Abstract
Background: Patient-reported outcomes (PROs), including health-related quality of life, are recommended to be routinely collected in clinical trials, but data are limited from trials of sarcoma patients. In this analysis, pooled PRO data are reported from patients with advanced or metastatic soft tissue sarcoma (STS) enrolled to the ANNOUNCE phase III trial of doxorubicin-based therapy. Methods: ANNOUNCE was a phase III trial that randomized 509 patients with STS to receive up to eight cycles of doxorubicin with olaratumab or placebo, followed by single-agent olaratumab or placebo. Dexrazoxane was allowed at any cycle of treatment. Participants completed the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire-Core 30 (EORTC QLQ-C30, which is scored 0–100), and Brief Pain Inventory Short Form Modified (mBPI-sf, scored from 0–10) at each treatment cycle. A descriptive analysis of the longitudinal data was conducted overall and by cumulative dose of doxorubicin received to inform the clinical care of patients with STS. Worsening on the QLQ-C30 was defined as a change of 10 points or more at any post-baseline assessment. Worsening on the mPBI-sf was defined as an increase of ≥2 points from baseline. Results: The majority of participants completed the baseline and at least one subsequent PRO assessment within the trial (n = 460, 90.4% EORTC QLQ-C30; n = 454, 89.2%, mBPI-sf). Patients with STS enrolled to the ANNOUNCE trial had clinically meaningful problems with physical function and pain before initiating doxorubicin. Overall, those with fewer symptoms or better function at baseline received higher cumulative doxorubicin dose throughout the study. At baseline, mean QLQ-C30 fatigue was 29.9 with a median time to first worsening of 0.9 months, and mean nausea/vomiting was 6.5 with 1.4 months until worsening; mean physical function was 78.3 with median time to worsening of 2.1 months and mean health status was 66.8 with median time to first worsening of 1.6 months. Median time to worsening of pain was 7.9 months. Conclusion: Patients with advanced or metastatic sarcoma reported a relatively rapid decline in PROs during doxorubicin-based treatment, with patients with poorer symptoms at baseline (specifically fatigue), subsequently receiving less doxorubicin therapy. The availability of detailed summary data from the patient perspective during doxorubicin-based treatment may inform future care of these patients and can provide a resource for the development of PRO endpoints in future trials.
Keywords
Introduction
Patient-reported outcomes (PROs), including health-related quality of life (HRQoL), are recommended to be routinely collected in clinical trials. 1 The rationale for collecting such data is based on the importance of patient-centered care, which ensures that all perspectives of treatment choices are considered in clinical decision making. In oncology, the sequelae of systemic therapy have an impact on patients that cannot be reliably evaluated from an outside perspective (e.g. the impact and severity of symptoms, physical and functional well-being, pain and discomfort) or from clinical efficacy endpoints alone. The recommendation to include PROs in trials is also based on the premise that the best source to understand the patient experience is from the patient directly without influence or interpretation from anyone else. 2 Unfortunately, despite this guidance, PROs have not consistently been included in the design or publications of clinical trials enrolling patients with soft-tissue sarcoma (STS).3,4 This may be due to a number of factors, such as the lack of inclusion of hypotheses or objectives related to outcomes from the patient perspective, heterogeneity of sarcomas, or lack of standardized instruments specific to sarcomas. While OS and PFS are clearly important primary endpoints in a clinical trial, it is essential that the patient experience related to these outcomes are captured alongside the clinical findings.
The primary result of a Randomized, Double-Blind, Placebo-Controlled, Phase III Trial of Doxorubicin Plus Olaratumab Versus Doxorubicin Plus Placebo in Patients With Advanced or Metastatic Soft Tissue Sarcoma (ANNOUNCE, NCT02451943) was published in 2020, 5 demonstrating that the addition of olaratumab to doxorubicin failed to improve OS or PFS. 5 There were also no statistically significant or clinically meaningful differences in any of the quality of life measures or subscales included in the study, and safety outcomes were similar between treatment arms. 5 Therefore, this analysis combined patient-reported data collected from each treatment group to provide a thorough evaluation of PROs longitudinally during treatment with doxorubicin-based therapy among patients with advanced or metastatic soft tissue sarcoma. These descriptive data are provided by cumulative dose of doxorubicin received to be consistent with prior work exploring the outcomes associated with doxorubicin dosing in the ANNOUNCE trial. 6 The primary objective of this work is to provide a depth of information about PROs in sarcoma that is currently lacking in the literature.
Methods
ANNOUNCE trial cohort
Patients who had enrolled to the ANNOUNCE study were included in this analysis of PROs, which were pooled across treatment arms due to the lack of any differences observed in the primary analysis. PRO data were described overall and by cumulative dose of doxorubicin received. The protocol was approved by the institutional review board at each participating center, and the study was done in accordance with the Declaration of Helsinki and the International Conference on Harmonisation Guidelines for Good Clinical Practice. All patients provided written informed consent to participate in the study.
Exploratory objectives and patient-reported outcome measures
The trial included a number of secondary objectives based on PROs, including time to first worsening on the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire-Core 30 (EORTC QLQ-C30) and time to first worsening of the Brief Pain Inventory Short Form Modified (mBPI-sf). The EORTC QLQ-C30 is a well-validated instrument that assesses health-related QoL in adult cancer patients. It is composed of scales that evaluate physical (5 items), emotional (4 items), role (2 items), cognitive (2 items), and social functioning (2 items), as well as global health status (2 items), which are scored from 0–100, with higher scores representing better functioning. There are also three symptom scales measuring nausea and vomiting (2 items), fatigue (3 items), and pain (2 items), and six single items assessing financial impact and various physical symptoms. These scales are also scored from 0–100, with lower scores representing fewer symptoms. For the functioning subscales (where higher scores represent better functioning), values below 83 for physical functioning, 58 for role functioning and social functioning, 71 for emotional functioning, and below 75 for cognitive functioning represent a clinically important problems. 7 For the symptom subscales (where higher scores represent more symptomology), values above 39 for fatigue, 25 for pain, eight for nausea/vomiting, 50 for sleep disturbances, 17 for dyspnea, diarrhea and financial impact, and above 50 for appetite loss and constipation represent clinically important symptoms. 7
For this combined analysis, QLQ-C30 subscales are reported for symptom subscales that corresponded to grade 3–4 toxicities reported among >1% of the study cohort: fatigue and nausea/vomiting. The global health status and physical functioning subscales are also reported as being particularly relevant in sarcoma. As defined in the study protocol, worsening on the functioning subscales was defined as decrease of at least 10 points from baseline. For the symptom subscales, worsening was defined as an increase of at least 10 points. The 10-point threshold has been established in prior research, but has not been validated for sarcoma. 8 Time to first worsening was calculated as the time from the first study drug dose (baseline date) to the first observation of worsening and was evaluated using the Kaplan-Meier method. 9 Patients with the lowest reportable score at baseline (because worsening could not be observed), a missing baseline assessment, or with no post-baseline assessments were not included in time-to-event analyses. Patients without events were censored at the last QLQ-C30 assessment.
The mBPI-sf is a validated instrument, developed to assess the severity of pain and its impact on functioning, where a score of 0 corresponds to no pain/no impact on a 0-10 scale. 10 Pain severity is estimated as “least, “worst,” and “average” during a 24-h recall period as well as “current.” Time to first worsening of the mBPI-sf was defined as the time from the date of the first study drug dose (baseline date) to the first date of a “worst pain in the past 24 h” score increase of ≥2 points from baseline.11,12 Time-to-event analyses used the Kaplan-Meier method. Patients with a baseline score of eight or more, with a missing baseline score, or with no post-baseline assessments were not included in the time-to-event analyses. Patients without events were censored at the last mBPI assessment.
Because of the high level of consistency in all PROs measured across treatment groups, 5 data were combined to provide a descriptive summary of longitudinal data from a cohort of patients treated with doxorubicin to inform clinical care and to support the development of future research. Given that there remains some controversy regarding the appropriate cumulative dose of doxorubicin in the setting of advanced or metastatic sarcoma, PROs are reported for three categories of cumulative doxorubicin dose: patients who received <450 mg/m2; patients who received 450–599 mg/m2; and patients who received ≥600 mg/m2, respectively. 6 There were no a priori hypotheses or planned comparisons between these groups; therefore, all data are reported descriptively. Data are presented through cycle 15, allowing for up to the study-designated eight cycles of doxorubicin, plus PROs recorded through an additional seven cycles of placebo or olaratumab monotherapy treatment (post-doxorubicin period). PRO data were not collected during receipt of subsequent therapy.
Results
Details of the ANNOUNCE trial population and their baseline characteristics have been previously published. 5 Overall, the median age of the 509 randomized patients was 57 years (70.7% were under age 65), 296 (58.2%) were female, and 234 (46.0%) had leiomyosarcoma. The eligibility criteria of ANNOUNCE required that all patients have locally advanced or metastatic disease; patients with metastatic disease comprised 82.9% of the enrolled population. Few patients had received prior adjuvant or neoadjuvant therapy (3.9%) or any other prior systemic therapy (26.7%) for locally advanced or metastatic disease. Approximately one-third of patients (33.8%) had received prior radiation therapy. Of the 506 randomized patients who received at least one dose of doxorubicin, and 485 patients completed at least one of the baseline PRO instruments. Of the 506 randomized patients, 324 (64.0%) received at least one dose of dexrazoxane; 106 (20.9%) initiated dexrazoxane at cycle 1 and 154 (30.4%) at cycle 5. The remaining 182 (34.0%) did not receive dexrazoxane during study treatment. 13 All of the baseline measures, time-to-event outcomes for each of the subscales of the QLQ-C30, and time to worsening on the mBPI-sf were not significantly different between treatment groups. 5
Of the 506 treated patients, 251 (49.6%) received <450 mg/m2 cumulative dose of doxorubicin, 165 (32.6%) received 450–599 mg/m2, and 90 (17.8%) received ≥600 mg/m2. Of all randomized patients, 90.4% completed a baseline assessment and at least one post-baseline assessment on the QLQ-C30. The number of evaluations available for analysis were dependent on duration of treatment in the study and decreased over time as patients discontinued therapy. Similarly, 89.2%, of randomized patients completed a baseline mBPI-sf and at least one post-baseline assessment; the number of assessments at each post-baseline visit was dependent on the duration of study participation.
The QLQ-C30 global health status subscale data from baseline through cycle 15 are presented in Figure 1 and the detailed data tables are provided in Supplementary Table 3. Mean baseline health status was 66.8 (SD = 23.4) overall, 63.3 (SD = 24.8) for those who subsequently received <450 mg/m2, 69.5 (SD = 20.8) for those who subsequently received 450–599 mg/m2, and 72.2 (SD = 22.8) for those who subsequently received a cumulative doxorubicin dose of ≥600 mg/m2. By subsequent cumulative doxorubicin dose groups, 68.3% of those who received <450 mg/m2, 90.6% of those who received 450–599 mg/m2 and 92.4% of those who received ≥600 mg/m2 experienced worsening of 10 points or more in global health status. In time-to-event analyses, there was a median time to worsening of 1.6 months (95% CI: 1.5, 2.1). Mean scores for the Cancer Quality of Life Questionnaire-Core 30 (EORTC QLQ-C30) global health status and physical functioning subscales by subsequent cumulative doxorubicin dose. Note: higher scores indicate better functioning.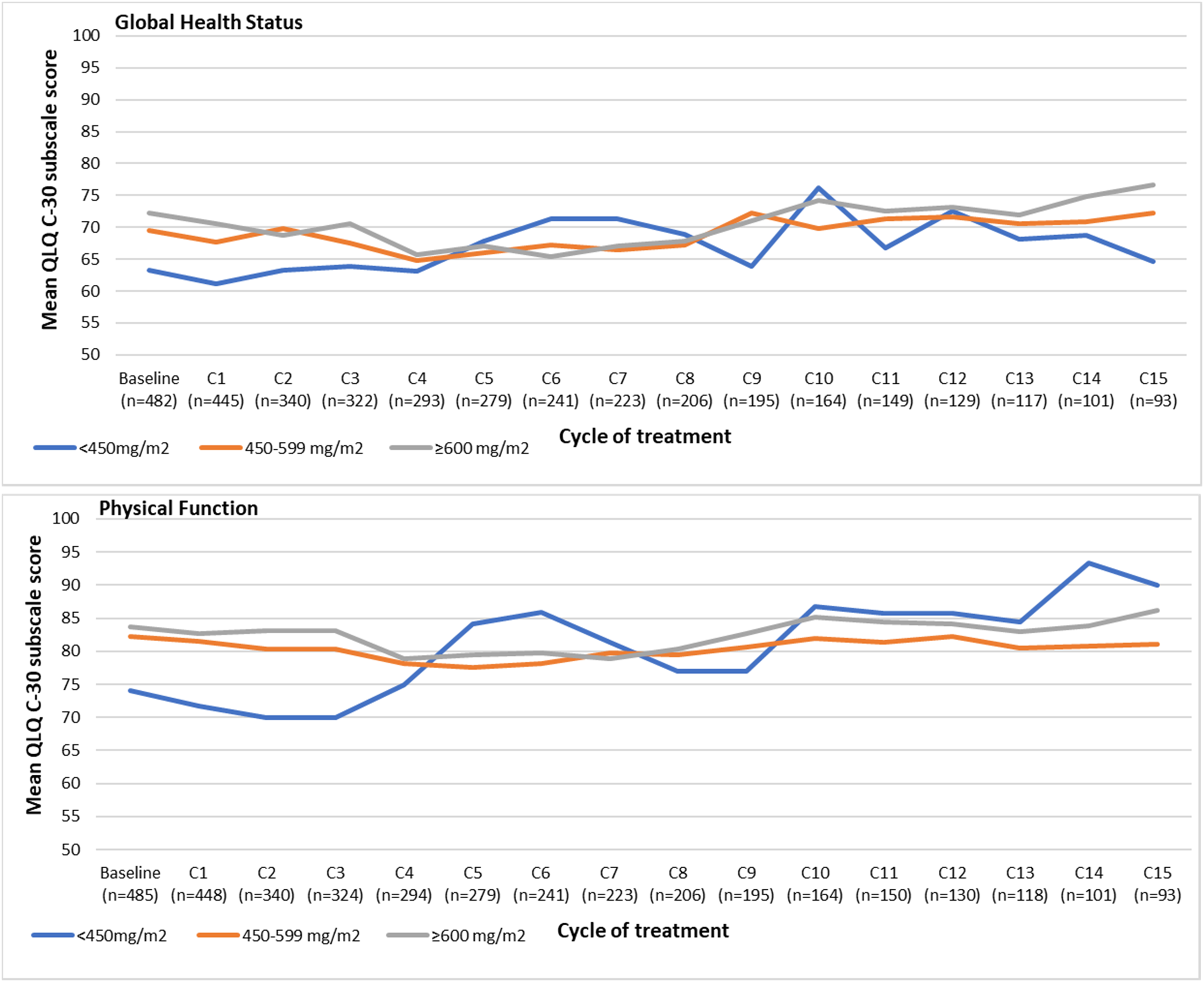
Physical functioning status is presented in Figure 1. Mean baseline physical functioning was 78.3 (SD = 21.7), overall and was 74.0 (SD = 23.2), 82.3 (SD = 19.0), and 83.7 (SD = 19.4) for patients who subsequently received a cumulative dose of doxorubicin of <450 mg/m2, 450–599 mg/m2 and ≥600 mg/m2, respectively. All baseline mean scores (except for among patients who subsequently received ≥600 mg/m2 doxorubicin represent a clinically important problem for patients) were below the 83-point threshold). The detailed data tables for the physical function subscale are provided in Supplementary Table 4. Among patients receiving <450 mg/m2, 64.9% experienced at 10-point or more worsening in physical functioning from baseline, whereas 86.2% and 78.1% experienced worsening in the 450–599 mg/m2 and ≥600 mg/m2 cumulative dose subgroups. In time-to-event analyses, the median time to worsening was 2.1 months (95% CI: 1.7, 2.8).
The QLQ-C30 fatigue and nausea/vomiting subscales from baseline through cycle 15 are presented in Figure 2 and the detailed summary data tables are provided in Supplementary Tables 1-2. At baseline, mean fatigue was 29.9 (standard deviation, SD = 24.5) for the study population. Patients who subsequently received <450 mg/m2, 450–599 mg/m2 or ≥600 mg/m2 cumulative dose of doxorubicin had baseline fatigue of 34.0 (SD = 26.1), 27.3 (SD = 22.7) or 22.6 (SD = 20.4), respectively. Mean baseline values for fatigue did not reach a clinically important threshold. All patients who received 450–599 mg/m2 or ≥600 mg/m2 doxorubicin experienced a 10-point or more worsening of fatigue, and 78.5% of patients who received <450 mg/m2 experienced worsening of fatigue. Median time to first worsening of fatigue was 0.9 months (95% confidence interval, CI: 0.8, 1.0) in time-to-event analyses. Mean scores for the Cancer Quality of Life Questionnaire-Core 30 (EORTC QLQ-C30) fatigue and nausea/vomiting subscales by subsequent cumulative doxorubicin dose. Note: higher scores indicate greater symptoms.
Baseline mean nausea/vomiting was 6.5 (SD = 14.8). Among patients who received <450 mg/m2 doxorubicin, mean nausea/vomiting was 8.1 (SD = 16.3), for those who received 450–599 mg/m2 mean nausea/vomiting was 5.2 (SD = 12.5), and for those receiving ≥600 mg/m2, mean nausea/vomiting was 4.6 (SD = 14.0). Mean baseline nausea/vomiting was considered clinically important problem (above the 8-point threshold) for those who subsequently received <450 mg/m2 doxorubicin. All patients who received 450–599 mg/m2 or ≥600 mg/m2 doxorubicin experienced worsening of nausea/vomiting, and 69.1% of patients who received <450 mg/m2 experienced worsening of nausea/vomiting. Median time to worsening of nausea/vomiting was 1.4 months (95% CI: 1.4, 1.5).
At baseline, patients reported a mean 24-h recall worst pain score of 2.7 (SD = 2.9), 24-h recall of least pain was 1.3 (SD = 1.8), and 24-h recall average pain reported was 2.1 (SD = 2.2). Mean current pain at baseline was 1.5 (SD = 2.1). Current pain and 24-h recall of worst pain as measured by the mBPI-sf are summarized in Figure 3 from baseline through cycle 15. By doxorubicin cumulative dose, baseline mean 24-h worst pain was 3.3 (SD = 3.1), 2.2 (SD = 2.4) and 2.1 (SD = 2.6) and current pain was 1.8 (SD = 2.3), 1.3 (SD = 1.9), and 1.2 (SD = 1.8) among patients receiving <450 mg/m2, 450–599 mg/m2, and ≥600 mg/m2 cumulative dose of doxorubicin, respectively. Pain worsened by >2 points in the 24-h recall of worst pain among 39.4%, 59.9% and 63.6% of patients, for those receiving <450 mg/m2, 450–599 mg/m2, and ≥600 mg/m2 cumulative dose of doxorubicin, respectively. Overall, time to worsening of pain was a median of 7.9 months (95% CI: 6.4, 9.5). The detailed summary data tables are included in Supplementary Tables 5 and 6. Current pain and 24-h recall of worst pain as measured by the modified Brief Pain Inventory-short form (mBPI-sf). Note: higher scores indicate greater pain.
Discussion
Patients with advanced or metastatic sarcoma receiving doxorubicin-based therapy in this study reported a decline in PROs during doxorubicin-based treatment. All treatment arms and study groups reported similar times to decline. Of note, patients with worse symptoms at baseline (specifically fatigue), subsequently received less doxorubicin therapy. Further research should explore baseline factors that may limit the planned treatment for patients with STS, and should also further investigate the role of dexrazoxane and other supportive care interventions on patient quality of life. Previous research has investigated the role of supportive care on patient outcomes, and found a relationship with reduced hospitalizations and adverse events 14 ; such therapies could also be hypothesized to similarly impact quality of life and such research is needed.
The use of standard, validated instruments in this trial allow for the ability to understand the patient perspective during treatment. The baseline scores observed in this study (78.3, physical functioning; 66.8, global health status; 29.9, fatigue; and 6.5, nausea/vomiting) are consistent with reviews of prior quality of life research conducted among patients with sarcoma, 15 and support the growing body of evidence showing that these patients experience clinically meaningful problems when diagnosed with advanced disease. In a study of patients with non-small cell lung cancer, mean QLQ-C30 scores for patients in the first-line treatment setting were 66.4 (physical functioning), 57.6 (global health status), 45.0 (fatigue) and 10.8 (nausea/vomiting). 16 In a randomized trial of doxorubicin versus paclitaxel for the treatment of patients with breast cancer, mean baseline QLQ-C30 scores were 65.5 (physical function), 60.1 (global health status), 37.2 (fatigue) and 8.8 (nausea/vomiting). 17 The values observed in the current study are comparable to those experienced by patients with other advanced/metastatic cancers.
Several other studies have used the QLQ-C30 in trials investigating sarcoma. In a single-arm phase II study of pegylated liposomal doxorubicin for soft tissue sarcoma, baseline scores were 63.5 (physical function), 50.0 (global health status), 33.8 (fatigue), and 11.6 (nausea/vomiting). 18 In a randomized trial of eribulin versus dacarbazine patients with advanced STS, mean baseline physical functioning was 76.6, global health status was 65.1, fatigue was 31.7, and nausea/vomiting had a mean score of 7.9. 19 This study, similar to others, shows that patients initiating systemic therapy for advanced or metastatic sarcoma present with clinically meaningful problems with physical function and nausea/vomiting, before initiating systemic therapy. Those with the fewest problems at baseline, not surprisingly, were able to receive the study intended dose, whereas those with greater problems at baseline received a lower cumulative doxorubicin dose throughout the study. This suggests that baseline patient-reported outcomes may help predict those least likely to receive their planned treatment, but additional research is needed to evaluate this.
It is important to note the limitations of this study. Patients were not assigned to doxorubicin dose groups; rather, all patients were eligible for and were enrolled to receive up to eight cycles of doxorubicin at 75 mg/m2 for a total cumulative dose of up to 600 mg/m2. As a result, the design of this study does not allow for statistical comparisons to be made between agents or by dose group. Furthermore, this trial design included doxorubicin therapy that was of longer duration/cumulative dose than is typically used in clinical practice, and the majority of patients received dexrazoxane during study participation, most frequently initiating at cycle 1 or cycle 5. Real-world data have shown that patients typically receive up to 5.5 cycles of doxorubicin-based therapy. 20 The findings from this study are consistent with real-world data observed in other real-world studies of doxorubicin that show a median duration of first-line therapy of only 4.1 months. 21 Therefore, the data from the ANNOUNCE trial may not adequately reflect what occurs in everyday practice as patients received a median of six cycles of therapy on the ANNOUCE trial. To account for this difference, patients were described in groups based on the cumulative dose of doxorubicin they received during the trial to be more applicable to the cumulative doxorubicin received in routine practice. Patients who received lower doses of doxorubicin in ANNOUNCE would have discontinued earlier than those receiving the highest doses remained on therapy longer without disease progression. Therefore, comparisons between these groups are not appropriate as patients were not randomized to these groups but resulted in them due to time to disease progression; future research is needed to investigate this relationship as the current study design does not allow for comparative or causal inference to be made.
It is important to note that the primary reason for study discontinuation was due to disease progression or death (87.7% randomized to doxorubicin plus olaratumab and 86.1% of patients randomized to doxorubicin plus placebo). 5 Discontinuation due to adverse events or inability to tolerate treatment was rare, and was also similar between arms (2.4% and 3.7% of all patients randomized to the olaratumab and placebo arms, respectively). 5 Therefore, it is unlikely that adverse events could have solely contributed to the observations in the patient-reported outcomes observed in the three doxorubicin dose groups, but this has not been explored in this study. Future research is needed to explore the relationship between patient-reported outcomes and adverse events in a different cohort of patients where higher rates of adverse events were observed. The role of disease progression, which was the primary reason for treatment discontinuation in this study, related to PRO outcomes may warrant future investigation.
The relationship between tumor response and patient quality of life has been explored in prior work to evaluate the relationship between disease growth and patient experience, primarily to estimate utility values for economic modeling purposes. 22 There were few tumor responses observed in this study (complete responses were observed in 0.8% versus 0.4% of patients and partial responses were observed in 13.2% versus 17.9% of patients randomized to the doxorubicin plus olaratumab arm versus doxorubicin plus placebo arm, respectively). 5 These low numbers do not permit further analyses by tumor response from this dataset, which limits the ability to further explore this question using these data.
The ANNOUNCE trial had relatively strict eligibility criteria, so the typical factors that are known to be associated with poor outcomes have already been accounted for (e.g. patients with poor performance status (ECOG >1) were excluded, patients with significant cardiovascular comorbidities were excluded, no prior exposure to anthracyclines was allowed, all patients had similar disease status upon study entry). 5 Therefore, the relationship between PROs and disease progression or tolerance to therapy may be investigated without the need to control for many of these covariates.
While data in this study are reported descriptively and no hypotheses were tested, there are a number of observations that can be made about this cohort. At baseline patients had comparable scores on physical function, global health status and nausea and vomiting, as well as similar current and prior 24-h pain recall scores. Patients who subsequently discontinued therapy before reaching a cumulative dose of 450 mg/m2 had average baseline fatigue score of 34, which was more than 10 points higher (representing worse fatigue) than those who were able to receive >600 mg/m2 in this study. This may suggest the need to further explore patient-reported fatigue and its causes in the context of this disease and the subsequent ability to receive planned doxorubicin-based therapy.
Future research may wish to explore the relationship between decline in PROs and clinical outcomes. While this study noted changes that met accepted criteria for a meaningful change, these changes were not evaluated to identify correlations with clinical outcomes in this study. There may be additional relationships between the adverse event profile of patients and PROs that could be explored. This study provides baseline data to generate hypotheses for future study. The detailed descriptive data provided are intended to be a resource for future study, so that studies can be adequately designed and powered to improve outcomes and address the short times observed to worsening of these patient-reported outcomes.
All outcomes measured reflected a median of approximately 2 months or less from treatment initiation until worsening. There is a need to identify treatments that not only extend life in this population, but importantly to reduce the burden on health-related quality of life, which declines quickly with doxorubicin-based therapy. These factors should be considered in the context of the number of cycles a patient is expected to receive in practice. Future trials should incorporate PRO outcomes and develop strategies to improve the outcomes experienced by patients with STS.
Supplemental Material
Supplemental Material - Quality of life of patients with soft tissue sarcoma treated with doxorubicin in the ANNOUNCE phase III clinical trial
Supplemental Material for Quality of life of patients with soft tissue sarcoma treated with doxorubicin in the ANNOUNCE phase III clinical trial by Brian A Van Tine, Anders Krarup-Hansen, Lisa M Hess, Albiruni R Abdul Razak, Victoria Soldatenkova, Jennifer Wright and Se Hoon Park in Rare Tumors
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LMH, VS and JW are employees of Eli Lilly and Company. VS and JW are stock shareholders of Eli Lilly and Company. ARAR serves on an advisory board to Eli Lilly and Company. Funding was provided to the following institutions: Washington University (BAVT), University Hospital Herlev, Denmark (AK-H), Princess Margaret Cancer Center (ARAR), and Samsung Medical Center (SHP) for patient enrollment and study conduct.
Funding
Ethical approval
The protocol was approved by the institutional review board at each participating center, and the study was done in accordance with the Declaration of Helsinki and the International Conference on Harmonisation Guidelines for Good Clinical Practice.
Informed consent
All patients provided written informed consent to participate in the study.
Contributorship
BAVT. AKH, ARAR, JW, and SHP all contributed to the concept and design of this study; VS and LMH developed the analysis plan, VS conducted the analysis; all authors contributed to the interpretation of findings; LMH drafted the manuscript; all authors contributed to the scientific content through important intellectual contributions. All authors have reviewed and approved the final work for submission.
Data availability statement
Lilly provides access to all individual participant data collected during the trial, after anonymization, with the exception of pharmacokinetic or genetic data. No expiration date of data requests is currently set once data are made available. Access is provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. Data and documents, including the study protocol, statistical analysis plan, clinical study report, blank or annotated case report forms, will be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at ![]() .
.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
