Abstract
Brain metastases in sarcoma are exceedingly rare, with few published series documenting ranges from 1% to 8%. This study investigated the outcomes of sarcoma patients with brain metastases using a population-based analysis. This was a retrospective review of 5933 patients with high-grade sarcoma identified from the Surveillance, Epidemiology, and End Results database between 2010 and 2015. Of the eligible 5933 patients, 0.7% (n = 44) had brain metastasis. Kaplan–Meier was used to estimate survival and follow-up (reverse Kaplan–Meier), and a multivariable Cox proportional hazards model analyzed prognostic factors of disease-free survival (DFS). Median (IQR) follow-up of all eligible patients was 28 months (12; 47). Patients who developed brain metastasis had a higher proportion of N1 stage disease (p < 0.001), as well as synchronous metastasis to bones, liver, and lungs compared to those without brain metastasis (all p < 0.001). The median (IQR) DFS with brain metastasis was 6 months (2; 12), and survival with brain metastasis was significantly worse than DFS in patients without brain metastasis (p < 0.001). Among those with brain metastasis only, there was no difference in DFS with respect to sex, race, primary tumor origin, T stage or N stage disease, synchronous metastasis to bone, liver or lung, nor with respect to chemotherapy or radiation for treatment of the primary tumor (all p > 0.05). For sarcoma patients with brain metastasis, the outcomes are poor and do not appear to differ by clinicopathologic factors. However, patients with certain histologies and synchronous metastases may warrant more frequent surveillance as there was an association of brain metastasis with these factors.
Background
Sarcomas are a rare and heterogeneous tumor group with a propensity for metastasis. Although metastases may commonly occur in the lungs, liver, and bones, distant spread to the brain is exceedingly rare. The incidence of sarcoma brain metastasis ranges from 1% to 8% according to the literature.1–5 However, given its rarity, there are few data that reliably describe the prognosis of these patients.
Historically, clinical outcomes of patients with sarcoma who develop brain metastasis are poor, and disease-free survival (DFS) ranges from about 2 to 7 months.6–10 Patterns of brain metastasis may vary by histologic subtype, and there is evidence to suggest certain histologies such as malignant fibrous histiocytoma, rhabdomyosarcoma, leiomyosarcoma, and osteosarcoma may metastasize to the brain more readily.11,12 Despite modern treatment advances, brain metastases from sarcoma are often refractory to multimodal management. Nonetheless, surgical excision and/or radiation are utilized when feasible, although the outcomes are still poor.2,7,10,13
Objectives
In an attempt to better characterize the rare event of brain metastasis in sarcoma, the current study reviewed the Surveillance, Epidemiology, and End Results (SEER) database in order to identify patients with high-grade bone or soft tissue sarcoma who developed distant spread to the brain. The primary objective was to determine patterns of brain metastasis by sarcoma subtype and clinical characteristics, and the secondary objective was to assess DFS of these patients.
Materials and methods
Selection
Ethical or institutional review board approval was not required for the completion of this study. The SEER database of the National Cancer Institute was queried from 2010 and 2015. 14 Inclusion criteria were diagnostically confirmed bone or soft tissue sarcoma and a yes/no indicator of brain metastases. Cases were first filtered by pertinent Histologic International Classification of Diseases for Oncology, Third Edition (ICD-O-3) codes. Patients with low-grade disease were excluded, as the association between high grade sarcoma and brain metastasis has been described in other studies.15–20 Cases with no available data on brain metastasis were also excluded, leaving 5933 cases for review. Variables of interest were age, sex, race, primary tumor histology, T and N stage defined by the American Joint Committee on Cancer (AJCC), and adjuvant therapy. Radiation (yes or no) and chemotherapy (yes or no/unknown) were recorded as dichotomous variables with respect to treatment of the primary tumor and not metastatic disease.
Statistical analyses
Categorical variables were described using frequencies and percentages and compared with Chi-squared or Fisher’s Exact test. Continuous data were tested for normality and reported as the median with interquartile range (IQR) and compared using Mann–Whitney U Test. DFS was estimated using Kaplan–Meier methods, and survival between variables of interest was compared using the log-rank test. DFS was defined as the survival in months from diagnosis until death or last clinical encounter per the SEER database. Censoring was of patients without death having occurred by the last recorded follow-up. The median follow-up time (months) was estimated using the reverse Kaplan–Meier method wherein survival is the event of interest and death is a censored event.
Multivariable Cox model of regression was used to assess the association of individual variables on DFS for all eligible patients. The final model was constructed using a backward stepwise elimination method of sequential variable exclusion with the highest p-value variable being excluded at each step, until only those with p < 0.15 remained. Values of 0.05 < p < 0.15 were retained in the final model as potential confounders, though were not considered to be significantly associated with DFS. Preselected variables were those of clinical interest and included diagnosis year, age, sex, race, N, and T stage, metastasis elsewhere, and radiation and chemotherapy for the primary tumor. Metastasectomy was considered for inclusion in this model, however too few patients underwent metastasectomy. Similarly, factors with less than five events per variable were excluded. Statistical significance was set to p < 0.05, and all analyses were performed on SPSS version 26.0 (IBM, Armonk, NY, USA) and R-Studio version (RStudio: Integrated Development for R. RStudio, PBC, Boston, MA, USA).
Results
Clinicopathological characteristics
The rate of brain metastasis among eligible patients with high-grade sarcoma was 0.7% (n = 44). The age-adjusted crude incidence rate of brain metastasis was 1.0 per 100,000 at-risk patients from 2010 to 2013, which increased to 1.2 per 100,000 at risk-patients from 2014 to 2015. The median age of patients who developed brain metastasis was 55.5 years (interquartile range (IQR), 42; 67). Between those who developed brain metastasis and those without, there were differences with respect to race (p = 0.021) and the proportion of primary sarcoma originating from soft tissue, bone, or cardiac/nervous tissue (p < 0.001) (Table 1). Tabulation of histologies that developed brain metastasis is shown in Supplemental Table S1. Patients who developed brain metastasis also had a higher proportion of N1 nodal disease (p < 0.001), as well as a higher proportion of synchronous metastasis to bones, liver, and lungs compared to those without brain metastasis (all p < 0.001), respectively. Among all patients, chemotherapy for the primary disease was more often used in patients with brain metastasis (p = 0.012).
Baseline clinicopathologic data among all eligible patients grouped by presence or absence of brain metastasis.

BM: brain metastasis.
median (interquartile range), compared with Mann-Whitney U Test.
AJCC seventh edition staging.
Significant difference on Mann–Whitney U test, or Fisher’s Exact or Chi-squared tests.
Adjuvant and/or neoadjuvant with respect to primary disease.
Survival and prognostic factors
According to quantiles of potential follow-up, the median estimated follow-up of all eligible patients (n = 5933) was 28 months (IQR, 12; 47). A multivariable Cox proportional hazards model of regression of all eligible patients is shown in Supplemental Table S2. For all eligible patients, 1-, 3-, and 5-year DFS estimates were 80.1% (95% confidence interval (CI), 78.9%–81.4%), 60.8% (95% CI, 59.0%–62.5%), and 53.0% (95% CI, 50.8%–55.2%), respectively (Figure 1(a)).
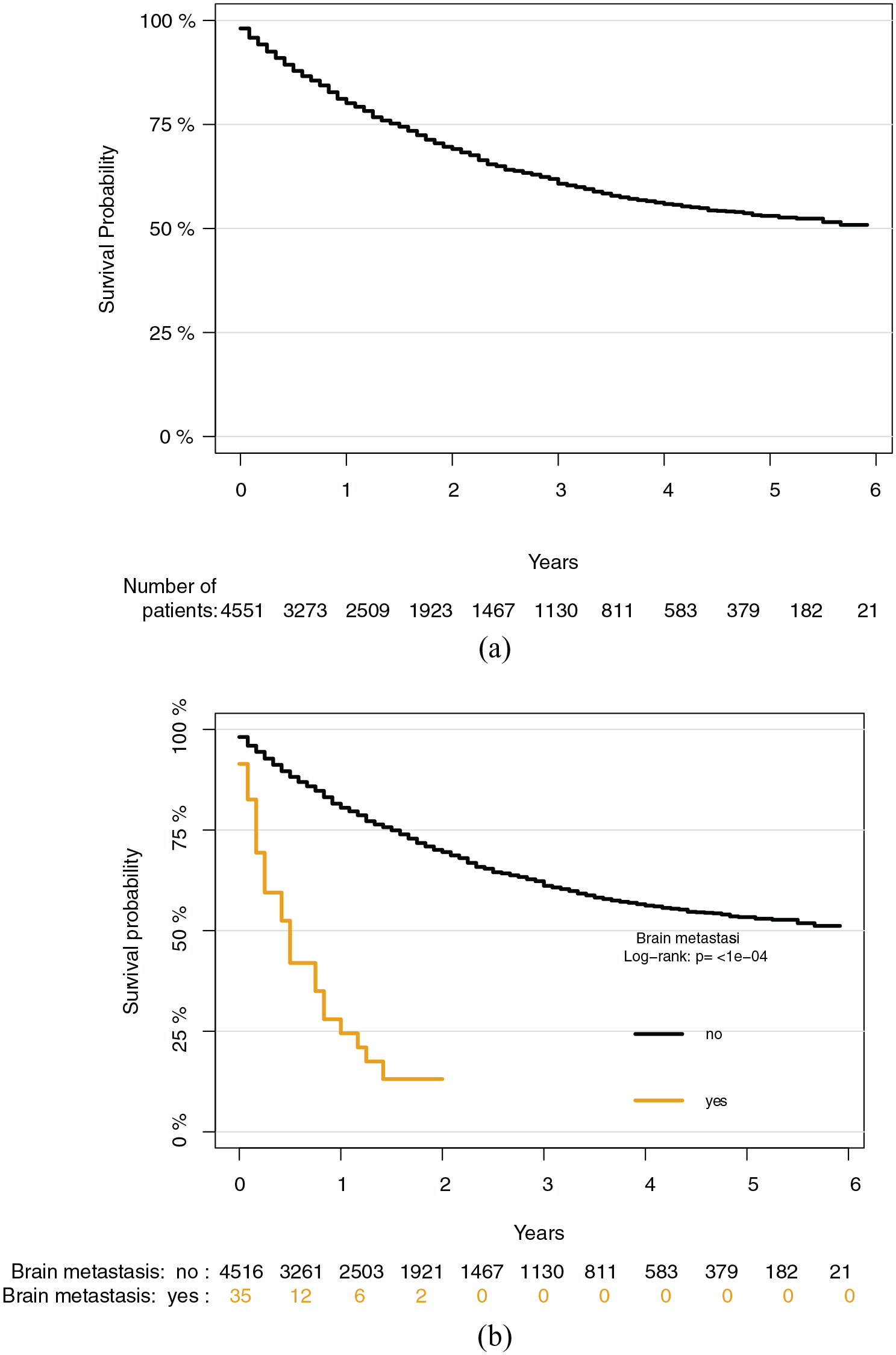
(a) Kaplan–Meier survival analysis demonstrating disease-free survival of all eligible patients with high-grade bone and soft tissue sarcoma. At-risk individuals are shown along the x-axis. (b) Kaplan–Meier survival analysis demonstrating disease-free survival between sarcoma patients with brain metastasis compared to patients without brain metastasis. At-risk individuals are shown along the x-axis. BM, brain metastasis.
For patients with brain metastasis, the median DFS was 6 months (IQR, 2; 12), with a 1-year DFS of 24.5% (95% CI, 8.9%–40.1%). In comparison, the 1-, 3-, and 5-year estimates without brain metastasis were 80.5% (95% CI, 79.3%–81.8%), 61.1% (95% CI, 59.3%–62.9%), and 53.4% (95% CI, 51.2%–55.6%), respectively (p < 0.001; Figure 1(b)). A subset survival analysis among brain metastasis-only patients is tabulated in Table 2. Among these 44 patients, there was no difference in DFS with respect to sex, race, primary tumor origin, T stage, or N stage disease, synchronous metastasis to bone, liver or lung, nor with respect to chemotherapy or radiation for the primary tumor (all p > 0.05).
Median disease-free survival and corresponding 1-year survival probabilities among brain metastasis-only patients (n = 44).

CI: confidence interval; DFS: disease-free survival. Int.: intermediate. IQR: interquartile range; NA: not available.
AJCC seventh edition staging for primary sarcoma.
Chemotherapy and radiation given for the primary tumor.
Log-rank test.
Discussion
The rate of brain metastasis in the current study was 0.7%. Furthermore, the crude, age-adjusted incidence appears to have steadily increased over the years according to the SEER database. Although this was a small increase (1.0–1.2 cases per 100,000 at-risk patients was recorded from 2010 to 2015), it is important as high-volume sarcoma centers may disproportionately encounter patients with brain metastasis.
Limited series of brain metastasis in sarcoma have been published, and the current study is the third largest identifiable study to date.3,5,6,8,12,21,22 Historically, sarcoma brain metastasis has a predilection for males, and the current study found slight male predominance.2,12,13 The current study also found that patients with brain metastasis were younger than those without, however the median age of 55.5 years in this study is quite higher than other modern reviews by Malouf et al. and Al Sannaa et al., who report median ages of 23 years and 34.8 years, respectively.8,22 A generally lower median age of brain metastasis in sarcoma may be related to more extensive use of imaging in recent years, which may result in detection at a younger age. For example, Salvati et al. describe a mean interval from sarcoma diagnosis to brain metastasis recognition of 19.8 months, which is considerably shorter than older studies in which time-to-recognition intervals were closer to 30 months. 12 Similarly, Salvati et al., along with other studies, suggest imaging usage may play a role in earlier recognition of brain metastasis in younger patients.12,13,23 This may account for an increased incidence at perhaps a younger age.
Although the natural history of brain metastasis from sarcoma has been infrequently characterized, the majority of available data document synchronous lung metastases in 50%–80% of patients.4,12,13,23 The current study recorded lung metastases in 59.1% of patients, which is consistent with other studies. Additionally, the rate of lung metastases in the current study was higher than metastases to the bones (45.5%) and liver (29.5%), and furthermore these patients also had a higher proportion of nodal disease. Thus, there is strong evidence that brain metastasis from sarcoma is seen in the setting of metastatic disease elsewhere, and this finding may help identify patients at a higher risk of brain metastasis or provide a framework for optimal surveillance of such patients.
There are certain histologic subtypes of sarcoma that have demonstrated a higher proclivity toward development of brain metastasis than others. For example, malignant fibrous histiocytoma, rhabdomyosarcoma, and leiomyosarcoma have been reported to metastasize to the brain most often, and for primary tumors of bone, osteosarcoma is most frequently identified.6,12 In the current study of 44 cases with brain metastasis, the most frequent histologic subtypes were sarcoma: unspecified (18.2%), leiomyosarcoma (15.9%), hemangiosarcoma (15.9%), spindle cell sarcoma (13.6%), osteosarcoma (13.6%), and undifferentiated sarcoma (11.4%).
Interestingly, hemangiosarcoma and spindle cell sarcoma exhibited disproportionately higher rates of brain metastasis in the current study than have been previously described. Despite limitations to the SEER database insofar as the accuracy of the subtype recorded, this may suggest a role for strategic management of high-grade hemangiosarcoma and spindle cell sarcoma patients. However, larger studies are needed to confirm this trend. Of note, there are few studies that document disproportionately higher rates of brain metastasis in alveolar soft-part sarcoma (ASPS). However, the current study failed to identify any ASPS with metastases to the brain, though the lack of ASPS may be related to reporting tendencies inherent to the SEER database.5,22,24
The disease course of sarcoma brain metastasis is aggressive, and the estimated median survival ranges from 2 to 7 months as mentioned above.6–10 The median overall survival in the current study was 6 months, which aligns with the literature that describe poor outcomes in this setting. Surgery is typically the mainstay of treatment for these patients, and while metastasectomy may have a survival benefit, the overall survival following brain surgery is still poor.2,7,10,13 Chaigneau et al. found surgery, whole-brain radiotherapy, and stereotactic radiotherapy were each associated with a lower risk of mortality. 6 However, the authors do note the magnitude of the benefit appears to be limited. Of note, the SEER database does not clearly distinguish metastasectomy by site, and therefore any distant site surgery may have been on the brain, liver, lungs, or for bone metastasis. Thus, the utility of surgical intervention in these patients at least from the current analysis is unclear, and although few studies have demonstrated a survival benefit, the true role of metastasectomy requires future prospective efforts.
Chemotherapy is typically utilized for the treatment of metastatic sarcoma, though in the current study chemotherapy failed to demonstrate a survival benefit in patients with brain metastasis. This is contrary to larger modern studies like Chaigneau et al. and Al Sannaa et al., who each identified a survival benefit with chemotherapy.6,8 For systemic disease such as brain metastasis, there is no clear consensus on the utility of chemotherapy, and furthermore these tendencies to treat are often institutionally dependent. While the results of these other larger studies are promising, the lack of specific drugs utilized for chemotherapy in the SEER database made it difficult to draw a conclusion on the role of chemotherapy in this setting. Chemotherapy was also indicated for the primary disease in SEER. Thus, in a subset survival analysis of brain metastasis only patients, radiation and chemotherapy did not influence survival. Brain metastasis may therefore be too far advanced to be successfully treated with current strategies.
The paucity of prognostic data describing sarcoma brain metastasis has recently been addressed by Patrikidou et al., 21 who identify subgroups of sarcoma patients with brain metastasis in whom aggressive therapy may improve survival. While this concept remains to be validated externally, there is likely no substitute for randomized, prospective efforts. The current study attempted to address any survival differences among brain metastasis patients by a subgroup survival analysis. However, there were no patients who we found to be at a survival disadvantage based on any of the included variables. This concept is promising and may have utility in informing physicians which patients should be considered to be screened for brain metastasis
For example, a disproportionately higher number of hemangiosarcoma and spindle cell sarcoma patients who developed brain metastasis. In addition, there were a higher number of cardiac sarcoma patients who developed brain metastasis. These findings may warrant earlier surveillance pattern in such patients. Ultimately, however, there is a need for large, multi-institute studies to identify groups of sarcoma patients with brain metastasis that can prognosticate outcomes with respect to treatment, or also with respect to histology and other demographic variables. Such studies would potentially allow for earlier intervention in high-risk patients which could drastically improve this disease course.
Limitations
There are limitations to this study, most of which are inherent to the use of a multi-institutional database. First, this study was retrospective in nature, and although it collected data from a variety of participating hospitals, the cohort of brain metastasis patients is relatively small. This is likely because the SEER database only recorded brain metastasis data from 2010 to 2015. Second, by nature of the SEER database, it was impossible to know whether these patients had, for example, multiple and bilateral metastasis not amenable to complete resection. Last, there was ambiguity in the SEER with respect to which metastatic site underwent metastasectomy (e.g. brain, liver, bone, or lung). Therefore, any metastatic surgical intervention was not studied, and we were unable to accurately distinguish brain metastasectomy only. This limits the conclusions drawn with respect to multimodal management.
Conclusions
Using a large, multi-institutional database, patients with advanced sarcoma and brain metastasis were identified and the survival of these patients was assessed. Similar to other modern series, the current population-based analysis found that sarcoma patients with brain metastasis had poor short-term outcomes. Furthermore, we found no survival difference with respect to any clinicopathological data, and therefore risk stratification of brain metastasis patients needs to be further studied. Last, it appears there may be an association brain metastasis with synchronous metastasis and histologies such as hemangiosarcoma and perhaps other cardiac sarcomas, which may have implications for surveillance in such patients.
Supplemental Material
sj-docx-1-rtu-10.1177_20363613211026151 – Supplemental material for Outcomes of brain metastasis in high-grade bone and soft tissue sarcoma: An analysis of clinicopathological characteristics and survival data
Supplemental material, sj-docx-1-rtu-10.1177_20363613211026151 for Outcomes of brain metastasis in high-grade bone and soft tissue sarcoma: An analysis of clinicopathological characteristics and survival data by Charles A Gusho, Alan T Blank and Marta Batus in Rare Tumors
Supplemental Material
sj-docx-2-rtu-10.1177_20363613211026151 – Supplemental material for Outcomes of brain metastasis in high-grade bone and soft tissue sarcoma: An analysis of clinicopathological characteristics and survival data
Supplemental material, sj-docx-2-rtu-10.1177_20363613211026151 for Outcomes of brain metastasis in high-grade bone and soft tissue sarcoma: An analysis of clinicopathological characteristics and survival data by Charles A Gusho, Alan T Blank and Marta Batus in Rare Tumors
Footnotes
Author contributions
Study concept and design: M.B., A.T.B.; analysis and interpretation of data: C.A.G.; drafting of the manuscript: C.A.G.; critical revision of the manuscript for important intellectual content: C.A.G., M.B., A.T.B.; statistical analysis: C.A.G.
Disclosure statement
ATB: BMJ Case Reports: Editorial or governing board; Clinical Orthopaedics and Related Research: Editorial or governing board; exparel/pacira: Stock or stock Options; Journal of Oncology Practice: Editorial or governing board; Journal of Surgical Oncology: ad hoc reviewer; Lancet - Oncology: Editorial or governing board; Musculoskeletal Tumor Society: Board or committee member; Onkos Surgical: Paid consultant; Pediatric Blood and Cancer: Editorial or governing board; Rare Tumors: ad hoc reviewer. All other authors have no pertinent financial disclosures or pertinent conflicts of interest.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alan Blank is an ad hoc reviewer/editorial governing board for the journal of Rare Tumors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not required for the completion of this study.
Informed consent
Informed consent was not required for the completion of this study.
Availability of data
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
