Abstract
Primary bone malignancies - including chordoma, chondrosarcoma, osteosarcoma, and Ewing sarcoma - originate from bone or cartilage cells and often develop in anatomically complex or surgically challenging regions. While surgical resection remains the standard of care for most localized tumors, radiation therapy (RT) has become an increasingly integral component of multidisciplinary management, particularly when complete surgical excision is not feasible, margins are close or positive, or the tumor is adjacent to critical structures. Historically, conventional photon-based RT has shown limited efficacy in many of these tumors due to factors such as relative radioresistance and proximity to radiosensitive normal tissues. However, advances in conformal photon techniques such as intensity-modulated radiation therapy (IMRT) and stereotactic body radiation therapy (SBRT), along with hadron-based approaches like proton beam therapy (PBT) and carbon ion radiation therapy (CIRT), have expanded the therapeutic potential of RT in bone sarcomas. This review highlights the evolving role of RT in the management of primary bone malignancies, with a focus on technological advances, clinical outcomes, ongoing trials, and future directions in the field.
Keywords
Introduction
Primary bone malignancies are rare but aggressive cancers that originate from mesenchymal cells within the bone, accounting for approximately 0.2% of all cancers and 5% of pediatric cancers. 1 The most common subtypes include osteosarcoma, chondrosarcoma, and Ewing sarcoma, while less common forms include chordoma, undifferentiated pleomorphic sarcoma, adamantinoma, fibrosarcoma, and giant cell tumor of bone. 1 Surgical resection, often combined with chemotherapy, remains the cornerstone of treatment for most of these tumors.
Radiation therapy (RT) also plays a pivotal role, particularly when surgery is infeasible due to anatomical complexity, close or positive margins, or proximity to critical structures. Historically, the effectiveness of RT has been constrained by the relative radioresistance of certain bone tumors and the risk of toxicity to surrounding healthy tissue. However, recent advances in RT technology have significantly improved the precision, safety, and efficacy of treatment. These innovations have broadened the clinical utility of RT and strengthened its role within the multidisciplinary management of bone sarcomas. This review explores the evolving role of RT in the treatment of chordoma, chondrosarcoma, osteosarcoma, and Ewing sarcoma, with an emphasis on how modern techniques have contributed to improved patient outcomes.
Radiation Therapy – Classical Versus Next Generation Techniques
RT has long been a component of primary bone malignancy treatment, with its application tailored to each tumor's biology and anatomical context. It is commonly used as an adjunct when complete surgical resection is precluded or questionable. In these situations, RT contributes to improved local control (LC), reduced recurrence risk, and better quality of life.2–5 Ewing sarcoma is a notable exception due to its high radiosensitivity; here, RT often plays a more prominent role, used preoperatively to reduce tumor volume or postoperatively to manage residual disease.6,7
Recent advancements in RT technology have revolutionized the treatment landscape for bone sarcoma management. Modern techniques – including intensity-modulated radiation therapy (IMRT), stereotactic body radiation therapy (SBRT), proton beam therapy (PBT), and carbon-ion radiation therapy (CIRT) - enable the precise delivery of high radiation doses while sparing adjacent healthy tissue. This precision is particularly important in tumors located near critical structures such as nerves and organs.
IMRT utilizes multileaf collimators - movable tungsten leaves positioned at the beam's aperture - to shape the radiation beam with high precision. These collimators can be adjusted individually to block or allow specific portions of the beam, conforming the radiation dose to the tumor's three-dimensional geometry. 8 In addition to shaping the beam, IMRT modulates the intensity across different regions of the tumor, delivering higher doses to denser tumor areas while reducing exposure to adjacent healthy tissues.
While IMRT focuses on shaping the radiation dose and adjusting intensity across the treatment field, SBRT optimizes both the dose per session and the overall treatment timeline. Conventional RT is typically administered in numerous small fractions over several weeks to minimize damage to surrounding tissues and allow normal cells time to recover. However, this prolonged schedule also gives tumors time to repopulate, increases healthcare costs, and places a greater logistical burden on patients. SBRT addresses these limitations by combining advanced imaging modalities, such as computed tomography (CT), magnetic resonance imaging (MRI), and positron emission tomography (PET), with highly conformal dose delivery techniques. Leveraging the precision of IMRT, SBRT delivers larger doses per session over fewer treatments. 9 It also employs multiple radiation beams from various angles, concentrating high doses at the tumor while sparing nearby healthy tissues. This makes SBRT particularly effective for treating tumors in anatomically sensitive areas or for consolidating metastatic lesions, offering durable LC with minimal toxicity. 10
In addition to innovations in delivery techniques, recent advances have also transformed the physical nature of the radiation itself. PBT replaces traditional photon-based radiation with ionized hydrogen atoms (protons). Each accelerated proton carries the energy equivalent of approximately 108-109 photons, but this energy is deposited at a more focused and localized point. As a result, PBT enables highly precise tumor targeting while sparing adjacent normal tissues. 11
Building on this principle, CIRT utilizes carbon ions, which are approximately twelve times more massive than protons. 12 This increased mass allows for even greater biological effectiveness, but also necessitates specialized high-energy particle accelerators, currently available only at a select few centers worldwide. 13 Both PBT and CIRT - collectively referred to as hadron therapies – offer distinct advantages over conventional photon-based RT, particularly in delivering high doses with remarkable precision and reduced toxicity to surrounding critical structures.5,14 These modalities have shown promising results in improving LC, survival outcomes, and reducing side effects, even in tumors previously considered difficult to treat.
The following sections detail how these innovations have advanced treatment outcomes for chordoma, chondrosarcoma, osteosarcoma, and Ewing sarcoma.
Chordoma
Chordomas are rare primary bone tumors, occurring in about 1 per 1,000,000 people. They represent 1–4% of all bone cancers and about 17% of primary bone tumors in the spine, with a median age of diagnosis around 60 years.14,15 The reported median survival for chordoma patients is approximately 6 years, with 5-, 10-, and 20-year survival rates declining significantly from 67.6% to 39.9% and 13.1%, respectively, according to SEER data.
While chordomas tend to grow slowly, their location near critical structures such as the brainstem, nerve roots, and the spinal cord significantly complicates the prospect of achieving clear margins without neurological damage. As a result, adequate radiation doses are crucial for effective LC. Conventional x-ray RT provides only palliative relief, with doses of 50–60 Gy failing to significantly improve median survival and leading to recurrence rates between 80–100%. 14 Doses above 70 Gy are necessary for better outcomes, but achieving this is challenging due to the proximity of neural structures, which have much lower radiation tolerance. 5
Modern RT techniques, such as IMRT,16–18 SBRT,18–21 and hadron therapies,22,23 have improved dose conformity and reduced toxicity. Although no head-to-head trials have directly compared photon-based RT with hadron therapies in chordoma, a systematic review by Amichetti et al found that PBT achieved superior outcomes at 10 years compared to conventional photon irradiation, with relatively few significant complications, considering the high doses employed. 24 However, due to the high cost and limited availability of proton therapy – currently offered at only 47 centers in the United States – its use must be prioritized for cases where its advantages are most critical.
To address the limited availability and high demand for proton therapy, researchers have investigated hybrid strategies that combine IMRT with a reduced dose of PBT. In this approach, most of the radiation is delivered
Ongoing Clinical Trials Involving RT in Primary Bone Malignancies.
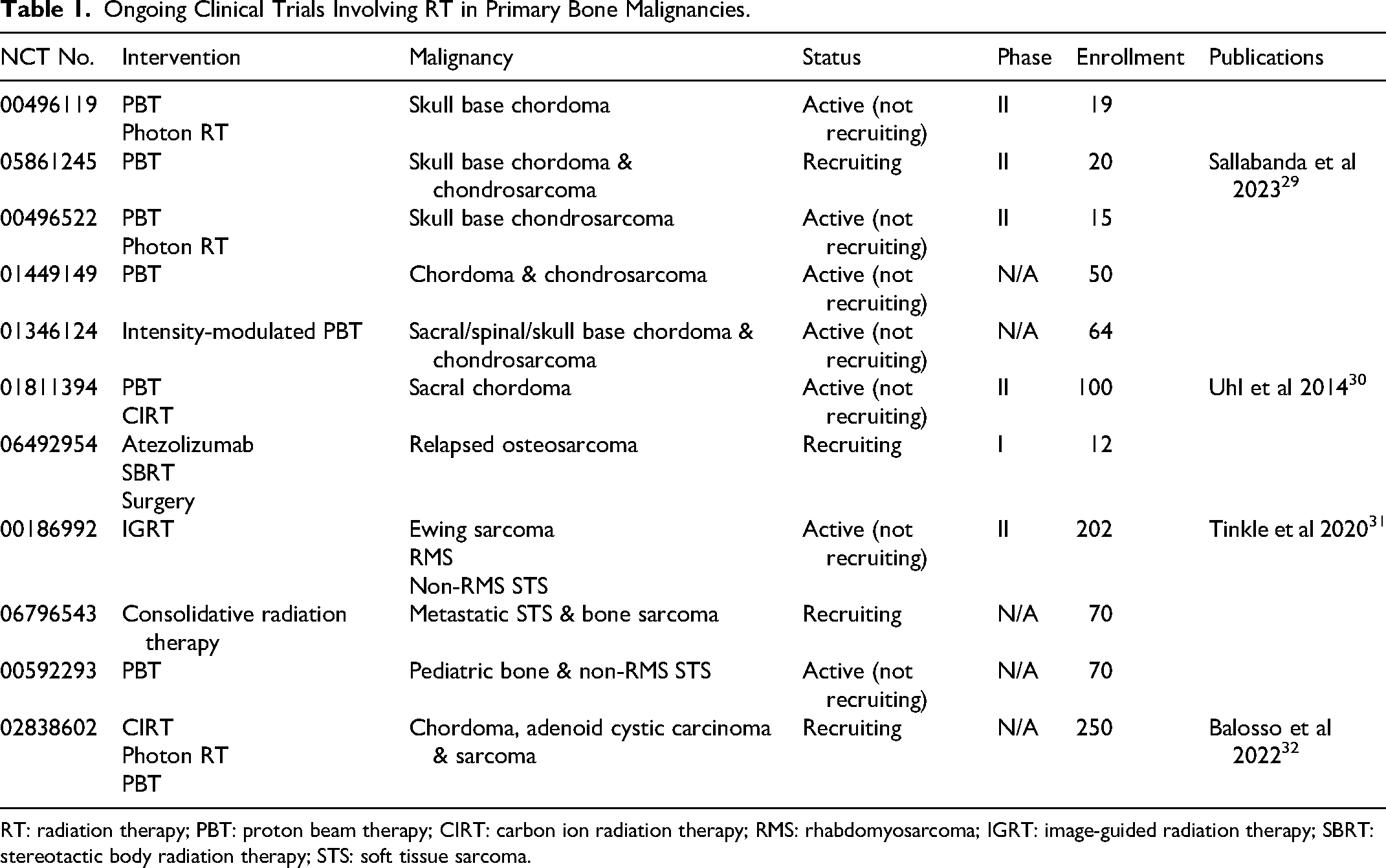
RT: radiation therapy; PBT: proton beam therapy; CIRT: carbon ion radiation therapy; RMS: rhabdomyosarcoma; IGRT: image-guided radiation therapy; SBRT: stereotactic body radiation therapy; STS: soft tissue sarcoma.
While the functional – if not logistical – superiority of PBT over conventional photon RT is well established, a new question has emerged: does CIRT offer additional clinical advantages over and above those of PBT? Several comparative studies have explored this, with the consensus being that PBT and CIRT provide similar efficacy.33–36 Some suggest a slight advantage for PBT, though this may be skewed by selection bias, as CIRT is often reserved for more advanced or inoperable cases due to its limited global availability. 37 A comprehensive meta-analysis by Rodrigues et al reviewed 14 independent studies involving 671 patients treated with PBT and 474 with CIRT for skull base chordoma, concluding that both modalities yield comparable long-term outcomes. 23 Tables 2 and 3 summarize studies of PBT and CIRT in chordoma patients, respectively, presenting the most frequently reported statistics of LC and overall survival (OS).
Published LC and OS Rates for Chordoma Treated with PBT.

LC: local control; OS: overall survival.
Published LC and OS Rates for Chordoma Treated with CIRT.
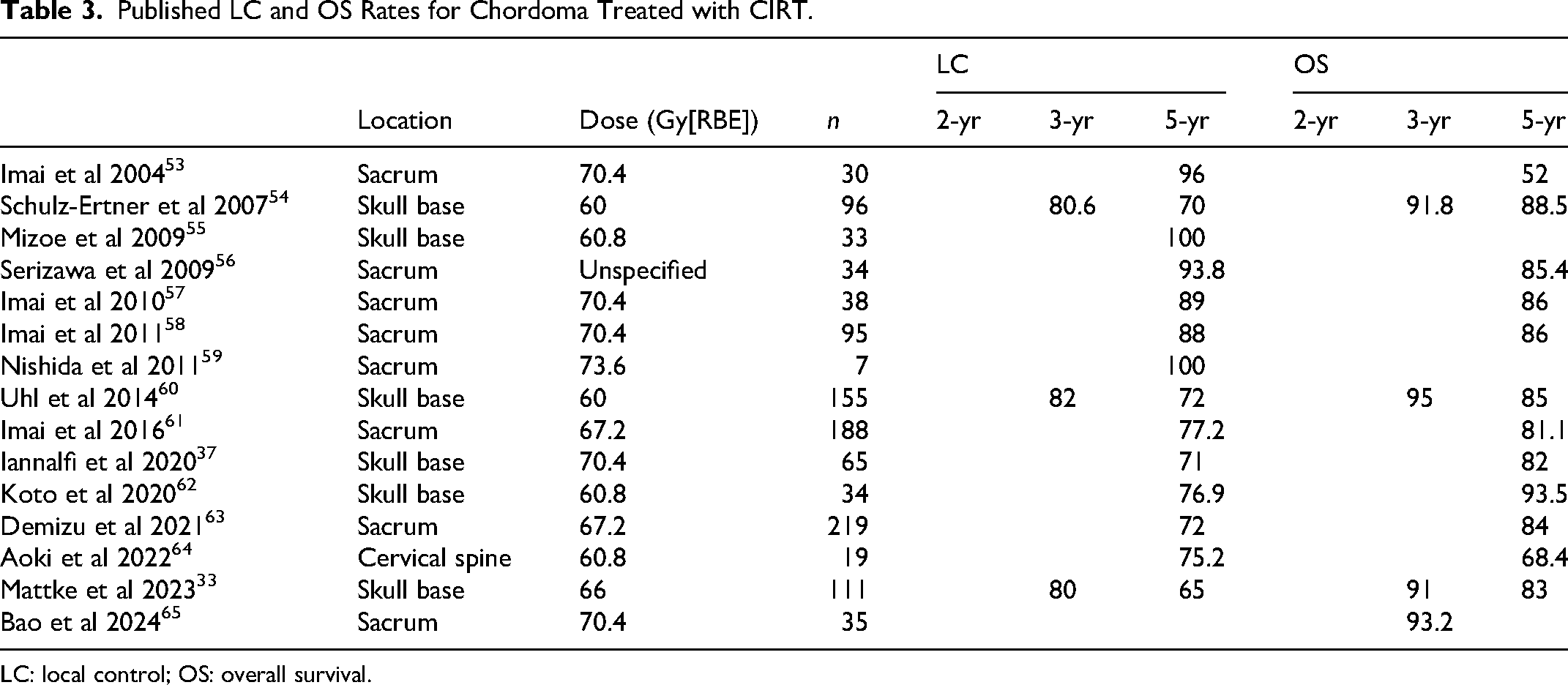
LC: local control; OS: overall survival.
Chondrosarcomas
Chondrosarcoma is a rare and heterogeneous group of malignant cartilage tumors that most commonly arise in the appendicular skeleton and pelvis, but can also occur in the spine and, more rarely, at the skull base.66,67 It is the second most common primary bone cancer, following osteosarcoma, and accounts for more than 20% of such cases. The estimated annual incidence in the United States is approximately 1 in 200,000, with most patients diagnosed after the age of 50, although it can occur at any age.68–70 Conventional chondrosarcoma represents about 85–90% of all cases. The remaining 10–15% consist of less common subtypes, including dedifferentiated, mesenchymal, and clear cell variants, each with distinct clinical, radiographic, and histopathological characteristics.71–73 Prognosis is closely linked to tumor grade: low-grade (grade I) tumors are indolent, with low rates of recurrence and metastasis, while high-grade (grade III) tumors are significantly more aggressive, often recurring locally and metastasizing – most commonly to the lungs – with markedly poorer long-term survival outcome. 72
Chondrosarcoma is known for its resistance to both chemotherapy and conventional radiation therapy, attributed to factors such as low mitotic activity, poor vascularity, a dense extracellular matrix, and the overexpression of drug resistance proteins.74,75 Recent molecular profiling has identified recurrent mutations – most notably in isocitrate dehydrogenase (IDH) 1 and 2 – which occur in approximately 50–75% of chondrosarcoma cases and represent potential therapeutic targets.76–80 Nonetheless, early clinical trials of IDH inhibitors in advanced disease have shown limited benefit, highlighting the need for combination strategies to enhance efficacy. 81
Given the limited effectiveness of systemic therapies, LC remains central to the management of chondrosarcoma. Tumors in the appendicular skeleton are typically more amenable to complete surgical resection with negative margins. In contrast, lesions located in the pelvis or vertebral columns are often near critical structures, making wide-margin resection technically challenging or impossible. 82 In such cases, adjuvant RT is frequently employed to enhance LC, and definitive RT may be considered for patients who are not surgical candidates.83–86 However, chondrosarcomas often require doses exceeding 70 Gy for adequate tumor control. Achieving such doses with conventional photon RT is often constrained by the tolerance of surrounding organs at risk, such as the spinal cord or bowel.74,85 Recent advances in RT technologies now make it possible to deliver high-dose radiation more precisely to the tumor while reducing exposure to nearby healthy tissues.4,14,74,87,88
Catanzano et al retrospectively analyzed 5427 chondrosarcoma patients from the National Cancer Database (NCDB), of whom 680 received RT, including both conventional external beam RT as well as advanced modalities such as IMRT and PBT. Their findings support the use of RT in selected high-risk patients – particularly those with tumors in surgically challenging locations or with unplanned positive margins. Notably, the use of advanced RT modalities delivering doses greater than 60 Gy was associated with improved OS compared to patients who did not receive RT. 88 These results suggest that, despite the traditionally perceived radioresistance of chondrosarcoma, modern radiation techniques may offer meaningful clinical benefit in selected cases.
As in chordoma, there has been a wealth of research conducted on evaluating PBT and CIRT in chondrosarcoma. The role of PBT in the management of skull-base chondrosarcoma is well established and has shown improved clinical outcomes compared to conventional RT, with excellent 5- and 10-year LC rates as high as 99% and 98% respectively, and 5- and 10-year disease-specific survival rates as high as 99%. 5 For non-skull-base extracranial chondrosarcoma, in a retrospective review of 44 patients by Bhatt et al, high-dose PBT was found to achieve LC rates of 76% at 2 years and 57% at 5 years, with an OS of 90% at 2 years and 68% at 5 years. Notably, PBT enabled safe dose escalation (>70 Gy), even in challenging spinal locations. 87 The year prior to that study, Weber and colleagues at the Paul Scherrer Institute (PSI) in Switzerland reported even more auspicious results, with a 5-year LC rate of 93.6% in a patient cohort of 71 individuals. 47 The year following, Mattke and team at the Heidelberg Ion Beam Therapy Center (HIT) reported a 100% LC and OS rate at 4 years in a patient cohort of 22 individuals. 89 More recently, a large retrospective analysis of pediatric patients with skull base chondrosarcoma once again confirmed that high-dose PBT following surgical resection achieves excellent disease control with minimal toxicity. 90 An overview of the various LC and OS rates reported for PBT in chondrosarcoma across various studies can be found in Table 4.
Published LC and OS Rates for Chondrosarcoma Treated with PBT.
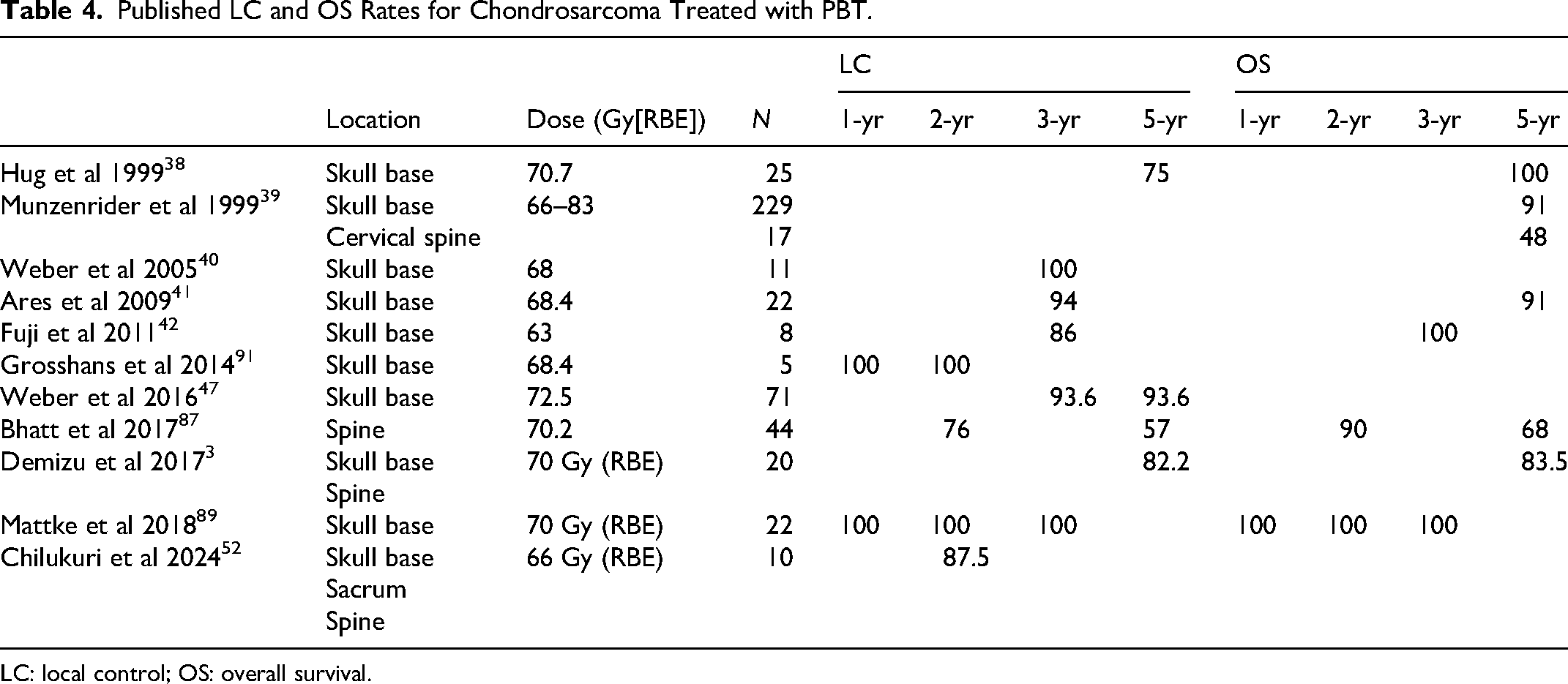
LC: local control; OS: overall survival.
A handful of studies have also evaluated the efficacy of CIRT in chondrosarcoma. Schulz-Ertner et al at the Gesellschaft für Schwerionenforschung (GSI) in Switzerland reported an actuarial 3-year LC rate of 96.2% and a 5-year OS rate of 98.2%. 92 Seven years later, Uhl et al at HIT reported 3- and 5-year LC rates of 95.9% and 88%, respectively, and a 5-year OS rate of 96.1%. 93 In 2017, Imai and colleagues at the National Institute of Radiological Sciences (NIRS) in Chiba, Japan reported decidedly more pessimistic actuarial statistics of 5-year LC and OS rates of 53% each, albeit in more difficult cases of unresectable chondrosarcoma. 94 Finally, the study previously mentioned by Mattke et al that evaluated PBT in chondrosarcoma also looked at CIRT, finding 1-, 2-, and 4-year LC rates of 98.6%, 97.2%, and 90.5%, respectively. Summary statistics of the aforementioned studies can be found in Table 5.
Published LC and OS Rates for Chondrosarcoma Treated with CIRT.

LC: local control; OS: overall survival.
Osteosarcoma
Osteosarcoma is the most common primary malignant tumor of the bone, with a bimodal pattern of peak incidence in adolescents and adults > 60 years of age. It is also the most frequent bone tumor in children and adolescents, with an incidence of 4.4 cases per million in this age group. 95 Although osteosarcoma can occur in any bone, the most frequent sites are around the knee and the proximal humerus. 96 Conventionally, osteosarcomas are classified into three types based on their predominant differentiation pattern: osteoblastic, chondroblastic, or fibroblastic. Additionally, they can be categorized into three major groups: low, intermediate, and high grade. Low-grade osteosarcomas tend to be indolent, whereas high-grade tumors are more aggressive and have a higher risk of metastasizing to the lungs, lymph nodes, and other bones.97,98 The standard treatment for osteosarcoma involves a combination of surgery and chemotherapy.97–99 This approach has resulted in long-term survival for over 60% of patients with localized osteosarcoma. However, outcomes for patients with metastatic osteosarcoma remain poor, with OS rates still below 30%.97–100
Although osteosarcoma is regarded as a radioresistant disease, RT may be considered in certain cases. Preoperative RT may be recommended in patients with tumors at high risk for resection with microscopically positive margins, while adjuvant RT is typically indicated in the setting of positive margins or for patients at high risk for local relapse. DeLaney et al found that RT was beneficial in enhancing LC in osteosarcoma patients for whom achieving negative surgical margins was infeasible. Osteosarcomas treated with RT following surgery had a LC rate of 78% for gross total or subtotal resection, compared to 40% for biopsy only, with OS being 74% for total or subtotal resections
With the incorporation of hadron therapies, unresectable or incompletely resected osteosarcomas have been able to be safely and effectively treated. Ciernik et al showed that PBT allows organ-preserving, locally curative treatment for unresectable or incompletely resected osteosarcoma, with a 5-year LC rate of 72%, a 5-year disease-free survival (DFS) rate of 65%, and a 5-year OS of 67%. 2 The Bone and Soft-Tissue Sarcoma Working Group in Japan investigated CIRT for unresectable pediatric and adult osteosarcoma, demonstrating a 5-year LC rate of 62% with a hypofractionated regimen of 70.4 Gy relative biological effectiveness (RBE) in 16 fractions.104,105 Researchers at HIT investigated the combination of PBT and CIRT in inoperable high-grade osteosarcoma. Their regimen, which they coined combined ion-beam radiotherapy (CIBRT), consisted of PBT up to 54 Gy RBE and a carbon ion boost of 18 Gy RBE. Using this hybrid technique, Seidensaal et al demonstrated 2-year progression-free survival (PFS) and OS rates of 45% and 68%, respectively. 106 For comparison, the 3-year OS rate of highly malignant osteosarcoma patients treated with surgery alone is less than 10%. 107
Thus, as in chordoma and chondrosarcoma, hadron therapies have achieved favorable toxicity profile and promising outcomes, especially for patients with inoperable osteosarcoma.
Ewing Sarcoma
Ewing sarcoma is the second most common primary malignant bone tumor in children, adolescents, and young adults (AYAs), with approximately 1.5 cases per million individuals in this age group worldwide.
108
At the genetic level, it is characterized by pathognomonic
Unlike the previous three bone malignancies, Ewing sarcoma is radiation-sensitive. However, the use of radiation alone for primary tumor treatment has decreased over the past decades due to advances in orthopedic surgery and concerns about radiation's long-term effects, such as secondary malignancies and growth disturbances, in children. The standard of care for Ewing sarcoma involves a multimodal treatment regimen, typically combining surgical resection with local RT and/or intensive multi-agent chemotherapy. 108 In patients with large tumors (>200 mL), poor histological response, or inadequate surgical margins, adjuvant RT (45-60 Gy) has been found to significantly reduce local recurrence. 7 For patients with bone Ewing sarcoma, RT with definitive intent should be used instead of surgery if complete surgical excision is not possible, particularly in cases with challenging local sites such as axial or spinal tumors, where surgery would result in unacceptable morbidity. These instances thus represent prime contenders for more focused RTs.
Modern treatment for Ewing sarcoma often incorporates advanced imaging techniques such as MRI/PET-CT fusion imaging to precisely delineate the tumor target area. This approach is crucial for treatment planning, especially with the advent of advanced RTs like IMRT, volumetric modulated arc therapy (VMAT) – a type of IMRT that uses rotating beams to conform the radiation dose to the tumor shape even more precisely and PBT. These techniques allow for better tumor targeting, reducing the dose to surrounding healthy tissues.
Among these advanced modalities, PBT has garnered increasing attention, particularly in pediatric cases, due to its ability to minimize late radiation effects, such as growth impairment and secondary malignancies.111–114 A retrospective review by Rombi et al of 30 children with Ewing sarcoma treated with PBT at a median dose of 54 Gy RBE reported 3-year actuarial rates of 60% event-free survival, 86% LC, and 89% OS. It was well tolerated among patients, with mostly mild-to-moderate skin reactions observed. 113 Subsequent studies conducted by the PSI 115 and the University of Florida Proton Therapy Institute 114 have generally placed 5-year OS rates at 83% and 85%, respectively with a 5-year LC rate exceeding 90% in the latter study. Additionally, most survivors regained long-term neurologic function with minimal side effects, highlighting the potential benefits of PBT in preserving quality of life while achieving excellent clinical outcomes.
An overview of the published LC and OS rates for PBT in Ewing sarcoma can be found in Table 6. The benefits of aggressive treatment of metastatic Ewing sarcoma with local therapy have been consistently supported by multiple studies,116–118 and, in cases of pulmonary metastases, whole-lung irradiation (WLI) remains the standard of care.119–121 However, SBRT has emerged as a promising alternative for consolidating metastatic sites and achieving durable tumor control. 10
Published LC and OS Rates for Ewing Sarcoma Treated with PBT.
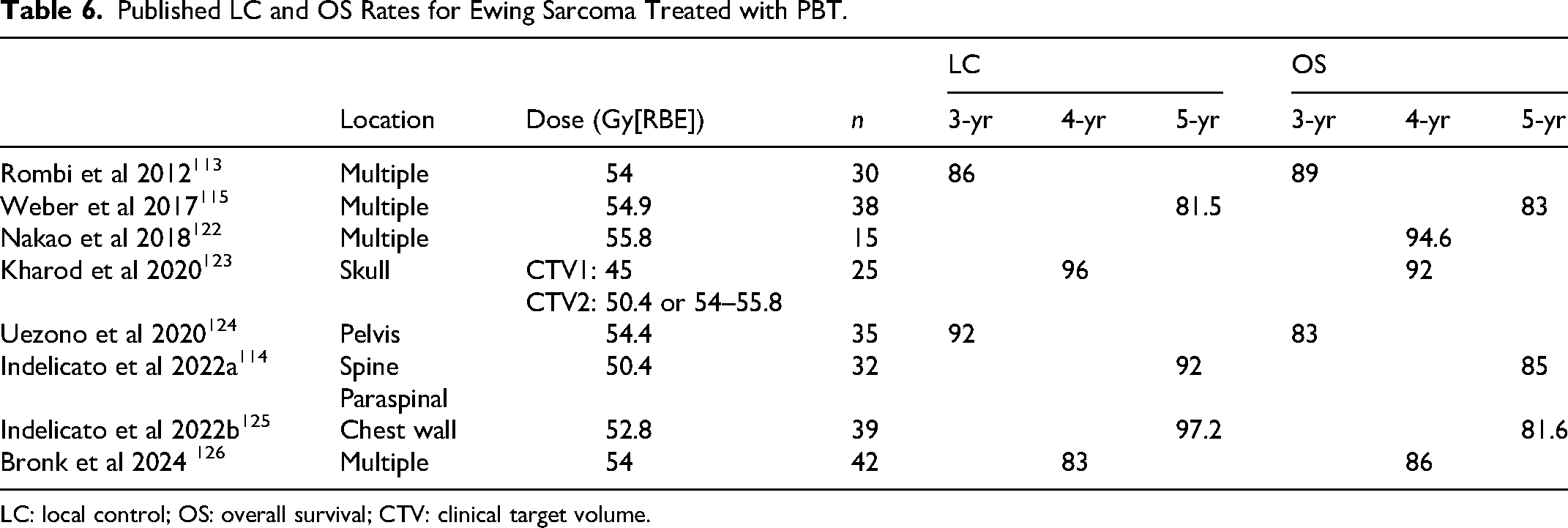
LC: local control; OS: overall survival; CTV: clinical target volume.
Adverse Events
Even with image guiding, beam shaping, or Bragg peak sharpening, off-target toxicity and secondary malignancies still occur following RT. Adverse events arise in both the short and long-term, and run the gamut from mild radiation dermatitis to permanent loss of vision or hearing, brain damage, and secondary malignancies. 7 Pediatric patients are particularly vulnerable, as their ongoing development renders them more susceptible to radiation injury. In addition to the toxicities noted above, children may experience chronic, serious, and permanently debilitating sequelae, including stunted growth, limb asymmetries, bone fractures, and learning disabilities.7,23
Refinements to RT, both photonic and hadronic, have fortunately reduced the incidence and severity of these side effects, 127 though severe toxicities still affect a sizeable minority of patients. For example, Kacar et al reported that, among 32 patients with Ewing sarcoma treated with photon or proton RT, eight experienced grade ≥3 acute dermatitis, and nine experienced grade ≥3 late toxicities involving in the bones, joints, skin, and subcutaneous tissue, including one secondary malignancy. 128 Bao et al observed no grade ≥3 acute toxicities among 35 patients with unresectable sacrococcygeal chordoma treated with hypofractionated, image-guided CIRT, and only three cases of grade 3 late toxicities. 65
Direct comparisons of the long-term toxicities between older and newer RT modalities are thin on the ground. Much of the supposed advantage of PBT derives from dosimetric modeling that compares administered PBT plans with hypothetical photon-based treatments. 129 In a rare comparative study published in 2012, Childs et al compared pediatric rhabdomyosarcoma patients receiving PBT to two previously published cohorts treated with conventional external beam RT (EBRT). 130 Among those few PBT patients available for long-term follow-up, the authors reported reduced rates of stunted growth, facial hypoplasia, dental deformities, chronic nasal congestion, and – critically – secondary malignancies relative to the historical EBRT patient data. Collectively, the existing data support modest improvements in long-term outcomes with advanced RT techniques. Hadron therapies appear to offer the most consistent reductions in severe adverse events, 131 and are therefore often preferred for pediatric patients. However, this preference is not universal due to limited availability, high costs, and the absence of definitive evidence establishing their superiority.132,133
Current Challenges and Future Directions
Despite the advancements in RT that have been realized over the past two decades, RT remains largely an adjunctive modality, with surgical excision preferred as first-line treatment when feasible. 7 When surgery is not feasible, RT is commonly employed as a second-line option. Although technical advancements have improved outcomes for patients with bone malignancies, substantial challenges and opportunities for further improvement remain.
While emerging evidence indicates superior outcomes with hadron therapies compared with photon-based therapies, this evidence remains incomplete and contested. Despite the increasing number of treatment centers and continued technological refinement (eg, pencil beam scanning and cone beam computed tomography), hadron therapy remains a limited and heavily rationed resource. This restricted availability has hindered direct comparisons of clinical outcomes between photon and hadron-based therapies. Hadron therapy patients represent a relatively small population, and bone cancers are already a rare subset, making large, randomized controlled trials particularly difficult to conduct. Consequently, robust comparative data within this patient population are limited. Nevertheless, most studies that have attempted the comparison have found that hadron therapy meets or exceeds patient outcomes achieved with x-ray RT – at least in bone malignancies,134–136 with little to no significant difference observed between PBT and CIRT. 23
Focusing on comparing PBT and x-ray RT, then, the question becomes whether this enhancement in outcome – which can range from substantial to negligible – is great enough to justify referring a given case for PBT, with the consideration that, even setting aside the cost, treating every case with this technology simply is not feasible. There is no simple answer to this, but the ongoing expansion of PBT technology and infrastructure, paired with the development of ethical patient selection criteria,137–139 suggest growing clinical confidence in the role of PBT for treating bone malignancies when and where available.
In deciding which cases should be referred for hadron therapy, consideration should be given to the location of the tumor (ie its proximity to vital/eloquent organs) and patient age, with pediatric patients given priority access to hadron therapies to limit off-target effects. Chordoma and Ewing sarcoma are good candidates, given the former's proximity to the central nervous system and the latter's radiosensitivity. In both cancers, the available evidence favors PBT, though determination of which treatment to pursue should, of course, be made on a case-by-case basis by the presiding physician. In contrast, chondrosarcoma and osteosarcoma, owing to their indolent, osseous natures, remain radio-insensitive. 140 As a result, RT is unlikely to assume a primary role in their management, instead being reserved for unresectable disease or margin control adjacent to surgery. In these cases, hadron therapy again appears to yield superior outcomes compared with conventional photon RT. 74
While efforts to expand access to PBT for appropriately selected patients continue, several ongoing and planned clinical trials aim to provide the evidentiary justification for this expansion (Table 1). The majority of ongoing studies are currently evaluating the utility of PBT in chordoma (NCT00496119, 01346124, and 01811394) and chondrosarcoma, (NCT05861245, 00496522, 01449149, and 01346124), as well as in pediatric bone sarcoma (NCT00592293). In one of these trials (NCT01346124), the proton therapy employed will also be intensity modulated, adding a further layer of refinement to what is already a promising therapy. In addition to these, SBRT is currently being evaluated in conjunction with the anti-PD-1 antibody atezolizumab in relapsed osteosarcoma (NCT06492954). Lastly, CIRT is being assessed in chordoma in two independent trials (NCT01811394 and 02838602).30,32
While today, the number of available hadron therapy centers remain limited, this number is increasing, and, as it does, the treatments made possible by these technologies will become increasingly accessible to more patients. In the meantime, improvements in traditional photon RT have led to better outcomes, providing an effective intermediate option that has significantly enhanced the quality of life for many patients. Furthermore, the development of targeted therapies, such as those targeting mutant IDH in chondrosarcoma, holds the potential for synergistic effects when combined with radiation therapy, offering hope for more effective treatments in the future.
Conclusion
While surgery and chemotherapy remain the cornerstone of bone sarcoma treatment, RT has become an increasingly critical component in managing these aggressive tumors. Advances in RT techniques, such as IMRT, SBRT, PBT, and CIRT, have significantly enhanced treatment precision. These innovations enable clinicians to deliver higher radiation doses to tumors while minimizing damage to surrounding healthy tissues. As a result, outcomes such as LC, survival rates, and reduced recurrence have improved, while long-term side effects have been minimized. Looking toward the future, the integration of advanced RTs with systemic treatments such as immunotherapy, targeted therapies, and novel chemotherapy regimens holds great potential to transform the landscape of bone sarcoma care and further improve patient outcomes. These combination approaches may optimize therapeutic efficacy, particularly in difficult-to-treat or metastatic cases. As the field continues to evolve, ongoing research will be critical in refining RT strategies, enhancing their overall effectiveness, and enabling more personalized, tailored treatment options for patients with bone sarcomas.
Footnotes
Abbreviations
Acknowledgment
We thank Genevieve Bertolet for her contributions to this review.
Ethics Statement
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute of the NIH under award P30CA240139 (Sylvester Comprehensive Cancer Center/University of Miami).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
