9
The diagnostic hallmarks of PHAT include hyalinized angiectatic vessels, which are dilated, thin-walled vessels with perivascular fibrin deposition, 4 and pleomorphic stromal cells 10 that range from spindled to epithelioid with nuclear atypia, often raising concerns about malignancy. Immunohistochemically, tumor cells are typically positive for vimentin and CD34 but negative for S100, desmin, and smooth-muscle actin (SMA),11,12 which helps distinguish them from mimics such as myxofibrosarcoma.
The imaging characteristics of PHATs are described in a limited number of published reports. On magnetic resonance imaging (MRI), they present as well-circumscribed, T2-weighted hyperintense masses with heterogeneous enhancement due to vascular and hyalinized components; however, irregular margins have been reported in some cases.13–20 Computed tomography (CT) reveals a soft-tissue density mass without calcifications,19,21–24 whereas ultrasound may reveal hypoechoic regions with internal vascularity.15,24,25
Emerging evidence suggests an association with the TGFBR3-MGEA5 fusion gene, 20 also observed in hemosiderotic fibrolipomatous tumor (HFLT),26,27 indicating a tumor spectrum. However, the molecular drivers of PHAT remain poorly defined.
Wide local excision 1 remains the mainstay of treatment, with recurrence rates of 30%–50% when surgical margins are positive. 2 Although metastases are exceedingly rare, adjuvant therapies (e.g. radiation) are still recommended; however, evidence-based medical support for these recommendations is limited due to the tumor’s rarity.1,2,20,28 Long-term follow-up is recommended due to potential late recurrences. 27
Case presentation
Clinical findings
A male in his early 20s with no significant medical history presented with a 6-year history of a painless mass over the anterolateral aspect of the right distal thigh. Written informed consent for surgical treatment was obtained from the patient prior to the procedure. The patient was admitted to The Second Affiliated Hospital of Wannan Medical College, Wuhu City, Anhui Province, on 1 July 2025. Over the past year, the lesion had increased in size and become mildly tender. He denied constitutional symptoms, tobacco or intravenous drug use, and prior tuberculosis exposure. A remote history of right knee sprain was noted.
On physical examination, a 4 cm × 5 cm, soft, well-defined, nonfluctuant mass was palpable 3 cm proximal to the superior patellar pole. There was no overlying erythema, ulceration, or purulent discharge. The mass was minimally tender and fixed to deeper planes. Range of motion of the knee joint was full, and distal neurovascular status was intact.
Imaging studies
Ultrasonography revealed a 93 mm ×44 mm × 95 mm hypoechoic mass with clear margins, homogeneous echotexture, and marked internal vascularity on color Doppler. No calcifications or necrosis were identified (Figure 1).
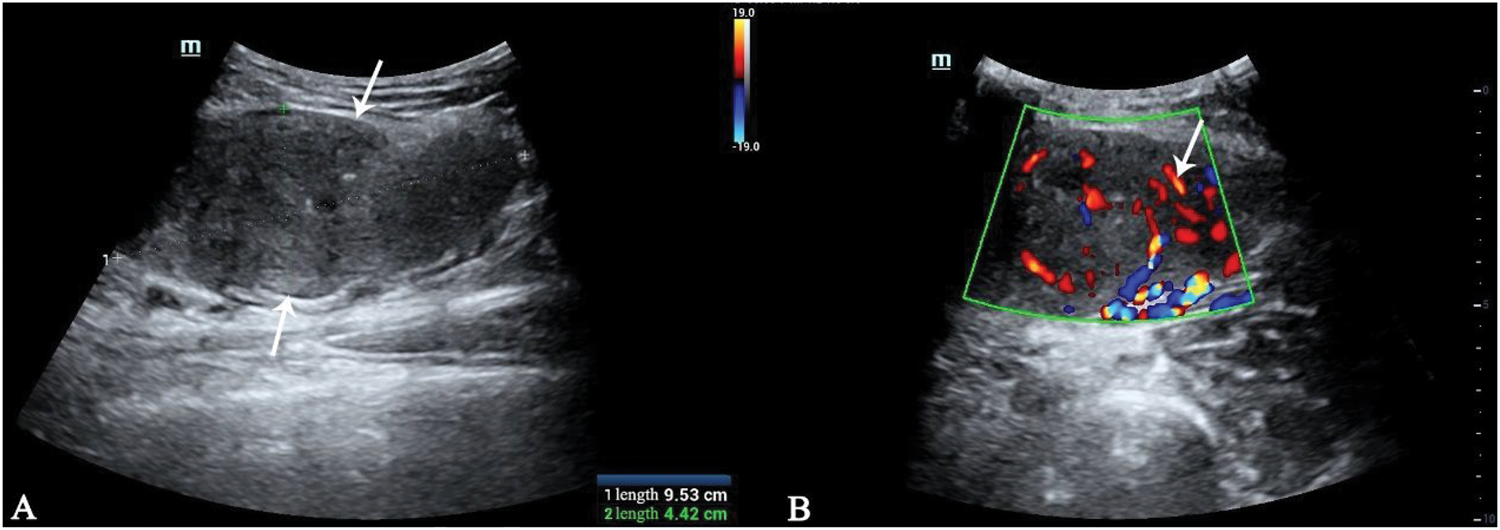
Ultrasonography images of the right distal thigh lesion. (a) Longitudinal view showing a 93 mm × 44 mm × 95 mm hypoechoic mass with clear margins and homogeneous echotexture (arrow indicates the mass boundary) and (b) color Doppler imaging demonstrating marked internal vascularity within the mass (arrow indicates blood flow signals). No calcifications or necrosis were identified.
Plain MRI revealed a lobulated subcutaneous mass superficial to the distal quadriceps tendon. The lesion was isointense to skeletal muscle on T1-weighted sequences and heterogeneously hyperintense on T2-weighted sequences, with punctate foci of high T2-weighted signal suggestive of cystic change. Following gadolinium administration, intense heterogeneous enhancement was observed (Figure 2). Radiologic differential diagnoses included myofibroma and intramuscular hemangioma.
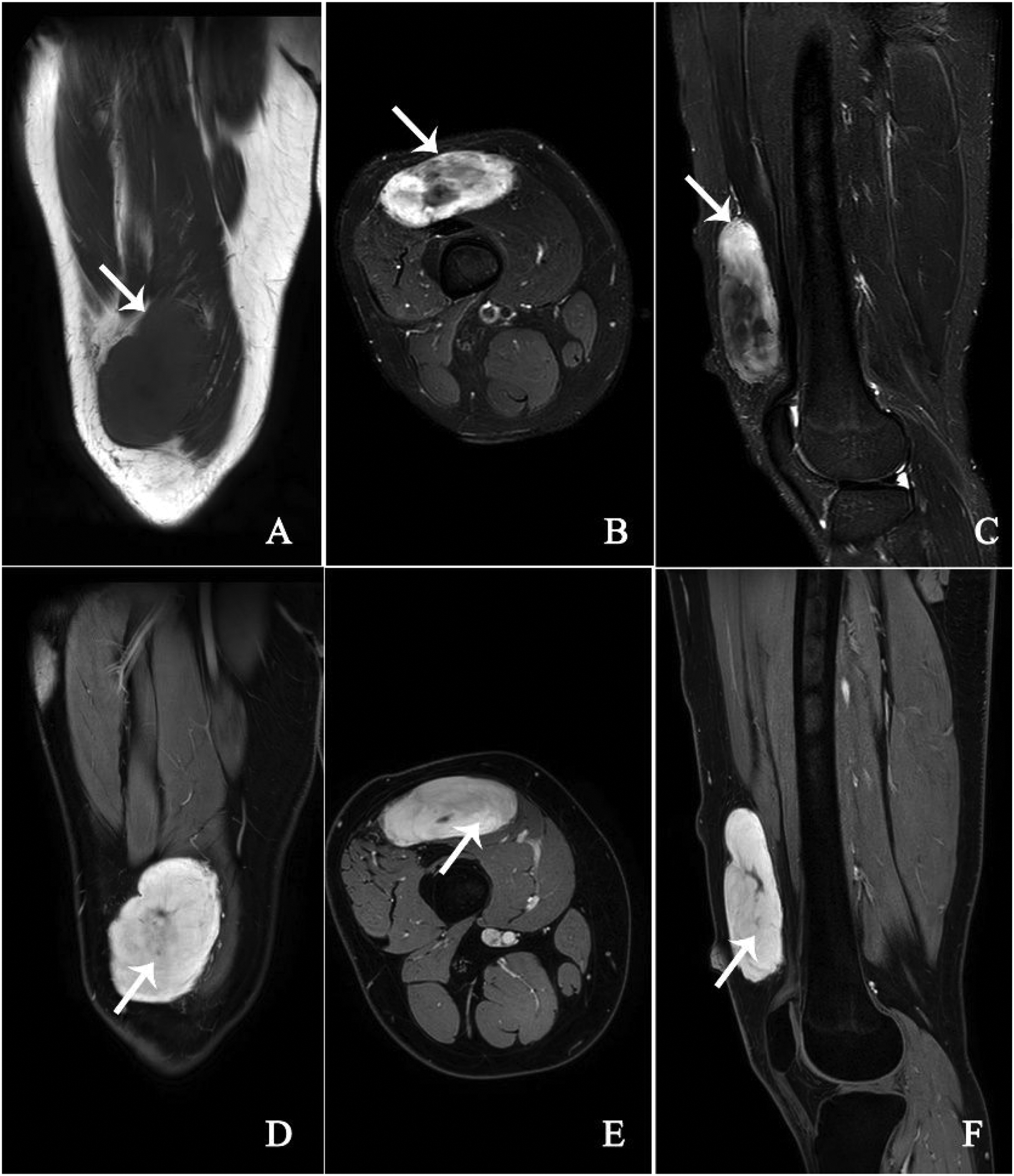
Magnetic resonance imaging (MRI) findings of the right distal thigh lesion. (a) T1-weighted sequence showing an isointense mass relative to the skeletal muscle (arrow indicates the mass); (b and c) T2-weighted sequences revealing heterogeneous hyperintensity with punctate foci of high T2-weighted signal (arrow indicates cystic change) and (d–f) gadolinium-enhanced images (coronal, axial, and sagittal planes) demonstrating intense heterogeneous enhancement of the mass (arrows indicate enhanced areas).
Surgical management
Under general anesthesia, an extended marginal excision was performed on 4 July 2025. A 12 cm longitudinal incision was made over the anterolateral thigh, allowing en bloc removal of the mass with a 1 cm cuff of uninvolved subcutaneous tissue and underlying deep fascia. Hemostasis was meticulous; no intraoperative complications occurred.
Pathologic findings
Gross examination yielded an 11 cm ×8 cm × 5 cm well-encapsulated, yellow-tan mass with a glistening, lobulated cut surface (Figure 3). Microscopically, the tumor displayed variably sized, ectatic, thin-walled vessels lined by attenuated endothelium and surrounded by dense hyalinized stroma. Scattered pleomorphic spindle cells with hyperchromatic nuclei were present; however, mitotic activity was low (<1/10 high-power field (HPF)). No necrosis or vascular invasion was noted. Histopathological examination confirmed negative surgical margins (R0 resection), with no tumor cells identified at the edges of the excised specimen (Figure 4).

Gross appearance of the surgical specimen.

Histopathological findings of the surgical specimen stained with H&E (100× field of view (FOV) and 400× FOV). H&E: hematoxylin and eosin; FOV: field of view.
Immunohistochemistry demonstrated strong cytoplasmic positivity for vimentin, SMA, S-100 protein, CD31, and CD34. Desmin, SOX-10, AE1/AE3, and CD30 were negative. The Ki-67 labeling index was 5%. The constellation of findings confirmed the diagnosis of PHAT (Figure 5).

Immunohistochemical staining of PHAT tissue (100× FOV). (a) Vimentin positivity; (b) SMA positivity; (c) S-100 positivity; (d) CD31 positivity; (e) CD34 positivity; (f) desmin negativity; (g) SOX-10 negativity; (h) AE1/AE3 negativity; (i) CD30 negativity; and (j) Ki-67 labeling index of 5%. PHAT: pleomorphic hyalinizing angiectatic tumor of soft tissue; FOV: field of view; SMA: smooth-muscle actin.
Follow-up
The patient was followed up for 2 months postoperatively (as of September 2025). Clinical examination showed no local tenderness, swelling, or palpable mass at the surgical site. No postoperative complications (e.g. wound infection and motor/sensory deficits of the lower limb) were reported.
The reporting of this study conforms to the Case Report (CARE) guidelines. 29
Discussion
PHAT is a rare soft-tissue neoplasm first definitively characterized in 1996. 1 In the 2020 World Health Organization (WHO) classification of soft-tissue tumors, PHAT is categorized as an “intermediate (rarely metastasizing)” tumor because of its locally aggressive behavior and low metastatic potential. 12 Epidemiologically, PHAT predominantly affects adults (median age, 50–60 years), with no significant sex-based predilection,5,6 and most cases arise in the subcutaneous tissues of the lower extremities (ankle/foot, 70%).7–9 Despite its rarity (<100 reported cases), PHAT has garnered attention owing to its unique histomorphology and diagnostic challenges.10–12 Notably, this case broadens the demographic and anatomical spectrum of PHAT. Most reported cases occur in patients aged 50–60 years;1–3 in contrast, our patient was in his early 20 s, with only 3 cases of PHAT in patients aged <30 years documented in the literature. 19 Furthermore, PHATs predominantly arise in the ankle/foot region,4,18 whereas in our case the distal thigh was involved, a location reported in only 5% of PHAT cases. 27 Therefore, this case provides valuable data on PHAT in unusual populations and anatomical sites.
Radiologically, PHAT presents as a well-circumscribed mass demonstrating characteristic T2-weighted hyperintensity and heterogeneous enhancement on MRI, reflecting its mixed vascular and hyalinized components.13,15,18,20 The absence of perilesional edema and infiltrative margins helps differentiate PHAT from myxofibrosarcoma, 30 whereas the lack of phleboliths and uniform hyperintensity distinguishes it from hemangiomas. 31 Unlike synovial sarcoma, PHAT typically lacks calcifications, and its slower contrast washout patterns differ from the rapid enhancement observed in angiosarcoma. 32 Schwannomas, although similarly well-demarcated, can be differentiated by their characteristic “target sign” appearance and association with nerve thickening. 33 Functional MRI features of PHAT, as reported in recent studies, 34 include isointense to slightly hyperintense signals on diffusion-weighted imaging (DWI) and slightly lower apparent diffusion coefficient (ADC) values compared with those of normal soft tissue. Perfusion-weighted imaging (PWI) often shows progressive enhancement, reflecting the tumor’s vascular perfusion characteristics. However, functional MRI sequences and angiography were not performed in this case, which may have limited the comprehensive evaluation of the lesion’s vascular properties, representing a key limitation of our study.
Pathologically, PHAT is characterized by two hallmark histologic features: ectatic, hyalinized blood vessels with perivascular fibrin deposition and a surrounding population of pleomorphic stromal cells exhibiting nuclear atypia. 2 Although the degree of nuclear pleomorphism may initially raise concern for high-grade sarcomas such as UPS, PHAT can be distinguished by its lower mitotic activity, absence of tumor necrosis, and preservation of the distinctive vascular architecture. 1 The differential diagnosis also includes myxoinflammatory fibroblastic sarcoma (MIFS), which shares a similar clinical presentation but differs in its prominent inflammatory infiltrates, virocyte-like cells, and lack of CD34 expression. 35 Another key mimic is HFLT, which demonstrates overlapping TGFBR3-MGEA5 gene fusion but demonstrates adipocytic differentiation and hemosiderin deposits, features not observed in PHAT. 36 Immunohistochemically, PHAT typically expresses vimentin and CD34 and is negative for S100, desmin, and SMA, aiding in its distinction from neural or myogenic tumors.11,12
Molecular studies have identified recurrent TGFBR3-MGEA5 gene fusions in 50%–70% of PHAT cases, which are also observed in HFLT, suggesting that these entities represent a spectrum of related neoplasm.36,37 This fusion gene is believed to contribute to PHAT’s characteristic vascular hyalinization through dysregulation of the extracellular matrix remodeling and angiogenesis pathways.36,37 However, the exact oncogenic mechanisms remain unclear, warranting further genomic studies.
Current management emphasizes complete surgical excision with wide margins (R0 resection), as positive margins correlate with a 30%–50% local recurrence rate.1,2 Given its exceptionally low metastatic potential, radical resection or adjuvant therapies are generally necessary;1,2,20 however, long-term follow-up is recommended to monitor for late recurrences. 27 Current challenges in management stem from the tumor’s rarity, which limits evidence regarding the role of radiation or systemic therapies. 28 Future research should focus on elucidating the oncogenic mechanisms of the TGFBR3-MGEA5 fusion to identify potential targeted therapies, particularly antiangiogenic agents that may exploit PHAT’s vascular phenotype.36,37 Additionally, multicenter collaboration is needed to establish standardized diagnostic criteria and to investigate novel biomarkers that may improve differentiation from histologic mimics. Prospective studies evaluating the efficacy of conservative versus aggressive surgical approaches in preventing recurrence would further optimize clinical management of this rare tumor.
This study has two main limitations. First, as a single-case report, its findings cannot be generalized to all patients with PHAT. Second, long-term follow-up (>1 year) is required to further assess the risk of late recurrence in young patients.
Conclusion
PHAT is a rare, indolent soft-tissue neoplasm that can clinically and radiologically mimic benign vascular or fibroblastic lesions. Awareness of its characteristic histopathologic and immunohistochemical features (vimentin+, CD34+, and S100±) is essential to avoid misdiagnosis. Extended excision with clear margins (R0 resection) remains the mainstay of treatment for durable local control, and short-term follow-up in this case showed no recurrence.
Supplemental Material
sj-zip-1-imr-10.1177_03000605251410284 - Supplemental material for Pleomorphic hyalinizing angiectatic tumor of soft tissue in the distal thigh: A case report and literature review
Supplemental material, sj-zip-1-imr-10.1177_03000605251410284 for Pleomorphic hyalinizing angiectatic tumor of soft tissue in the distal thigh: A case report and literature review by Xiu-Fang Yan, Ying Li, Qiang Wan, Guang-Bao Mei, Chun-Shuang Li and Tong-Hua Zhang in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank the Department of Pathology for expert histologic interpretation.
Author contributions
XF Y and YL were involved in the drafting of the paper as well as the conception and design of the study.
Q W and GB M were involved in the analysis and interpretation of the data, and literature retrieval.
TH Z was involved in revising the manuscript critically for intellectual content and gave final approval of the version to be published.
CS L performed histopathological diagnosis, immunohistochemical analysis, and revised the pathological section of the manuscript.
Conflicts of interest
The authors declare no conflicts of interest.
Declaration of funding
None.
Data availability statement
The original clinical, imaging, and pathological data supporting the conclusions of this article are available from the corresponding author (TH Z) upon reasonable request.
No artificial intelligence (AI) tools were used in the research methods of this study.
Ethics statement
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Ethics committee statement
Case reports do not require ethical committee approval in accordance with local regulations, and no such approval was obtained for this study.
Image copyright
Necessary permissions for using all clinical images in this manuscript have been obtained from the Medical Records Department.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
