Abstract
Endometrial stromal sarcomas (ESS) account for 10-15% of uterine malignancies and are classified into four categories: endometrial stromal nodule (ESN), low-grade endometrial stromal sarcoma (LG-ESS), high-grade endometrial stromal sarcoma (HG-ESS) and undifferentiated uterine sarcoma (USS). Depending on stage, ESS is treated with surgery, hormone therapy, chemotherapy or targeted therapy. A rare complication of ESS with metastatic pulmonary disease is recurrent, bilateral pneumothoraces. The current literature has reported on patients with ESS who either presented with pneumothoraces at their initial diagnosis, and thus were untreated, or after treatment with surgical resection and hormone therapy. There have been no case reports of patients with ESS who presented with pneumothoraces while receiving chemotherapy. Furthermore, there have been no reported cases of patients with HG-ESS presenting with this rare complication. We would like to expand the literature by reporting on two patients with HG-ESS who presented with pneumothoraces while concurrently receiving chemotherapy.
Keywords
Introduction
Uterine sarcomas account for 3% to 7% of uterine cancers and can be divided into mesenchymal tumors or mixed epithelial and mesenchymal tumors. 1 Endometrial stromal sarcomas account for 10% to 15% of uterine malignancies. 2 The current World Health Organization recognizes four categories of endometrial stromal tumor: endometrial stromal nodule (ESN), low-grade endometrial stromal sarcoma (LG-ESS), high-grade endometrial stromal sarcoma (HG-ESS) and undifferentiated uterine sarcoma (UUS). 3 Microscopically, LG-ESS typically involves the endometrium and infiltrates the myometrium as irregular projections often with lymphovascular invasion. The tumor cells are small, oval to fusiform, imparting a monotonous appearance sometimes, with low cytologic atypia and low mitotic activity (usually <5/10hpf). In the past, mitotic counts have been used to stratify endometrial stromal tumors. However, this is no longer convention as ESSs with high mitotic counts, but otherwise typical low-grade morphologic features, do not appear to have a significantly different prognosis from ESSs with low mitotic counts. HG-ESS are composed of atypical cells bearing histologic resemblance to endometrial stromal cells, but lacking the degree of pleomorphism necessary for a diagnosis of UUS. Most of the HG-ESS have a round cell portion that tends to have larger nuclei compared with LG-ESS, often with more angulated and irregular nuclear contours, supported by a delicate capillary network. The mitotic activity is consistently higher (>10 mitoses/10 HPFs) than the traditional LG-ESS, and tumor cell necrosis and vascular space invasion is more commonly seen. 3
The mainstay of treatment for most patients diagnosed with ESS is total hysterectomy and bilateral salphingo-oophorectomy. 4 Given that low-grade endometrial stromal sarcomas are often hormone sensitive tumors with overexpression of ER and PR reported in approximately 70% and 95% of cases, respectively, adjuvant targeted hormone therapy has shown to increase median overall survival when compared to an observational cohort not receiving hormone therapy. 5 Currently, hormone therapy is considered for any recurrent or advanced stage disease (stage 3 or 4) in patients with ER/PR positive tumors. 5 Agents used include megestrol or medroxyprogesterone, gonadotropin-releasing hormone analogs and aromatase inhibitors. For patients who continue to progress or develop recurrence, chemotherapy can then be considered.4,5 Systemic chemotherapy agents include doxorubicin and ifosfamide although gemcitabine, docetaxel, liposomal doxorubicin and paclitaxel have also been used. 4
The prognosis of Stage 1 LG-ESS is favorable with a 5 and 10 year prognosis of 98% and 89%. 6 Although ESS tends to follow an indolent nature, about one-third of patients have recurrent disease that requires long-term follow up. 1 Most commonly these patients develop recurrences in the pelvis and abdomen and less often in the lungs and vagina.6,7 In some cases, patients with metastatic disease in the lungs have been reported to present with recurrent pneumothoraces. The literature has reported on patients with ESS who either presented with pneumothoraces at their initial diagnosis, and thus were untreated, or after treatment with surgical resection and hormone therapy. There have been no case reports of patients with ESS who presented with pneumothoraces while receiving chemotherapy. We would like to expand the literature by reporting on two patients with endometrial sarcoma who presented with pneumothoraces while concurrently receiving chemotherapy.
Case #1
The patient is a 59-year-old female with no significant history of chronic lung disease or tobacco use who was diagnosed with ER+/PR+ metastatic, high-grade endometrial stromal sarcoma (HG-ESS) in April 42013. A surveillance CT chest on 1/2018 showed an incidental large left-sided pneumothorax while receiving chemotherapy. Her previous treatment for ESS included a hysterectomy at diagnosis (4/2013) followed by adjuvant systemic therapy with Gemcitabine and Taxotere. She developed relapse of her disease in the right upper lung in January 2016 with subsequent wedge resection. She was then started on anastrozole on 2/2016 but was switched to megestrol and later tamoxifen on 7/2016 due to significant bone pain. Unfortunately, she progressed and was started on cabozantinib, a phase 2 clinical trial, which was dose reduced due to nausea and vomiting (9/2017). She was then started on doxorubicin and olaratumab on 11/2017, however her third cycle of chemotherapy was complicated by a large left-sided pneumothorax on 1/2018, requiring ICU admission. CT Chest at the time of admission showed a new large left pneumothorax with multiple bilateral solid and cavitary pulmonary nodules (Figure 1(a)) which required chest tube placement. She then resumed cycle 4 of doxorubicin and olaratumab combination therapy and was switched to maintenance olaratumab for cycle 7 on 4/2018. Restaging CT scans continued to show response to therapy until cycle 18, when scans showed progression of her disease. At this time, the patient reported feeling dyspnea on exertion which progressed to dyspnea at rest and a CT Chest (1/2019) showed upper and lower lobe consolidative, ground glass opacities as well as multiple right-sided pulmonary nodules with cavitation and a large left-sided pneumothorax. She required chest tube placement and supplemental oxygen. She ultimately required a left diagnostic thoracoscopy with talc pleurodesis and was discharged to a skilled nursing facility for physical rehabilitation.

(a) 59 year-old woman with metastatic endometrial stromal sarcoma treated with chemotherapy. CT chest with contrast (upper row) demonstrates scattered peripheral cystic nodules compatible with treated metastatic disease. CT chest without contrast 2 months later (bottom row) demonstrates interval development of a spontaneous large left pneumothorax. Note the subpleural location of several of the left lung nodules, which may result in a pneumothorax secondary to pleural rupture, (b) 56 year-old woman with metastatic endometrial stromal sarcoma. CT chest with contrast (upper row) demonstrates multiple bilateral pulmonary parenchymal nodules compatible with metastatic disease. CT chest with contrast following 2 months of chemotherapy (bottom row) demonstrates interval development of spontaneous large right and moderate left pneumothoraces. Many of the parenchymal nodules have undergone cystic transformation following chemotherapy.
Case #2
The patient is a 56-year-old Hispanic non-smoking female with no past medical history of chronic lung disease who was diagnosed with ER-/PR- metastatic, high-grade endometrial stromal sarcoma (HG-ESS) in 2019 A CT-guided biopsy of a lung nodule showed tumor morphology consistent with metastatic disease. She underwent a total abdominal hysterectomy with bilateral salpingo-oophorectomy on 2/2019 to control her bleeding. She then received doxorubicin, ifosfamide, and mesna (AIM) (AIM) chemotherapy on 4/2019, but after cycle 2, AIM was discontinued due to encephalopathy. She resumed cycle 3 with doxorubicin alone on 6/2019, but was hospitalized for ten days after CT revealed bilateral pneumothoraces. . A CT angiography during admission redemonstrated innumerable solid pulmonary nodules, now with central cavitation (Figure 1(b)). After placement of bilateral chest tubes resulting in re-expansion, the patient was discharged, but readmitted for recurrence of bilateral pneumothoraxes that necessitated talc pleurodesis bilaterally. She remained pneumothorax free for the duration of her doxorubicin therapy, but eventually her disease progressed. She continues on second line chemotherapy with stable disease at this time.
Discussion:Pulmonary metastases are a rare complication of ESS with a reported incidence ranging from 7% to 28%. 7 In patients with pulmonary metastases, patients have been found to develop unilateral and bilateral pneumothoraces. A literature review identified 10 other reported cases of women who were found to have pneumothoraces due to either previously diagnosed or unknown metastatic ESS (Table 1).7,8 –16 In 8 of the 10 patients, a diagnosis of low-grade ESS was made while the other two had an unknown grade. The majority of the patients presented with bilateral pneumothoraces (7/10 cases) and some patients experienced multiple episodes of pneumothoraces over a series of months. In four of the case reports, the pneumothorax was the first presenting sign of the malignancy while in five of the cases, the patient had been previously treated with surgery. There was one patient who had received six cycles of doxorubicin, 3 years prior to the pneumothorax, followed by hormonal therapy and 4 months of pazopanib at the time of the pneumothorax. 15 There was no report of any patient who experienced a pneumothorax while receiving chemotherapy. Furthermore, while LG-ESS with pneumothoraces have previously been described in the literature, our case series is the first to describe high-grade endometrial sarcoma presenting with pulmonary metastases leading to pneumothoraces after the start of chemotherapy.
A literature review of documented pneumothorax cases caused by metastatic endometrial stromal sarcoma.
Regarding the mechanism by which ESS lung metastases lead to pneumothoraces during chemotherapy, the pathophysiology is likely multifactorial and can be attributed to the ESS histopathology, the location of the lung metastases, and the chemotherapy mechanism of action.
In our case report, the tumor cells consisted predominantly of atypical round cells with irregular nuclear outlines and a delicate capillary network. The mitotic count was high (18/10 hpf) and with areas of necrosis present (Figure 2(a) and (b)). Immunostain for CD10 was positive in both cases (Figure 2(c)) and ER immunostain was positive in one case (Figure 2(d)). As a result of the variable cell necrosis seen on histopathology and rate of tumor growth, ESS is reported to have variable imaging findings from well-defined or infiltrative masses to unilocular or multilocular cystic appearances. This can result in simultaneous cystic and solitary nodular lesions as was seen in Figure 1.
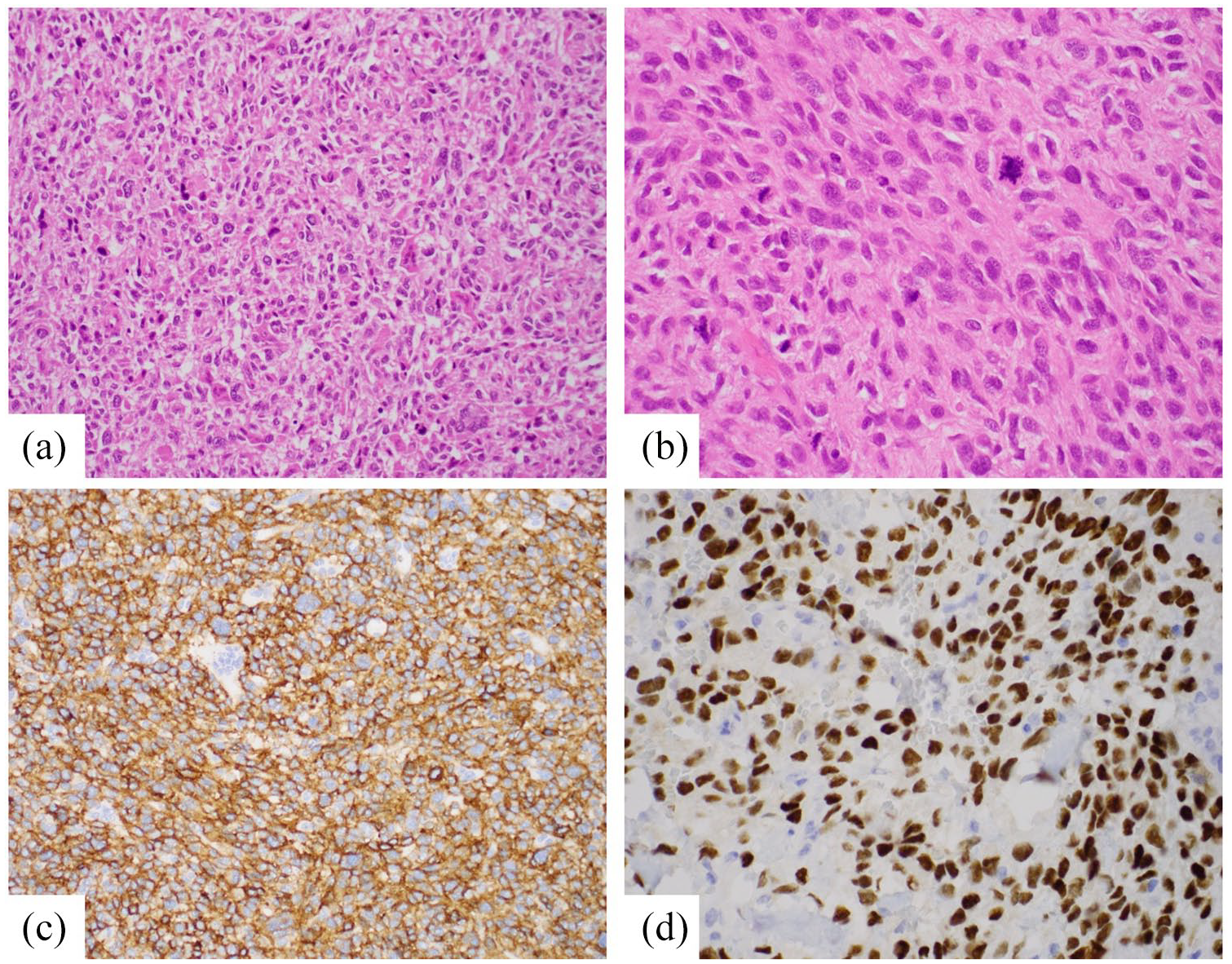
(a) Atypical round cells with more angulated and irregular nuclear outlines, HE × 200×, (b) cells with pleomorphism and prominent mitotic figures, HE × 400×, (c) tumor cells showing CD10 positivity, CD10 × 200×, and (d) tumor cells showing ER positivity, ER × 200×.
As seen in Figure 1(b), as these parenchymal nodules evolve, they can undergo cystic transformation and depending on their location within the lung, can result in pneumothoraces. This cystic progression is often seen as a result of extensive tumor necrosis that can either be caused by aggressive tumor growth or exacerbated by chemotherapeutics and has been reported in previous series of chemotherapy associated pneumothoraces in solid tumor patients that included sarcoma patients, but none with ESS. 17 Similarly, other studies and case reports of systemic therapy induced pneumothoraces include eribulin in some types of sarcomas as well as pazopanib, an anti-VEGF targeted tyrosine kinase inhibitor, which was reported and associated with a case of high grade ESS.15,18 A subsequent study looking at the use of pazopanib in sarcoma patients concluded that it’s use did not confer a higher risk of pneumothorax than those not taking pazopanib. 19 Interestingly, the only risk factor associated with a higher risk of pneumothorax on multivariate analysis was the development of cavitary lesions within lung nodules.
In this case report, the pneumothoraces presented while the patients were being treated with doxorubicin. By inducing necrosis, doxorubicin can lead to cystic or cavitary changes. If these changes occur at susceptible regions such as the subpleural spaces or adjacent bronchi, a pneumothorax could ensue leading to emergent symptoms such as in our two cases. There are currently multiple other theories that attempt to explain the mechanisms by which these pneumothoraces occur. Another theory suggests that the tumor nodules at the lung periphery obstruct the bronchioles and lead to a “ball valve-type effect” that can lead to distension and rupture of the lung. 20 We will likely need more studies to better elucidate this mechanism and it is likely that each pneumothorax is caused by various factors without a single unifying etiology.
Conclusion
In conclusion, HG-ESS can be associated with pneumothoraces in patients receiving chemotherapy. This is the first report in such patients and given that systemic agents have the potential to induce tumor necrosis in ESS patients, clinicians should be made aware of this rare complication and should consider pneumothorax in their differential diagnosis in patients who develop shortness of breath during chemotherapy.
Footnotes
Contributorship
Angela Duvalyan and Kirk Tran researched the current literature and drafted the manuscript. Dr. Shefali Chopra and Dr. Christopher Lee provided their expertise for the pathology and radiology findings, respectively. Dr. James Hu was involved in conceiving, editing and organizing the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval (include full name of committee approving the research and if available mention reference number of that approval)
No ethical approval was required for this case report.
Informed Consent
Informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
