Abstract
Schwannoma, otherwise known as neurilemmoma, is a benign tumor that originates from Schwann cells. Ancient Schwannoma is a long-standing Schwannoma with degenerative and xanthomatous changes. The Head and neck region is one of the most common sites for nerve sheath tumors but Involvement of the jaw is a considerably infrequent event. We report a rare case of intraosseous ancient Schwannoma of the mandible in a 24-year-old male patient which was initially misdiagnosed with carcinoma. The clinical features, radiographic findings, and treatment plan are discussed. Furthermore, a thorough literature review demonstrated nine published cases of intra-osseous ancient Schwannoma with their summarized features. Recognition of various histopathologic features of ancient Schwannoma is essential to prevent over-treatment.
Introduction
Schwannoma is a benign, slow-growing, peripheral nerve sheath tumor that derives from Schwann cells of myelinated nerves.1–3 Approximately 25%–40% of all Schwannoma occur in the head and neck, especially in the soft tissue of this region. 4 While the incidence of Schwannoma is uncommon, 5 Intraosseous Schwannoma are very rare and account for less than 0.2% of all primary bone tumors.6–9 Ancient Schwannoma of the intraoral region is still extremely rare and so far only few cases have been reported in English literature.10,11 The term ancient Schwannoma which was first presented by Ackermann and Taylor in 1951, refers to a long-standing Schwannoma with degenerative changes such as calcification, cystic formation, necrosis, xanthomatous changes, vascular hyalinization, and obvious nuclear atypia.12–14 These Intra-tumoral- related degenerative changes are attributed to an increasing in tumor size and tumor aging.12,13 In this case report, we represent a rare case of recent intraosseous ancient Schwannoma in the mandible with extensive xanthomatous changes.
Case report
A 24-year-old man was referred to the Otolaryngology department at Cancer Institute of Imam Khomeini hospital complex, Tehran, Iran in January 2017, with right facial swelling without any pain and paresthesia for more than 1 year. Intraoral examination revealed right mandibular expansion with firm consistency and no tenderness. The patient had undergone an incisional biopsy under local anesthesia at another center and the diagnosis of Carcinoma with clear cell features was designated.
Panoramic radiography revealed a well-defined multilocular radiolucent lesion with a cortical border in the right posterior mandible, extending from the second premolar to the ascending ramus and sub-condylar area. Some degree of apical root resorption is seen in both right first and second mandibular molars (Figure 1).
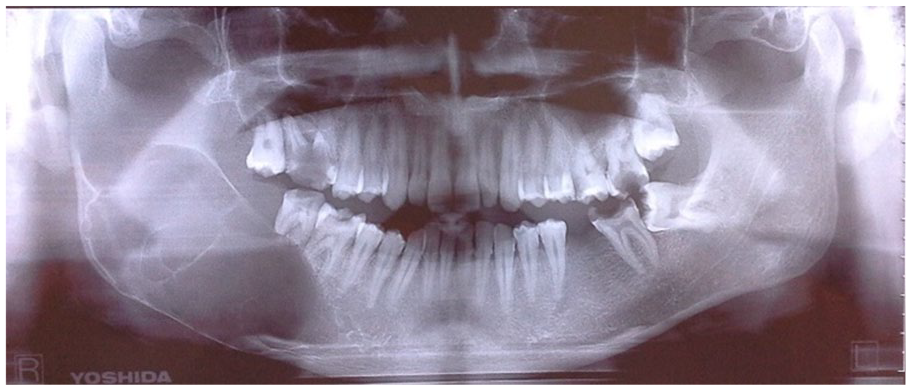
Panoramic view illustrates a well-defined multilocular radiolucent lesion at the right posterior area of the mandible.
In the axial and coronal computed tomography (CT) scan slices without contrast, a radiolucent lesion measuring 50 mm × 40 mm × 30 mm was seen in right mandibular angle with a homogenous density which caused mandibular expansion and cortical plates perforation (Figure 2(a) and (b)). Based on radiographic and clinical findings differential diagnosis comprised odontogenic tumors and cysts including Ameloblastoma, long-standing odontogenic keratocyst, and non-odontogenic lesions such as central giant cell lesions, intraosseous carcinoma, and infrequently central neural and vascular neoplasms or abnormalities. 15 Since various benign and malignant differential diagnoses were considered based on clinicoradiographic features, the specimen was reviewed for the second time by a pathologist at another center. This time, the histopathologic report proposed low-grade carcinoma with xanthomatous changes, and the definite diagnosis was deferred to whole surgical excision.

Axial (a) and coronal (b) CT scan images reveal mandibular expansion and cortical bone perforation.
Subsequently, the patient underwent a right Hemi-mandibulectomy under general anesthesia. The mandible was reconstructed at the region by fibular bone. On gross examination, the specimen was consisting of a segment of the right posterior mandible from lower right first molar tooth to ascending ramus measuring 6.5 cm × 4.5 cm × 2 cm and 9 cm × 4.5 cm × 3 cm, respectively with a mass within the mandible bone measuring 9 cm × 4.5 cm × 3 cm which led to expansion, lateral mandibular cortex perforation and expansive deformity of the inferior alveolar canal (Figure 3). Cut sections revealed a solid non-homogeneous tan- brown tissue which covered by thin capsule in some area. Microscopic examination showed a neoformed tissue composed of hypo and hypercellular areas of elongated to spindle-shaped cells with fusiform nuclei demonstrating some degrees of atypia and ill-defined cytoplasmic border arranged in short fascicles. Tumoral cells were mainly infiltrated by a few foamy to clear histiocytes in the whole lesion. Immunohistochemical (IHC) staining for S100 protein and Vimentin revealed diffuse strong positive reactivity in elongated spindle cells. CD68 was positive in numerous large foamy macrophages, perhaps Xanthoma cells. After multiple serial sectioning, more degenerative changes including sheets of hemorrhage and Xanthomatous cells were detected (Figure 4(a)–(c)). Immunohistochemical staining for Ki67 indicated 2%–3% positivity in tumoral cells (Figure 4(d)). No pan CK, P63, SMA, H-caldesmon was detected in tumor cells.

Gross examination of specimen revealing parts of the right mandibular body and ramus demonstrating tan-gray intrabody mass with buccal cortical plate perforation and 2.5 cm lobular protrusion.

Photomicrographic images of histologic sections: (a) H & E stained section illustrating encapsulated spindle cell neoplasm with extensive Xanthomatous degenerative changes (×40), (b and c) immunohistochemical stained section with S100 protein showing diffuse strong nuclear and cytoplasmic positivity of tumoral cells (b: ×40, c: ×100), and (d) immunohistochemical stained section with Ki67 revealing around 2%–3% proliferative activity in tumoral cells (×100).
Considering all these mentioned features, the diagnosis of ancient Schwannoma was completely confirmed. Postoperatively, the patient did well, and he experienced no major complications. Follow-up for 4 years duration declared no evidence of wound dehiscence, infection, or even recurrence (Figure 5(a) and (b)).
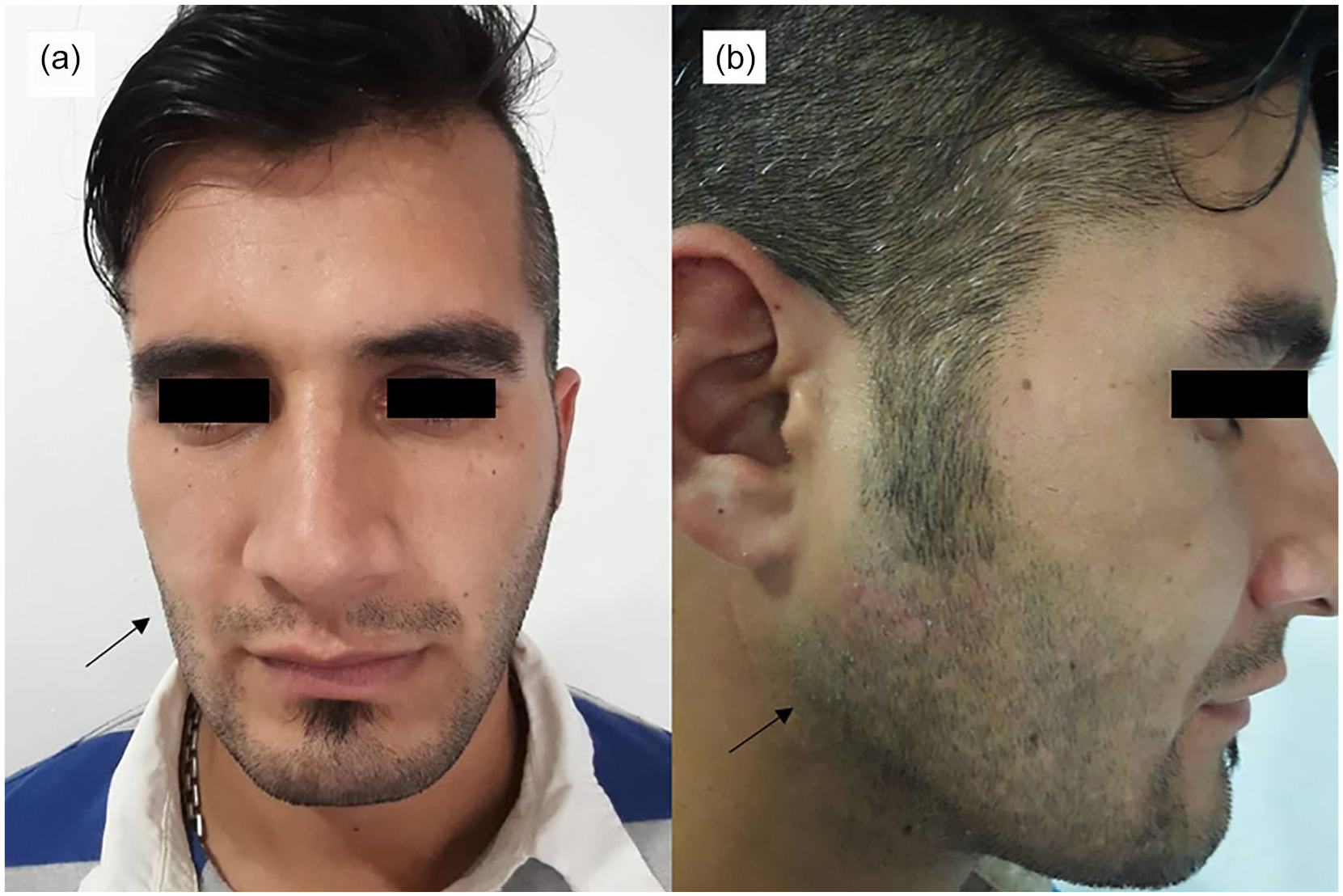
Frontal (a) and lateral (b) clinical view of the patient showing no recurrence of lesion 4 years post-operation.
It should be noted that we obtained written and signed informed consent from the patient for publishing this case report.
Discussion
Ancient Schwannoma is a slow-growing neurogenic neoplasm with a large size and long extent of duration.6,14 It rarely affects the head and neck region and intraoral lesions are even scarcer Eversole and Howell 13 reported the first ancient Schwannoma of the head and neck area. Intraosseous Schwannomas are rare, but when they occur, the mandible is the most common site, particularly in the posterior areas of the body and ramus. 12 In the scope of our knowledge, only nine cases reported ancient Schwannoma in the jaw (Table 1). This information reflected the rare incidence of called tumor in the jaw. In our literature review, based on available information, the lower jaw was the most common site of engagement (88%), only one of them occurred in the hard palate. 16 In the present case, the lesion was located in the posterior mandible of 24 years old man.
Summarizes of published case reports with ancient Schwannoma in the jaw.

F: female; M: male.
Clinically, Ancient Schwannoma seems to be a confined slow-growing, firm, non-tender mass. Although most of the cases are asymptomatic, pain and paresthesia may be found in some patients, especially in cases of Intraosseous engagement. 16 In our review, even though all the cases occurred in the jaw but only 44% of them had pain or paresthesia as a clinical symptom.
Histopathologically, the term “ancient Schwannoma” is applicable when hemorrhage, hemosiderin deposition, chronic inflammatory changes, and pleomorphic cells are detected in the lesion. 1 These features can make the diagnosis difficult and may lead to an erroneous diagnosis of malignancy by pathologists.24,25 The reason for sharing this case was its especial microscopic feature which was initially mistaken for malignancy. Immunohistochemistry is highly beneficial to confirm the diagnosis of Schwannoma, especially those with extensive degenerative changes and a lack of common histopathologic features. In this regard, immunostaining for vimentin, S100, LEU7, and myelin basic protein (MBP) markers are suggested.1,2 In such complicated cases, it is better to do a panel of immunohistochemistry markers to achieve the correct diagnosis and to prevent the application of inappropriate treatment protocol. 24 In the present reported case, Immunohistochemical staining was applied as a valuable approach for ascertaining the diagnosis. Accordingly, the diagnostic immunohistochemistry revealed strong positive signals of S100 and Vimentin in focal spindle-shaped cells. Also, large xanthoma cells showed immunoreactivity with cd68, a marker of histiocytes.
Complete excision of the lesion seems to be a golden standard treatment for ancient Schwannoma. 16 This is a similar approach to all the cases reported in this literature review including our case. It is important to note that a pathologist must be precautious about the atypical features of this type of tumor that may lead to misdiagnosis of a malignancy. 6 There are two case reports of ancient Schwannoma with malignant transformation, but no intra-oral ancient Schwannoma reveals such a malignant transformation.6,25 Long-term clinical and radiological follow-up of patients is mandatory to ensure good results. In our study, the patient was followed up for 4 years with no clinical and radiographic signs of recurrence.
Conclusion
In conclusion, ancient Schwannoma has no different clinical behavior and treatment than usual Schwannoma. Recognition of histopathologic features of ancient Schwannoma, especially those with subtle or absent typical microscopic criteria is crucial to prevent over-treatment. 12
Footnotes
Acknowledgements
We would like to thank our patient for his cooperation.
Contributorship
All authors reviewed and edited the manuscript and approved the final version of the manuscript
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article. It has been attached to the main file.
Ethical approval
Tehran University of medical science does not require ethical approval for reporting individual cases or case series.
