Abstract
The most common associated malignancies with BRCA mutations include breast and ovarian cancers. Less common malignancies associated with BRCA mutation include: pancreatic, prostate, colon, gastric, and biliary cancers. Esophageal cancer, particularly squamous cell carcinoma, has rarely been reported to harbor BRCA mutations. Here we report, to our knowledge, the first case of germline BRCA1 mutated associated esophageal squamous cell carcinoma.
Introduction
BRCA1 and BRCA2 are known tumor suppressor genes involved in DNA repair via homologous recombination. 1 It is well-established that deleterious mutations in these genes are associated with an increased risk of malignancy, namely breast and ovarian cancer. BRCA1 and BRCA2 mutations also increase the risk of gynecologic malignancies (fallopian tube and endometrial) along with prostate and pancreatic cancer. 2 In terms of increased risk of gastrointestinal malignancies, BRCA2 mutations have been described in stomach and biliary cancers. 3 A family-based association study noted that there was a relative risk increase of esophageal (relative risk [RR] 2.9, 95% CI 1.1–6) and stomach (RR 2.4, CI 1.2–4.3) cancer in families with BRCA1 mutations. 4 Additionally a review of 1072 malignancies with deleterious BRCA mutations revealed one case of BRCA1 mutated esophageal cancer (histology unspecified). 5
Here we report a patient with confirmed germline BRCA1 mutated esophageal squamous cell carcinoma (ESCC).
Case report
Our patient is a 69-year-old female of Ashkenazi Jewish ancestry with a past medical history significant for stage IIIA left-sided breast cancer diagnosed in 1994 (at the age of 46). She received bilateral modified radical mastectomies, adjuvant chemotherapy and radiation (chest wall and axilla, dose details unavailable) followed by autologous stem cell transplantation.
Genetic testing was performed and patient was found to harbor a deleterious BRCA1 mutation. She subsequently underwent prophylactic bilateral oophorectomy and hysterectomy. Of note, her sister was also positive for the BRCA1 mutation.
The patient then presented to the emergency department in 2017 with hematemesis. She reported progressive odynophagia in the preceding 6 months along with a 30-pound weight loss. Esophagogastroduodenoscopy (EGD) showed a partially obstructive, malignant appearing esophageal mass with esophageal stenosis at 20 cm from the incisors. The tumor was 5.5 cm in diameter and 10 cm in length (Figure 1). The mass was biopsied and pathology was significant for invasive, poorly differentiated, squamous cell carcinoma.
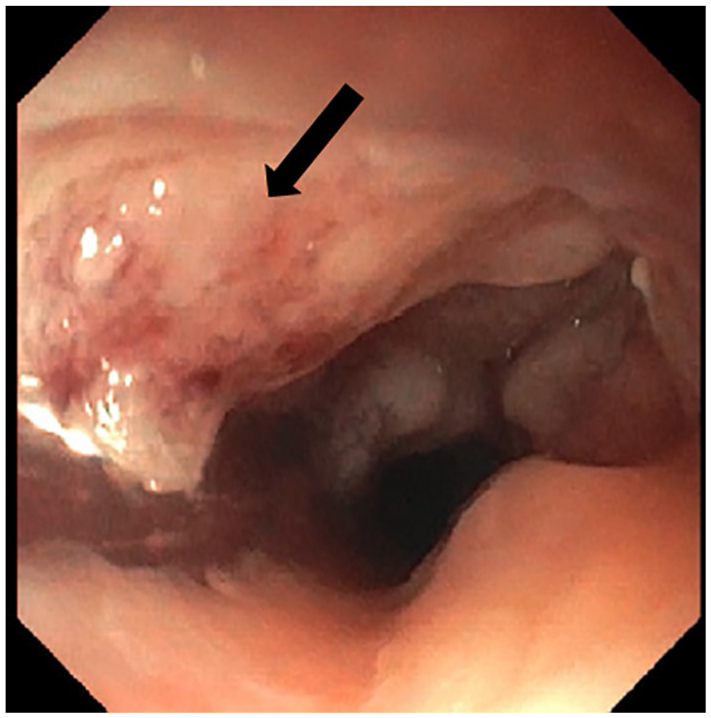
EGD. Endoscopic visualization of the esophageal mass (arrow) in the middle one-third of the esophagus.
A staging positron emission tomography with computed tomography (PET/CT) scan revealed irregular thickening of the middle-third of the esophagus, consistent with esophageal mass seen on endoscopy (Figure 2). Also noted was extrinsic compression of the left mainstem bronchus and a slightly prominent precarinal lymph node measuring 14 × 9 mm in size. No distant metastasis was noted. Clinical stage was T4bN0M0 (corresponding to Stage IIIC by AJCC 7th edition). She was deemed to not be a surgical candidate.
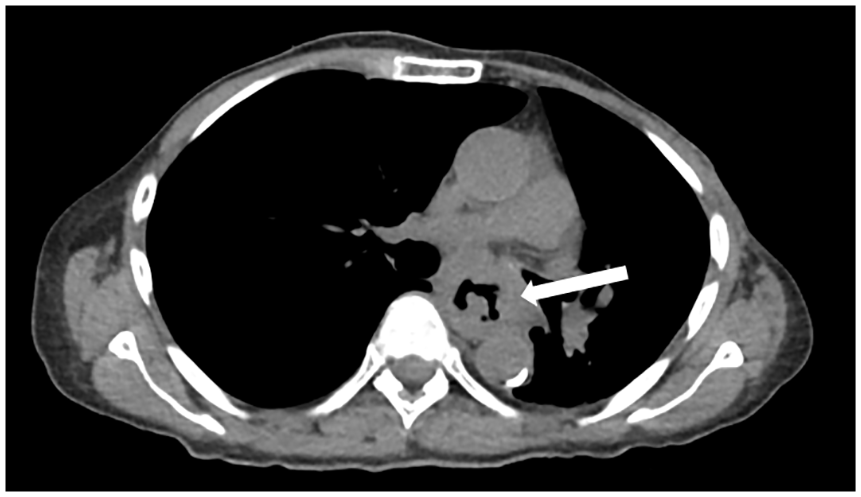
CT chest (axial view). Large esophageal mass (arrow) with infiltration of the mediastinum.
A bronchoscopy was done as a part of the work up and a bronchoesophageal fistula was noted in the left mainstem bronchus. A covered bronchial stent was subsequently placed. Combined modality therapy with chemotherapy and radiation was recommended for the patient. At the time of her diagnosis, the tumor was sent off for next generation sequencing (Foundation One®). The following genomic alterations were noted: BRCA1 (E23fs*17), CDKN2A (loss of function) and TP53 (loss of function).
The patient began weekly carboplatin/paclitaxel along with concurrent radiation therapy (55.8 Gy via image modulated radiation therapy). Response to therapy as assessed by PET/CT showed a marked response to therapy with no definite residual FDG uptake within the esophagus (Figure 3). Unfortunately despite response to treatment the patient died of complications of pneumonia related to the bronchoesophageal fistula.

PET/CT (coronal view): (a) intense fluorodeoxyglucose uptake in the region of the esophageal mass (arrow) and (b) complete resolution of fluorodeoxyglucose uptake in esophagus.
Discussion
This case report highlights a rare BRCA1 mutated associated malignancy. As mentioned above, a suggestion of an increased risk for esophageal (RR 2.9) and gastric cancer (RR 2.4) was noted in family members of patients harboring BRCA1 mutations. It is equally important to point out that these associations were mostly based on unconfirmed cases of BRCA mutation in those individuals. 4
As relates to BRCA2, researchers in China observed frequent loss of heterozygosity involving chromosome 13, specifically 13q12 (where BRCA2 resides), in patients with ESCC.6–8 This prompted an evaluation of the BRCA2 gene in 56 patients with esophageal squamous cell carcinoma and found that 5 (9%) patients had BRCA2 mutations, and 3 of these were determined to be germline mutations. 9 Interestingly, none of these cases exhibited the Knudsen two-hit inactivation of BRCA2, suggesting that BRCA2 may have been a passenger mutation as opposed to the mechanism of carcinogenesis.
Another analysis of BRCA2 mutations was performed among the Turkmen population who reside in the northeastern part of Iran. This population of patients has some of the highest rates of squamous esophageal cancer in the world. 10 Given the previous data from the Chinese studies of the possibility of BRCA2 playing a role in the carcinogenesis of ESCC, researchers set out to evaluate BRCA2 status in 197 Turkmen with ESCC and identified deleterious germline mutations of BRCA2 in 15 (7.6%) patients. 11
Armed with the knowledge that our patient had a BRCA1 mutation we were hopeful that incorporation of a platinum along with radiation would improve treatment response and disease control based on data showing increased platinum sensitivity in BRCA mutated breast,12,13 ovarian, 14 and pancreatic cancer 15 patients. Indeed our patient had a complete metabolic response (Figure 3) to combined modality chemotherapy and radiation. The increased sensitivity of BRCA mutated tumors to platinum chemotherapy is due to the impaired ability of the tumor to repair double stranded DNA adducts induced by platinum. 16 Similar to alkylating agents, ionizing radiation potentiates DNA damage by inducing double stranded breaks.
PARP (poly[ADP-ribose] polymerase) inhibitors have also been shown to have promising activity in BRCA mutated tumors (e.g. breast, pancreatic, and prostate cancers).17,18 PARP is a family of proteins that bind to damaged DNA and helps recruit proteins for DNA repair, including base excision repair, homologous recombination, and alternative non-homologous end joining. Because BRCA mutated cells have deficient DNA repair they become overly reliant, or “addicted,” to other repair pathways and proteins, such as PARP. Inhibiting PARP in patients with BRCA mutated tumors causes the replication fork to stall causing a double strand break and the cell then undergoes apoptosis. This concept is also referred to as synthetic lethality. 19 The PARP inhibitors, niraparib, olaparib, and rucaparib have been FDA approved for the treatment of ovarian cancer.
A logical question is whether or not the previous radiation the patient received years prior played a role in carcinogenesis. Interestingly the data in breast cancer suggests no significant increased risk of either contralateral breast cancer after adjuvant radiation and no clinically significant increase in the risk of sarcoma.20,21 Zablotska et al. evaluated the risk of squamous cell esophageal cancer following chest wall radiation in patients with breast cancer. After reviewing SEER data from 1973 to 2000 the authors identified a 2.83 (95% CI, 1.35–5.92) and 2.17 (95% CI, 1.67–4.02) increased relative risk for the development of squamous cell esophageal cancer at 5 to 9 and ⩾10 years, respectively. 22 This risk appeared confined to the upper and middle third of the esophagus.
An admitted limitation to our case report is whether or not the BRCA mutation was a driver mutation as opposed to a passenger mutation in the setting of the other genomic alterations. Jonsson et al. suggested that for tumors not traditionally associated with BRCA mutations, the BRCA mutation is likely a result of selective pressure from other genomic alterations, and thus a passenger event. To further prove this point they highlighted clinical data suggesting that tumors not traditionally associated with BRCA mutations did not respond as favorably to PARP inhibition as compared to BRCA mutated associated malignancies. 23 It should be noted that approximately 97% of the BRCA mutations in this study were somatic mutations.
Footnotes
Author contributions
There was equal contributorship among both authors in the construct of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Mayo Clinic and University of Florida does not require ethical approval for reporting individual cases.
Informed consent
Unfortunately the patient has since passed away. The patient verbally agreed to publication of her case while she was alive. The patient does not have any next of kin for us to contact to have a form signed. The manuscript is sure to anonymize the patient so that her identity will be protected. This manuscript will be a nice way to honor the patient’s life and she would like to have known that others could learn from her disease.
