Abstract
Introduction
Chronic hand eczema (CHE) is a prevalent and challenging skin disorder, affecting 10% of adults worldwide. 1 Characterized by persistent inflammation, pruritus, erythema, and scaling, CHE has a substantial impact on patients’ quality of life. The chronic nature of the disease often leads to frequent relapses, and current treatment options, such as topical corticosteroids, are limited by their side effects, including skin thinning and impaired skin barrier function.
Recent advancements in understanding the immunological mechanisms underlying CHE have identified Janus kinase (JAK) inhibitors as promising therapeutic agents. 2 JAK inhibitors target multiple signaling pathways involved in the inflammatory process, potentially offering improved control over the disease. Delgocitinib, a pan-JAK inhibitor, has shown efficacy in modulating immune responses and restoring skin barrier function in patients with CHE. This systematic review and meta-analysis aimed to synthesize the available evidence on the efficacy and safety of delgocitinib in treating CHE.
Methods
Study design
This systematic review and meta-analysis followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 3
Data sources and search strategy
A comprehensive search was conducted in the PubMed, Scopus, Embase, and Cochrane Central databases to identify relevant randomized controlled trials (RCTs) published from inception up to September 7, 2024. The search terms included combinations of “chronic hand eczema,” “delgocitinib,” “JAK inhibitor,” “randomized controlled trial,” “treatment success,” “adverse events,” and “safety.” In addition, reference lists of selected studies were manually searched to identify additional studies.
Eligibility criteria
Studies were included if they met the following criteria: RCTs that evaluated the efficacy of delgocitinib for CHE, and patients aged ≥18 years diagnosed with CHE. Outcomes of treatment success at weeks 8 and 16 reported, defined as achieving clear or almost clear skin on the Investigator’s Global Assessment (IGA) or Physician’s Global Assessment (PGA) scale with at least a two-point improvement from baseline. Data on adverse events reported, allowing for the assessment of the treatment’s safety profile.
The exclusion criteria were non-English publications, non-randomized trials, case reports, or observational studies and studies lacking data on the primary outcomes of interest (treatment success or adverse events).
Data extraction and quality assessment
Two independent reviewers (CSK and SSH) screened the titles and abstracts of all identified studies. Full-text articles were retrieved for potentially relevant studies, and disagreements were resolved by discussion with a third reviewer (KT). Data extracted included study design, patient characteristics, treatment regimens, and outcomes for treatment success at weeks 8 and 16, as well as reported adverse events.
The Cochrane Risk of Bias tool 4 was used to assess the quality of the included RCTs. Risk of bias was evaluated in terms of randomization, blinding of participants and personnel, incomplete outcome data, and selective reporting. For each of these domains, trials were categorized as having a low risk of bias, some concerns, or a high risk of bias, and an overall risk of bias rating was subsequently assigned. Trials that showed a low risk of bias in all domains were considered to have a low overall risk, indicating high methodological rigor and confidence in their findings.
Outcome measures
The Investigator’s Global Assessment (IGA) and Physician’s Global Assessment (PGA) are both standardized scoring systems used to evaluate the severity of skin conditions such as eczema. The IGA is generally employed by clinical investigators in trials, while the PGA is a broader term that encompasses assessments by healthcare professionals such as physicians. Both scales rank the severity of the disease on a five-point scale.
For the purpose of this meta-analysis, treatment success was defined as achieving a score of 0 (clear) or 1 (almost clear) with a reduction of at least two points from baseline on either the IGA or PGA scale. The studies also reported the incidence of adverse events, which were analyzed to assess the safety and tolerability of delgocitinib.
Statistical analysis
For the meta-analysis, pooled odds ratios (ORs) with 95% confidence intervals (CIs) were calculated for the primary outcomes of treatment success at weeks 8 and 16, using the Mantel-Haenszel random-effects model. Heterogeneity among studies was assessed using the I2 statistic, which quantifies the percentage of variation across studies due to heterogeneity rather than chance. I2 values greater than 50% were considered indicative of significant heterogeneity, suggesting that the true effects of the intervention might vary between studies due to differences in study populations, methodologies, or other factors. All analyses were conducted using MetaXL, version 5.3 (EpiGear International, Queensland, Australia), and statistical significance was set at a p-value less than 0.05.
Results
Study selection
Characteristics of included trials.
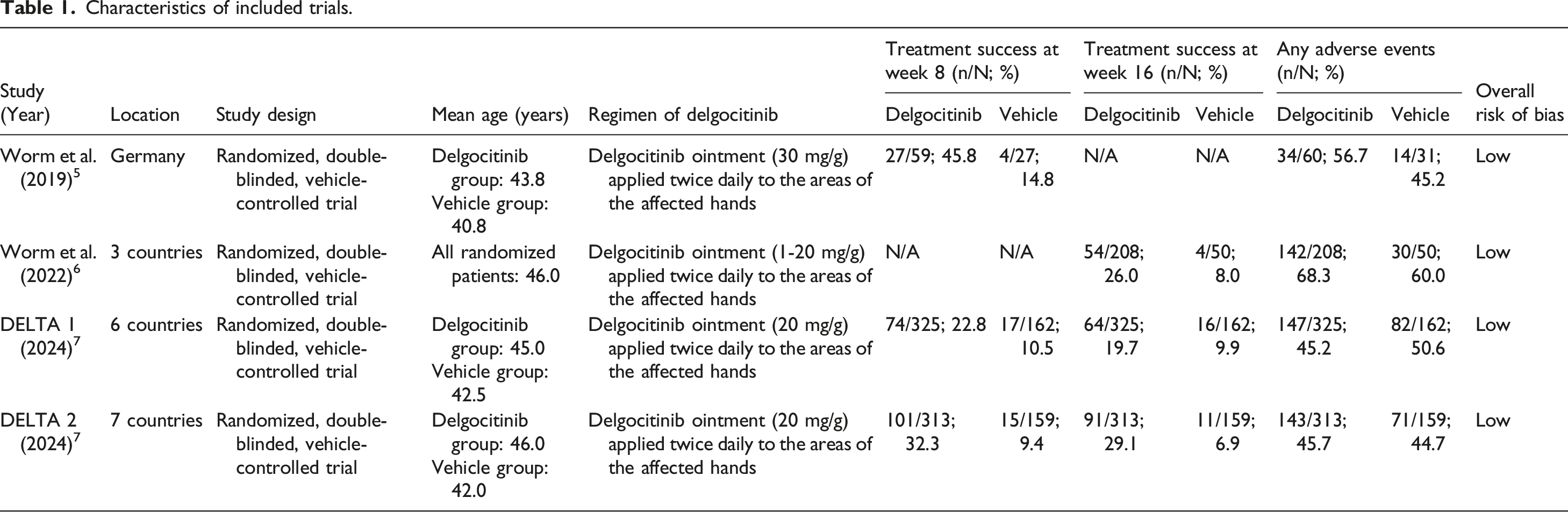
Study characteristics
The included studies5–7 comprised four randomized, double-blinded, vehicle-controlled trials conducted between 2020 and 2024, evaluating the efficacy and safety of delgocitinib ointment for the treatment of CHE. Participants were adults aged ≥18 years with moderate-to-severe CHE, defined by an Investigator’s or Physician’s Global Assessment (IGA/PGA) score of 3 or 4 at baseline, and were required to have an inadequate response or contraindication to topical corticosteroids.
Baseline characteristics were comparable between delgocitinib and vehicle groups, with mean ages ranging from 40.8 to 46.0 years and 40–60% of participants reporting a history of atopy. Patch testing was performed according to local clinical practice to identify allergic contact dermatitis, though positive but inactive sensitization was not exclusionary.
The trials were conducted across diverse geographic regions, ranging from a single-country (Germany) phase II study to multinational phase III programs involving up to seven countries. The delgocitinib concentration ranged from 1 mg/g to 30 mg/g, administered twice daily for 8 to 16 weeks. All studies permitted the use of non-medicated moisturizers under standardized conditions (prohibited within 2 h before or after study drug application) and reported treatment adherence exceeding 90%.
Treatment success at week 8
Pooled analysis from the four trials demonstrated a significant improvement in treatment success at week 8 for patients treated with delgocitinib compared to the vehicle. The pooled OR was 3.49 (95% CI 2.26 to 5.41; see Figure 1), indicating that delgocitinib significantly improved outcomes in chronic hand eczema during the initial treatment period. Pooled odds ratio of treatment success at weeks 8 and 16, and overall adverse events for patients treated with delgocitinib compared to the vehicle.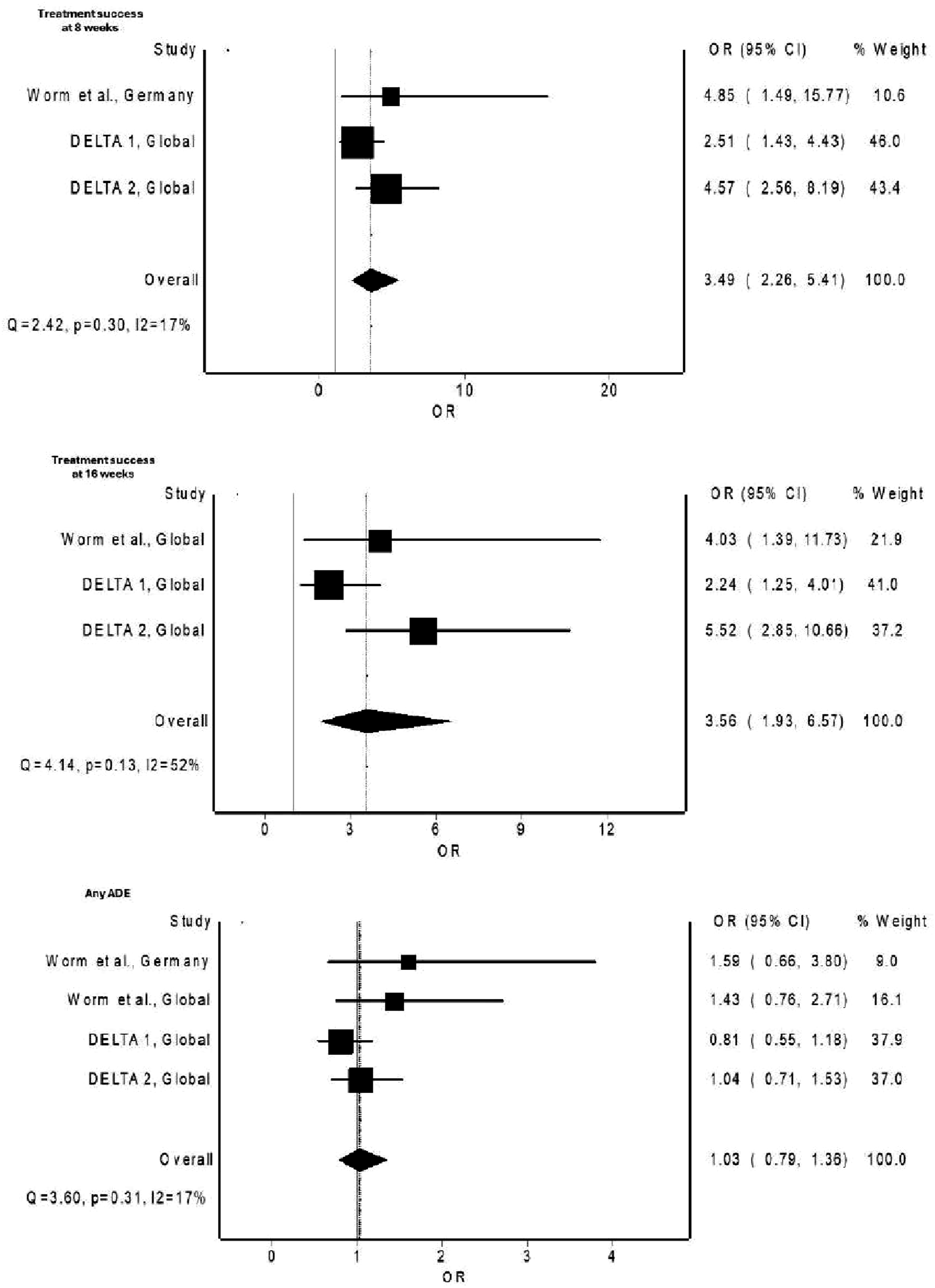
Treatment success at week 16
By week 16, the pooled results confirmed the sustained efficacy of delgocitinib. The pooled OR for treatment success at this time point was 3.56 (95% CI 1.93 to 6.57), further demonstrating the long-term effectiveness of delgocitinib in managing chronic hand eczema.
Adverse events
Adverse events were consistently reported across all included trials, with the overall incidence of any adverse event ranging from 45% to 68% in the delgocitinib groups, comparable to 45–51% in the vehicle arms. These figures represent the proportion of participants experiencing any event, regardless of causality, rather than treatment-related adverse events. The most frequently reported adverse events were nasopharyngitis, headache, eczema flare, and mild application-site reactions such as pruritus or erythema, all of which were transient and non-serious. No deaths or treatment-related serious adverse events occurred in any trial.
Adverse events were consistently reported across all studies. The pooled OR for overall adverse events was 1.03 (95% CI 0.79 to 1.36), indicating no significant difference in the risk of adverse events between delgocitinib and vehicle treatments.
Discussion
This systematic review and meta-analysis assessed the efficacy and safety of delgocitinib in the treatment of CHE by pooling data from four RCTs. The results demonstrate significant improvements in treatment success at both week 8 and week 16, with no significant increase in adverse events compared to vehicle treatments. These findings suggest that delgocitinib offers a valuable therapeutic option for managing CHE, combining efficacy with a favorable safety profile.
The therapeutic benefits of delgocitinib in CHE can be largely attributed to its role as a pan- JAK inhibitor, targeting key inflammatory pathways involved in the pathogenesis of eczema. The JAK-STAT (signal transducer and activator of transcription) pathway is central to regulating immune responses in the skin, and it has been implicated in various inflammatory and autoimmune diseases, including CHE. 8
In CHE, immune dysregulation leads to chronic inflammation driven by both innate and adaptive immune responses. This immune activation is mediated by pro-inflammatory cytokines such as interleukin (IL)-4, IL-13, IL-22, IL-31, and tumor necrosis factor-alpha (TNF-α), which rely on JAK signaling for downstream effects. 9 These cytokines promote skin inflammation, disrupt the skin barrier, and exacerbate symptoms such as itching, redness, and scaling. Delgocitinib acts by inhibiting multiple members of the JAK family (JAK1, JAK2, JAK3, and tyrosine kinase 2), thereby blocking the signal transduction of these cytokines and mitigating their inflammatory effects. 10
The broad inhibition of multiple JAK pathways allows delgocitinib to target various aspects of the inflammatory cascade. For example, blocking JAK1 and JAK3 inhibits signaling of the Th2 cytokines IL-4 and IL-13, which are crucial drivers of atopic inflammation. This helps reduce the chronic itch and inflammatory responses associated with eczema. Similarly, by inhibiting JAK2 and TYK2, delgocitinib interferes with the signaling of cytokines like IL-22 and IL-31, which are involved in skin barrier dysfunction and keratinocyte hyperproliferation, contributing to the physical symptoms of eczema such as thickened, scaly skin.
A key limitation of this analysis is the relatively short follow-up periods of the included studies, which were restricted to 16 weeks. This limits the ability to assess the long-term effects of delgocitinib, particularly in chronic conditions like CHE, where ongoing treatment is often required. Future research should focus on extended follow-up periods to determine the durability of delgocitinib’s benefits and to compare its long-term efficacy and safety with other emerging treatments for CHE.
Conclusion
In conclusion, delgocitinib ointment significantly improves treatment success in chronic hand eczema at early and sustained time points without increasing the risk of adverse events. These findings position delgocitinib as a promising therapeutic option for CHE, offering both efficacy and safety. Continued research with longer-term studies will be essential to further establish its role in the management of this debilitating condition and to support its potential for regulatory approval.
Footnotes
Authors contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
