Abstract
Adrenocortical carcinomas (ACC) are aggressive tumors with a poor prognosis. Histological scores are advised for the diagnosis, however, there are borderline cases that may be misjudged as adrenocortical adenomas (ACA). The three main scores used are: Weiss Modified System (WMS), Reticulin Algorithm (RA), and Helsinki Score (HS). We intend to compare the accuracy of the three scores in ACC diagnosis and to identify predictive factors of overall survival (OS). Retrospective study (2004–2016) at Centro Hospitalar e Universitário de Coimbra of the adrenal tumors, classified as ACC or ACA, with a history of posterior tumor relapse/metastases, without lesions in the contralateral adrenal gland: 13F and 6M, with a median age of 51 ± 12.41 years. Nodules’ median size was 9.20 ± 6.2 cm. Patients had a median OS of 52 ± 18.6 months, with 57.9% and 46.3%, at 3 and 5 years. Seven patients had local recurrence and nine had metastases. Thirteen cases were in stage II. The WMS and the HS allowed a diagnosis of ACC in 15 cases and the RA defined ACC in 17 cases. All cases had, at least, focal disruption of the reticulin framework. More than 5 mitosis/50 HPF was associated with worse OS: 49.67 ± 21.43 versus 108.86 ± 14.02 months (p = 0.026). In patients with stage II, tumor size ⩾10 cm was associated with worse OS: 19.25 ± 7.15 versus 96.11 ± 16.7 months (p = 0.007), confirmed by multivariate analysis (p = 0.031). The correct diagnosis of ACC is a pathologist responsibility. The RA seems the most accurate. Any loss of the reticulin framework should raise awareness for malignancy. In patients on stage II, a size ⩾10 cm is a predictor of worse prognosis.
Keywords
Introduction
Adrenal tumors affect 3%–10% world population, 1 being adrenocortical carcinoma (ACC) a rare endocrine tumor1,2 characterized by a poor prognosis2–4 and a high relapse rate, with an incidence of 0.5–2 people per million per year.5–7 ACC’s occur more frequently in females and commonly in adults it the fifth decade as well as in children.8,9 ACC has poor clinical outcome in patients with substantial morbidity and mortality. 6
ACC can be symptomatic, exhibiting symptoms due to hormonal excess production such as hypercortisolism and/or hyperandrogenism, abdominal/flank pain,1,9,10 fever (if necrotic—rare event) or as metastatic disease. 10
The diagnosis algorithm of ACC demands detailed hormonal evaluation to identify autonomous excess of glucocorticoids, mineralocorticoids, androgens and adrenocortical steroid precursors, and imaging modalities.1,10 The biopsy usually is not indicated 11 and surgery (complete en bloc resection with loco-regional lymphadenectomy) is usually the treatment of choice. 12 The final diagnosis is provided by histological examination, resorting to several scores that combine morphology and ancillary techniques. 13 Adjuvant mitotane treatment is recommended for patients with a high risk of recurrence. Other therapies, such as radiation treatment or cytotoxic drugs might also be considered in an adjuvant setting for selected patients. 14
ACC can be very aggressive, with stage-dependent survival, assessed according to the European Network for the Study of Adrenal Tumors (ENSAT): 66%–82%, 58%–63%, 24%–50%, and 0%–17% for stages I, II, III, and IV, respectively. 8 Even among the different stages, the prognosis is heterogeneous, prompting for incorporation of additional factors to a more accurate biological potential assessment of the disease.
Therefore, the correct diagnosis of ACC is fundamental for patient stratification. In the majority of cases the distinction between adenoma and ACC is easy, with the correct application of a histological score such as the Weiss Modified Score (WMS). 15
However, the pathologic diagnosis of ACC is not always straightforward and sometimes the distinction between adrenocortical adenoma (ACA) and ACC is difficult, even for expert pathologists.3,4,8,10,16,17 In nearly 10% of the cases, only one or two of the criteria recognized in the WMS are identified and thus the final score is not enough for diagnosing malignancy, despite the suggestion of uncertain malignant potential. 18 These borderline cases are worrying from the clinical perspective since they present a question regarding management and follow-up.
In the latest years other histological scores were developed, namely the reticulin algorithm (RA) and the Helsinki score (HS), being appointed as more specific than the WMS.
Once the diagnosis of ACC, there are some factors that have been described related with worse prognosis, such as higher size, weight, mitotic activity (over 5 mitoses/50 high-power fields), Ki67 proliferative index, p53, among others.12,15,19
The objective of this research is to compare the accuracy of the three histological scores, WMS, RA, and HS, in the diagnosis of ACC and to identify clinical and pathological features associated with the worst outcome.
Materials and methods
Retrospective study of all patients with age over 18 years-old submitted to adrenal gland surgery between 2004 and 2016 at our institution—Centro Hospitalar e Universitário de Coimbra, a tertiary and reference hospital. Patients’ data were retrieved from the hospital’s electronic clinical records.
Consent has been obtained from each patient or subject after a full explanation of the purpose and nature of all procedures used and the local ethics committee approved the study.
We included all the patients with adrenal cortex tumors, with the diagnose of ACC or ACA with a history of posterior tumor relapse or metastases reported, without lesions in the contralateral adrenal gland. Oncocytic adenomas were excluded.
Nineteen cases fulfill the criteria and were included: 13 (68.4%) female and 6 (31.6%) male, with a median age of 51 ± 12.41 years. Seven patients (36.8%) had age inferior to 50 years old and the remaining were under 70 years old.
Regarding clinical presentation 11 (57.9%) were incidentally discovered, 4 (21.1%) presented with pain and 4 (21.1%) had Cushing syndrome. Subsequent laboratory investigation showed that one patient with an incidental discovery of ACC and one patient that presented with pain also had hormone production (cortisol). One patient had diabetes and four had dyslipidemia.
All patients were submitted to single adrenalectomy. In 12 patients (63.2%) the tumor was located in the right gland and in seven (36.8%) in the left gland. The glands had a median weight of 203 ± 705.58 g; in 10 patients (52.6%), the gland weighed over 200 g. The median size of the nodules was 9.20 ± 6.2 cm and, and in nine patients (47.4%) the tumor had a size superior to 10 cm.
The tumors were classified according to three different histological scores. To perform the WMS the following features were assessed, according to the Johnson et al. 15 : clear cells comprising ⩽25% of the tumor (×2) + confluent necrosis + over than 5 mitosis/50 high-power fields (×2) + atypical mitoses + capsular invasion. To each feature is attributed a number, 0 if absent and 1 if present, with an assumption of malignancy if the final score is three or more.
The assessment of ACC with the RA was performed by the stated by Volante et al. 20 : ACC was assumed whenever there was an altered reticulin framework associated with at least one of the following parameters: necrosis, high mitotic rate, and vascular invasion. There was an alteration of the reticulin framework when there was a focal or a diffuse disruption.
The HS was determined according to the stated by Pennanen et al. 21 : 3 × mitotic rate (over 5/50 high-power fields) + 5 × presence of necrosis + proliferation index in the most proliferative area of the tumor. ACC is diagnosed if the final score is superior to 8.5.
Slides were reviewed by two experienced pathologists (RCO and MJM). A reticulin stain was performed as well as immunohistochemistry for P53 (DO7, Ventana, Tucson, AZ-USA) and Ki67 (MIB-1, Dako, Hamburg, Germany) in one representative block of the lesion.
P53 was considered overexpressed whenever there was a null phenotype or there was nuclear expression in more than 50% of the tumor cells.
The evaluation of the Ki67 proliferative index was registered as the percentage of cells with nuclear expression/number of total cells in a high-power field, at the site of its maximum expression, the so-called “hot-spot.” The number of cells were manually counted in a printed image, in a similar fashion to the evaluation performed on neuroendocrine tumors. 22
Staging was performed according to the European Network for the Study of Adrenal Tumors (ENSAT) classification 23 : stage I if tumor ⩽5 cm and confined to the adrenal gland; stage II for as intra-adrenal ACC with more than 5 cm; stage III if regional nodal involvement or extra-adrenal dissemination of the tumor; stage IV when there were distant metastases.
All stained slides were observed under a light microscope—Nikon Eclipse 50i—and images were obtained using a Nikon-Digital Sight DS-Fi1 camera.
Metric variables were described by median ± standard deviation (SD), and mean values were compared using Student’s t-tests. Categorical variables were described by absolute and relative frequencies, and the distributions were compared using Chi-square tests. Survival was obtained using Kaplan–Meier curves with log-rank comparison. Multivariate analysis was performed using Cox regression. A p-value <0.05 was considered representative of statistical significance. Statistical calculations were performed with SPSS (Version 22.0, Chicago, IL).
Results
The patients were followed up by a median of 20 ± 45.98 (range 3–162 months), with a median overall survival (OS) of 52 ± 18.6 months (48.6–121.8). The OS at 3 and 5 years was 57.9% and 46.3%, respectively. Seven patients (36.9%) had tumor local recurrence and nine (47.4%) had metastases. The median of disease-free survival (DFS) was of 12 ± 40.9 months (0–124).
Regarding staging, when applied the ENSAT staging system, the majority of the tumors were in stage II—13 cases (68.42%); four cases (21.05%) were in stage III and two cases in stage IV (10.52%). There were no stage I tumors. All patients underwent adjuvant therapy with mitotane.
The clinical and pathological features can be consulted in Table 1.
Clinical and pathological features of the adrenocortical carcinomas.

On gross examination, the majority of tumors were well delimited and yellow, with necrotic areas, cystic features and, sometimes had a soft consistency. Some of the gross features of the specimens can be observed on Figure 1.
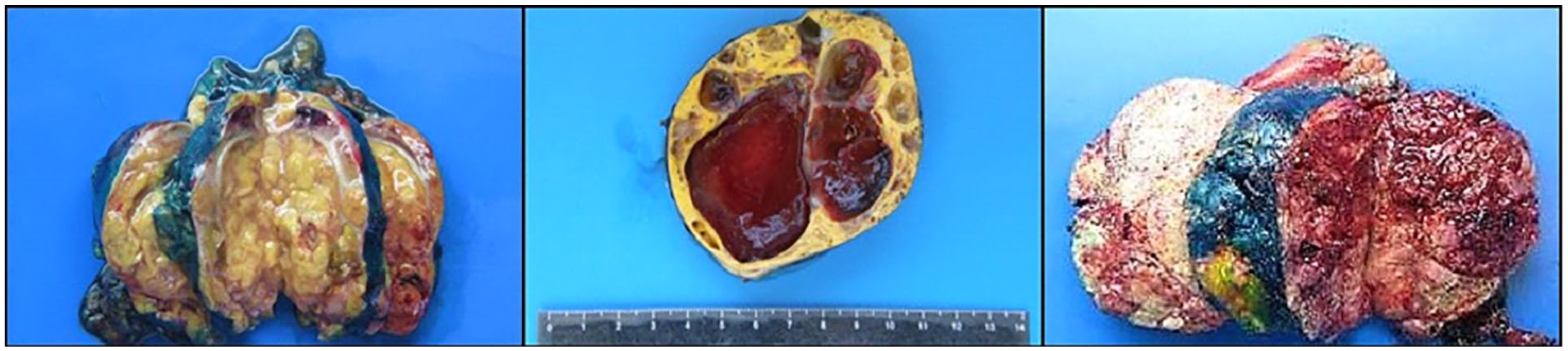
Gross examination of adrenal cortical carcinomas: soft consistency (left side), cystic degeneration (central portion of the image) and abundant necrosis (right side). Tumors show a characteristic yellow color.
When it comes to conventional histological examination, there was necrosis in 15 of the tumors (78.95%) and vascular invasion in 12 (63.16%). Ten of the nodules exhibited capsular invasion (52.63%).
There was a median of 10 ± 10.91 mitosis/50 high-power fields (range 1–40) and in 12 patients (63.16%) it was registered more than 5 mitosis/50 high-power fields. All lesions exhibited disruption of reticulin framework, 16 in a diffuse pattern and three with focal loss. P53 staining had an abnormal pattern in 12 patients (63.16%), with a null phenotype in six and expression in more than 50% in another six patients.
The Ki67 proliferative index had a median of 9.2 ± 13.55 (range 2–44), with 10 patients (63.16%) displaying an index superior to 5%.
Representative pictures of these findings can be observed in Figure 2.

Microscopic evaluation: nuclear pleomorphism and mitotic activity (a), necrosis (b), vascular invasion (c), reticulin stain with loss of fibers (d); Ki67 proliferative evaluation—60% (e) and diffuse p53 nuclear staining indicating p53 mutation (f).
Concerning the application of the histological scores, both the WMS and the HS allowed a diagnosis of ACC in 15 cases (78.9%) and the RA defined ACC in 17 cases (89.5%), as displayed in Table 2.
Classification of the tumors of the adrenal cortex and their diagnosis when applied to our cohort, based on the application of the three systems.

Higher staging was associated with worse survival with a mean of 77.23 months for stage II and a mean of 55.83 months for stage III/IV, but without statistical significance (p = 0.143). Vascular invasion, necrosis, and capsular invasion when analyzed individually had worse OS, but also without significance (p > 0.05). The same results were present for size superior to 10 cm (p = 0.067) and abnormal P53 expression (p = 0.673).
There was an association between more than 5 mitosis/50 high-power fields and worse OS—mean of 49.67 ± 21.43 versus 108.86 ± 14.02 months (p = 0.026), as seen in Figure 3. However, this was not confirmed by multivariate analysis (p = 0.058).
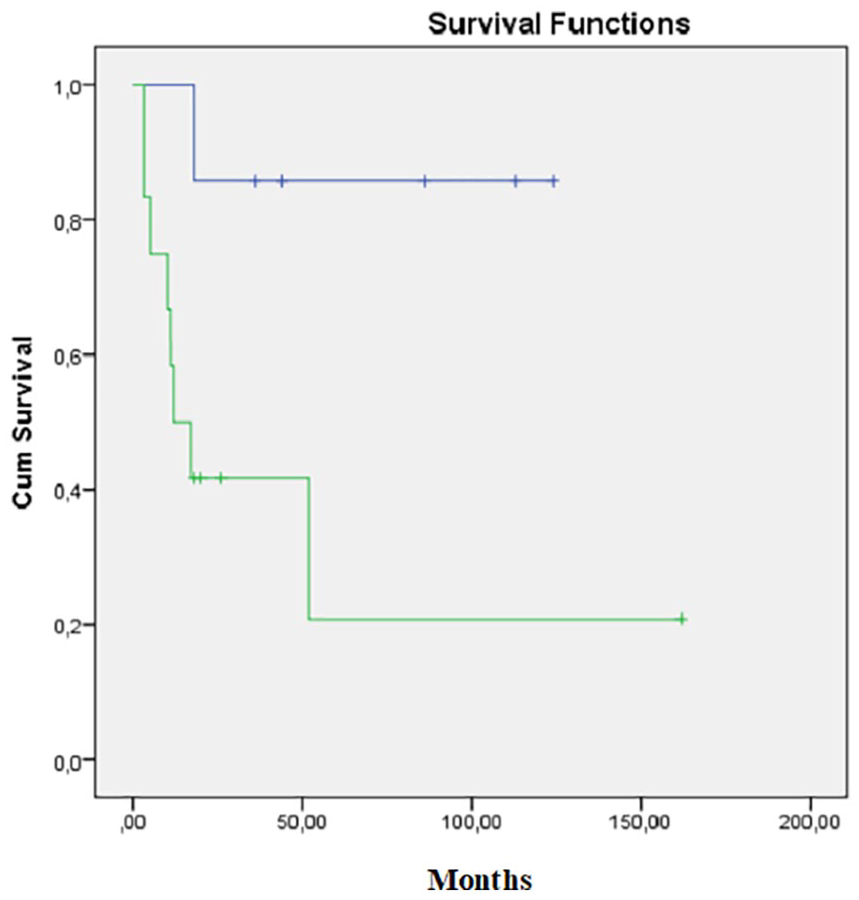
Kaplan–Meier curves regarding adrenocortical cancer with <5 mitoses/50 high-power fields (blue line) and more than 5 mitoses/50 high-power fields (green line).
More interestingly, when we study the cohort of patients with stage II disease, there was an influence of the tumor size in OS, with tumors with equal or more than 10 cm associated with worse OS—19.25 ± 7.15 versus 96.11 ± 16.7 months (p = 0.007), as expressed in Figure 4. The finding was confirmed by multivariate analysis (p = 0.031) with a hazard ratio (HR) of 13.023 (95% CI 1.273–133.261).
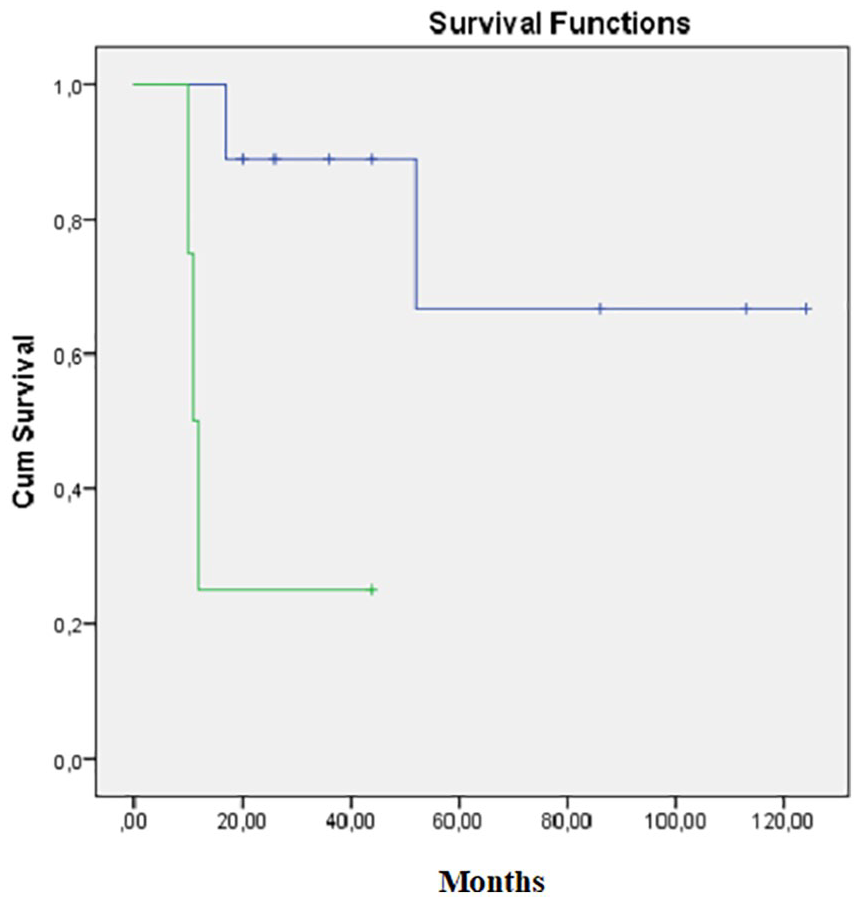
Kaplan-Meier curves regarding stage II adrenocortical cancer with less than 10 cm (blue line) and more or equal than 10 cm (green line).
In patients with stage II, the presence of more than 5 mitoses/50 high-power fields was also a predictor of a poor OS (p = 0.008), but that finding was not confirmed by multivariate analysis (p = 0.219).
No factors were identified as predictors of worse DFS.
Discussion
Adrenocortical carcinomas are rare and their diagnosis can be difficult and challenging.4,5,8 ACC are aggressive and have unfavorable overall survival, as demonstrated by our results with an OS of 52 ± 18.6 months (48.6–121.8) and 57.9% and 46.3% at 3 and 5 years. These results are in consonance with the literature.24–27
Therefore, the correct diagnosis of ACC is fundamental for patient stratification and prognosis. The final diagnosis can only be obtained after pathological examination, and in order to correct differentiate ACC from ACA the use of histological scores is recommended. 11
In 1984, Weiss studied a cohort of ACC and defined several criteria for their classification. This classification comprised the following criteria: (1) nuclear grade (1–4 according to Fuhrman classification); (2) mitotic rate (>5/50 HPF); (3) atypical mitoses (abnormal distribution of chromosomes); (4) cytoplasm features (percentage of clear or vacuolated cells resembling the normal zona fasciculata); (5) architecture of tumor cells; (6) necrosis; (7) invasion of venous structures; (8) invasion of sinusoidal structures; and (9) invasion of the capsule of the tumor (invasion of venous, sinusoidal, and capsule structures accepted only when unequivocal), thus establishing a final score from 0 to 9. 13
This classification was revised in 2002 by Aubert et al. in a study of 24 ACC with distant metastases, local invasion or recurrence and 25 adrenocortical adenomas, and developed a Weiss modified system based on the most reliable criteria: 2 × mitotic rate (>5/50 HPF) + 2 × cytoplasm + abnormal mitoses + necrosis + capsular invasion. In this system, a final score of three or more was a predictor of malignancy, achieving the specificity of 96% and the sensitivity of 100% for malignancy.13,17
Due to its simplicity, reproducibility and high specificity and sensitivity, the Weiss modified score rapidly became one of the most used histological scores. Nevertheless, some cases of ACC were not contemplated by this WMS and new scores have emerged, namely the RA and the HS, with validation in different cohorts.21,28
The best histological score is still on the debate, but judging our results the RA seems the most sensible classifying 17 lesions as malignant while the WMS and HS were only able to identify 15 lesions as malignant. Despite two of the cases were not assessed as malignant by the RA, both cases exhibit focal loss of the reticulin framework.
Based on these findings a full inclusion of the nodule may be advised on gross macroscopy and every case with focal loss of the reticulin framework, even if classified as an adrenocortical adenoma should have a very close follow-up.
Recently molecular profiling of ACC identified a consistently overexpressed gene: insulin growth factor type (IGF) 2. 29 This had led to several experiments on animal models and cell lines targeting the IGF receptor 1 that showed better diminution of tumor growth than mitotane, and when combined with both agents there was a synergistic mechanism of action. 30 Despite this finding, in 2014, Guillaud-Bataille et al. 31 performed a very interesting study where they conclude that IGF2 is associated with the growth of the tumoral cells, however, it is not the only tumoral driver for ACC, and suggest other pathways. This is also in line with the findings of Giordano et al. 32 where on a transcriptomic level the ACC were mainly associated with overexpression of IGF2, associated with a perturbation of the IGF2 locus, but also describe H19 alterations due to methylation of the H19 promoter. The study 32 also defines a two cluster molecular classification of the ACC, where the cluster I is composed of high-grade tumors and cluster II is composed of low-grade tumors, defined essentially by high and low-grade mitotic activity.
The molecular background of ACC is complex and not limited to IGF2 mutations and alterations in P53/Rb1, cyclins and topoisomerase pathways have been described. 33
Nevertheless, molecular characterization provides a lot of information that may be difficult to integrate, requiring fresh or frozen tissue. In a recent study from Assié et al. 34 molecular classification of ACC had a clinical impact only on stage IV tumors. In our cohort, the majority of tumors were in stage II, limiting this approach.
However, the molecular classification and clustering provided the emerging of new immunohistochemical markers for ACC: cyclin E was reported as a good marker for ACC when compared to ACA 32 and IGF2 has also emerged as a good immunohistochemical marker for maligancy.35,36
In the latest years p57 and SF-1 immunohistochemical detection has arisen as a promising marker for malignancy, 37 especially in pediatric tumors, 38 but other studies have revealed that SF-1 is a more sensitive marker for adrenal lesions and adrenal cortex rather than for malignancy. 39
However, despite the sensibility of the markers for the diagnosis of ACC, they do not translate the biological behavior of the tumor nor its impact on OS.
In the management of patients ACC, some authors have pointed out that higher size and weight, p53 status, mitotic activity, and Ki67 proliferative index are associated with worse prognosis and should be used to stratify patients with higher risk.12,15,19
The tumor stage is also associated with OS, with worse outcome in higher stages. 40 In the patients at stage II—the majority of our cohort, tumor size equal or superior to 10 cm, showed an effect on OS in multivariate analysis, with a high hazard ratio. This finding supports that simply diagnosing ACC is not enough for proper patient management and that more data should be integrated. In this study, despite association on univariate analysis, there was no association in the multivariate analysis for high-mitotic activity (defined as more than 5 mitoses/50 high-power fields). The mitotic activity has been recorded in the literature as a feature of worse prognosis 8 as we also believe it should be a very important characteristic for patient stratification.
In this context, the histological scores possess the double advantage of allowing lesion classification and the identification of features associated with a poorer outcome.
The phenotypic and immunohistochemical alterations should be in close relation with the molecular clustering, and in the future, an integrated classification should be employed, especially for patients in advanced stages of the disease.
Conclusion
The correct diagnosis of ACC is mainly the pathologist’s responsibility. An accurate histological score should be employed and any loss of the reticulin framework should raise concern for malignant behavior. In our cohort, the RA was the most effective for ACC diagnosis. In patients with ACC diagnosis and on stage II, a size equal or superior to 10 cm is a predictor of a worse prognosis.
Efforts should be performed in order to identify patients for correct stratification and individualized follow-up, and tumor size should be incorporated in patient’s reports and treatment algorithms.
Footnotes
Authors contribution
RCO and MJM designed the study, wrote the manuscript and performed histological analysis of the cases. CM, JC, RA and ETS collected clinical data. PT performed ancillary studies. MT performed statistical analysis. IP, AF and MAC supervised the research and added critical content to the manuscript. All authors have read and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The local ethics committee approved the study.
Informed consent
Consent has been obtained from each patient or subject after full explanation of the purpose and nature of all procedures used
