Abstract
Retroperitoneal chordomas are exceedingly rare and account for less than 5% of all primary bone malignancies. Their etiology remains unknown. We report a rare case of an extravertebral chordoma of the retroperitoneum in a 71-year-old man treated with surgical resection and post-operative spot-scanning proton beam therapy. We describe how to safely treat a retroperitoneal target to a prescription dose over 70 Gy (relative biological effectiveness) with spot-scanning proton beam therapy and also report a dosimetric comparison of spot-scanning proton beam therapy versus intensity-modulated radiation therapy. This case not only highlights a rare diagnosis of an extravertebral retroperitoneal chordoma but it also draws attention to the dosimetric advantages of proton beam therapy and illustrates a promising radiotherapeutic option for retroperitoneal targets.
Case presentation
A 71-year-old male presented with a deep venous thrombosis and pulmonary embolus. On further workup, contrast-enhanced computed tomography (CT) of the abdomen and pelvis demonstrated a 73 mm × 70 mm × 41 mm hypoattenuating retroperitoneal mass in the right paracaval region medial to the right kidney with 180° abutment of the inferior vena cava (Figure 1).

Contrast-enhanced CT of the abdomen and pelvis demonstrated a 73 mm × 70 mm × 41 mm hypoattenuating retroperitoneal mass in the right paracaval region medial to the right kidney encasing the inferior vena cava.
Gadolinium-enhanced magnetic resonance imaging (MRI) demonstrated a multiloculated T2-hyperintense extravertebral chordoma at the level of L2 in the right retroperitoneum that extended medially along the medial aspect of the right psoas muscle (Figure 2). Of note, there was no evidence of extension into the neural foramina and spinal canal or the presence of an epidural component. A CT-guided biopsy demonstrated a primary retroperitoneal chordoma. Immunohistochemistry was positive for pancytokeratin (A1/A3) and focally positive for CK7 and S-100 and negative for PAX8, MART-1, HMB45, CK20, CDX-2, and CD-10, supporting the diagnosis.

Gadolinium-enhanced MRI demonstrated a multiloculated T2 hyperintense extravertebral chordoma at the level of L2 in the right retroperitoneum that extends medially along the medial aspect of the right psoas muscle.
He underwent an en-bloc gross total resection. An 85-mm retroperitoneal chordoma was found to be involving the soft tissues adjacent to but not invading into the inferior vena cava, right renal vein, right renal artery, and right psoas muscle with tumor extending to the inked edge of the specimen. The mass was free of any nerve roots. There was no evidence of gross tumor remaining following the resection.
Final pathology demonstrated a microscopic-positive margin. He was referred for post-operative radiation therapy. We recommended proton beam therapy (PBT) to the surgical bed in the setting of a microscopic positive margin. We prescribed a total dose of 72 Gy (relative biological effectiveness (RBE))/(Gy) to be delivered with 2 Gy (RBE)/Gy daily fractions over 36 consecutive treatment days excluding weekends to the surgical bed. Using an Orfit board (Orfit Industries, Wijnegem, Belgium), custom Vac-LokTM, and a thermoplastic mask (Orfit Industries), the patient was immobilized on the treatment table in the supine position with his arms extended over his head. A treatment planning four-dimensional computed tomography (4DCT) scan demonstrated that respiratory motion of the treatment target was less than 2 mm isotropically. A clinical target volume (CTV) included potential sites of microscopic disease. A scanning target volume (STV) was defined as the CTV with a 5-mm isotropic expansion to guide PBT spot placement. Lexicographic-based optimization was performed based on CTV coverage constraints. Theoretically, PBT plans require utilization of an additional optimization target volume (OTV) to account for daily setup uncertainties. However, the proximity of critical structures can limit the amount of margin one can expand on the CTV. Under such space and dose-limiting situations, an alternate method composes of foregoing generation of an OTV volume and creating an STV margin from the CTV for the placement of Bragg peaks around the target. If the lateral margins of the STV account for lateral penumbra of proton spots placed outside the CTV, and the distal margins account for CT based range uncertainties, one can achieve nearly homogeneous dose coverage with adequate robustness in such plans as one would expect otherwise with plans involving an OTV. Treatment in this case was prepared based on the STV margin and CTV-based optimization.
Normal tissues including the spinal cord, kidneys, small bowel, and liver were contoured. A 5-mm isotropic expansion on the spinal cord was used to create a planning organ-at-risk volume for optimization purposes. Given that the CTV was in close proximity to healthy tissue including the duodenum, small bowel, right kidney, spinal cord, and liver, we created comparison plans of spot-scanning PBT versus intensity-modulated radiation therapy (IMRT) (Figure 3). To adequately treat the target volume to 72 Gy and maximally spare the kidneys and bowel, we elected to treat this patient with spot-scanning PBT. The patient was treated with two equally weighted posterior oblique fields. Daily kV image guidance and a single verification CT scan halfway through treatment was performed.

Axial view of the proton (above) and IMRT (below) dose distribution in color wash. CTV (red). Organs-at-risk: duodenum (cyan), large bowel (brown), ipsilateral kidney (magenta), spinal cord (yellow), contralateral kidney (dark blue), and small bowel (light green).
Both treatment modalities allowed greater than 99% of the CTV with 100% of the prescription dose. However, PBT maximally spared the organs-at-risk, especially the kidneys and bowel. The duodenum would have received a maximum dose of 55 Gy with IMRT versus 0 Gy with PBT. The ipsilateral and contralateral kidney would have received a mean dose of 8 and 4 Gy with IMRT, respectively. With IMRT, approximately 12% of the total kidney volume would have received 12 Gy and approximately 10% of the ipsilateral kidney would have received 18 Gy. Both kidneys received essentially no radiation with PBT (Table 1 and Figure 3).
Proton versus IMRT dose to organs-at-risk.
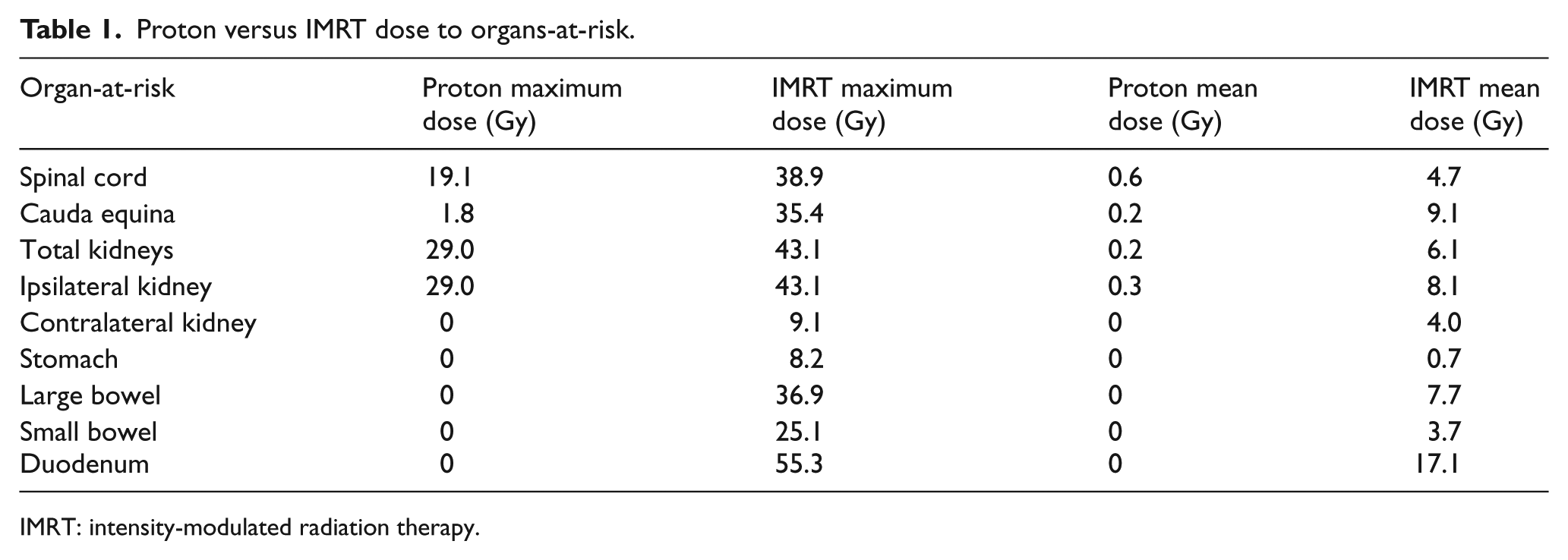
IMRT: intensity-modulated radiation therapy.
The patient completed PBT without any treatment-related toxicity. He has no treatment-related toxicity or evidence of recurrent disease 9 months following treatment.
Discussion
Chordomas are rare and locally aggressive malignancies of the bone arising from the notochord remnants. 1 They account for 1%–4% of all primary bone malignancies and most often occur in three axial skeleton locations: the sacrococcygeal region (50%), skull base (35%), and vertebral column (15%).2,3 Although reported in the literature, retroperitoneal chordomas are exceedingly rare.4,5 Here, we report a case of a retroperitoneal extravertebral chordoma, illustrate how to safely treat a retroperitoneal target to a prescription dose above 70 Gy with spot-scanning PBT, and highlight the dosimetric advantages of PBT over IMRT in retroperitoneal targets.
En bloc resection is the gold standard for chordomas. 6 There is a direct relationship between the extent of surgical resection and the length of progression-free survival. 7 However, chordomas are often difficult to completely resect. In these cases, adjuvant radiotherapy may improve outcomes. 8 A phase II trial of both proton and photon therapy after resection from the study by DeLaney et al. 9 favored a prescription dose of 77 Gy (RBE) definitively and 70 Gy (RBE) adjuvantly. Despite this, local recurrence remains the predominant pattern of treatment failure.10–14
Chordomas are often considered radioresistant. However, retrospective series of patients with chordomas who have been treated with radiation therapy have demonstrated dose–response in terms of local control between 60 and 70 Gy (RBE). Local control has been reported to be less than 25% when treated with less than 60 Gy (RBE), 50% when treated with 60 to 70 Gy (RBE), and approximately 75%–80% when treated with doses higher than 70 Gy (RBE). 15 Although a recent series of skull base chordomas treated with combined photon and proton radiation therapy to either 68.4, 70.2, 72, or 73.8 Gy (RBE) did not suggest a clear difference in local control. 16 However, the lack of a dose–response above 70 Gy is not yet well established.
Conventional radiation therapy is limited in its capability to safely deliver adequate dose to a target without delivering excessive dose to the surrounding healthy tissue. Charged particle therapy in the form of PBT is a promising treatment modality that has physical advantages over photon-based radiation therapy including stopping power and an ability to deliver a higher radiobiologic effect dose. 17 PBT is often delivered with a minimal number of beams, whereas IMRT requires many beams. Thus, PBT has a theoretical advantage of delivering escalated dose to a given target with maximal sparing of the surrounding normal structures.
Retroperitoneal targets are in close proximity to sensitive organs such as the kidneys and bowel. Radiation-induced renal damage can present acutely within 3 months of radiation or gradually over years. Cheng et al. 18 conducted a review of 12 publications on radiation-induced renal toxicity after total body irradiation and found that a total kidney dose of 9.8 Gy is associated with a 5% risk of renal toxicity, regardless of fractionation or concurrent chemotherapy. Also, radiation to a unilateral kidney is not free of risk and can lead to atrophy- and volume-dependent renal toxicity. The irradiated kidney has been reported to have a 10% or 24% diminished creatinine clearance after receiving ⩾26 Gy to more than half or more than 90% of a single kidney, respectively.19,20 After radiation-induced damage to a single kidney, the contralateral kidney can often compensate. However, low-dose radiation to >30% of the compensatory kidney can potentially blunt the compensatory response. 21 In our case, PBT reduced the dose to the ipsilateral kidney to a mean of 0.3 Gy with complete sparing of the contralateral kidney. In addition, Emami et al. 22 estimated a 5% risk of toxicity at 5 years when 1/3 of small bowel received 50 Gy and a 50% risk at 5 years when small portions receive 60 Gy. Verma et al. 23 reported increased duodenal toxicity with larger volumes receiving 55 Gy. In our case, PBT demonstrated dosimetric advantages when directly compared to IMRT. Although both modalities did meet target coverage as well as conventional spinal cord dose constraints, PBT maximally spared the organs-at-risk, in particular the duodenum and kidneys. PBT reduced the dose to the duodenum to 0 Gy from a maximum dose of 55 Gy with IMRT, and both kidneys received essentially no radiation with PBT. PBT allowed complete sparing of the contralateral kidney, stomach, and entire bowel. Further studies are needed to determine the long-term clinical significance of these dosimetric benefits in terms of gastrointestinal and renal toxicity.
PBT is particularly advantageous for retroperitoneal malignances. In the context of dose escalation for radioresistant tumors such as chordomas or sarcomas, the absolute benefit may be magnified.
Conclusion
Surgical excision plus adjuvant radiotherapy is considered the gold standard for the treatment of chordomas. Retroperitoneal chordomas pose unique challenges due to normal tissue tolerances of the bowel and kidneys. Optimal local control is seen with doses over 70 Gy. For these reasons, PBT provides the greatest improvement in the therapeutic ratio. Future prospective data are needed to assess long-term control and late toxicity.
Footnotes
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Author contributions
All authors were involved with conception and design, writing, and final approval of this teaching case.
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
