Abstract
Keywords
Introduction
Atopic dermatitis (AD) is a chronic, relapsing inflammatory skin disease characterized by intense pruritus, eczematous lesions, and substantial impairment of quality of life. 1 Current treatment options, such as corticosteroids and calcineurin inhibitors, are effective but come with limitations, including the potential for adverse effects with long-term use. These limitations have led to a growing need for safer, nonsteroidal therapeutic alternatives.
Tapinarof, a novel topical aryl hydrocarbon receptor (AhR) agonist, has emerged as a potential treatment option for AD due to its ability to modulate immune responses and improve skin barrier function. 2 By reducing inflammation and oxidative stress, tapinarof offers a promising approach for managing the symptoms of AD while minimizing risk of adverse effects associated with existing treatments. Given the increasing interest in tapinarof, this study aimed to synthesize the available evidence on its efficacy and safety. By pooling data from multiple studies, this analysis provided a comprehensive assessment of tapinarof’s therapeutic potential in AD.
Methods
This systematic review and meta-analysis was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 3
Search strategy and study selection
A systematic search was conducted across multiple databases, including PubMed, Scopus, Embase, Cochrane Library, and Web of Science, covering studies from inception to September 10, 2024. The search terms used were “tapinarof,” “atopic dermatitis,” “treatment success,” “adverse events,” “efficacy,” and “safety,” combined with Boolean operators (“AND” and “OR”) to refine results. Reference lists of key articles were manually screened to identify additional relevant studies. The search was restricted to English-language studies to ensure consistency in analysis. The detailed strategies were as follows: • PubMed: (‘tapinarof’ OR ‘AhR agonist’ OR ‘GSK2894512’) AND (‘atopic dermatitis’ OR ‘eczema’ OR ‘atopy’) AND (‘randomized’ OR ‘controlled trial’ OR ‘RCT’). Filters: Humans; Randomized Controlled Trial; English. • Embase: (‘tapinarof’ OR ‘GSK2894512’ OR ‘aryl hydrocarbon receptor agonist’) AND (‘atopic dermatitis’ OR ‘eczema’ OR ‘atopy’) AND (‘randomized controlled trial’ OR ‘clinical trial’). Search fields: title/abstract/keywords. • Scopus: TITLE-ABS-KEY((tapinarof OR GSK2894512 OR AhR agonist) AND (atopic dermatitis OR eczema OR atopy) AND (randomized OR trial)). • Web of Science: TS = ((tapinarof OR AhR agonist OR GSK2894512) AND (atopic dermatitis OR eczema OR atopy) AND (randomized OR controlled trial OR RCT)). • Cochrane CENTRAL: (tapinarof OR AhR agonist OR GSK2894512) in Title, Abstract, or Keywords AND (atopic dermatitis OR eczema OR atopy).
After retrieving citations, duplicates were removed using reference management software. The study selection process included three stages: title screening, abstract screening, and full-text review. Two independent reviewers (CSK and KT) screened titles and abstracts to assess eligibility, and full-text articles were obtained for studies deemed relevant. Any disagreements were resolved through discussion or by consulting a third reviewer (SSH).
Inclusion and exclusion criteria
Studies were selected based on the following inclusion criteria: patients of any age diagnosed with atopic dermatitis according to established clinical criteria (e.g., Hanifin and Rajka); intervention using topical tapinarof (any concentration); comparator groups involving placebo, vehicle cream, or other active comparators; and outcomes reporting treatment success after ≥8 weeks and adverse events. Only randomized controlled trials (RCTs) were included.
Studies were excluded if they were non-randomized, uncontrolled, did not report outcomes related to tapinarof, involved combination therapies without isolating tapinarof’s effect, or were preclinical studies, reviews, editorials, or case reports.
Data extraction
Data extraction was performed independently by two reviewers (CSK and SSH) using a standardized data extraction form. Discrepancies between reviewers were resolved through discussion or by consulting a third reviewer (KT). Extracted data included study characteristics (authors, publication year, country, and study design), population characteristics (sample size and age), intervention details (concentration and frequency of tapinarof application), comparator details (e.g., placebo or vehicle cream), and outcomes (number of patients achieving treatment success and experiencing adverse events).
Risk of bias and quality assessment
The quality of the included studies was assessed using the Cochrane Risk of Bias (RoB 2) tool, 4 specifically designed for RCTs. This tool evaluates bias across five domains: random sequence generation, allocation concealment, blinding, incomplete outcome data, and selective reporting. The overall risk of bias for each study was adjudicated as “low,” “some concerns,” or “high” based on the collective assessment of these five domains. Studies were considered to have a low overall risk of bias if they were judged as low risk across all domains. If one or more domains raised concerns but none were classified as high risk, the study was deemed to have some concerns. Studies were considered to have a high risk of bias if one or more domains were assessed as high risk, significantly affecting the reliability of the results.
Statistical analysis
Meta-analyses were performed using MetaXL (version 5.3, EpiGear International Pty Ltd, Queensland, Australia). Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated for dichotomous outcomes to assess the strength of associations between tapinarof and the key outcomes, including treatment success and adverse events.
A random-effects model was employed to account for heterogeneity among studies and to provide more generalizable results, as it assumes that variations exist not only within studies but also between studies.
Statistical heterogeneity was evaluated using the I2 statistic, which quantifies the proportion of variability in effect estimates that is due to heterogeneity rather than chance. The I2 values were interpreted as follows: 0%–25% Low heterogeneity; 26%–50%: Moderate heterogeneity; 51%–75%: Substantial heterogeneity; >75%: Considerable heterogeneity. Additionally, the Q-test (Cochran’s Q) was used to assess whether observed differences in study results are likely due to chance. A p-value <0.10 for the Q-test indicated significant heterogeneity.
Results
Study selection
Characteristics of included trials.

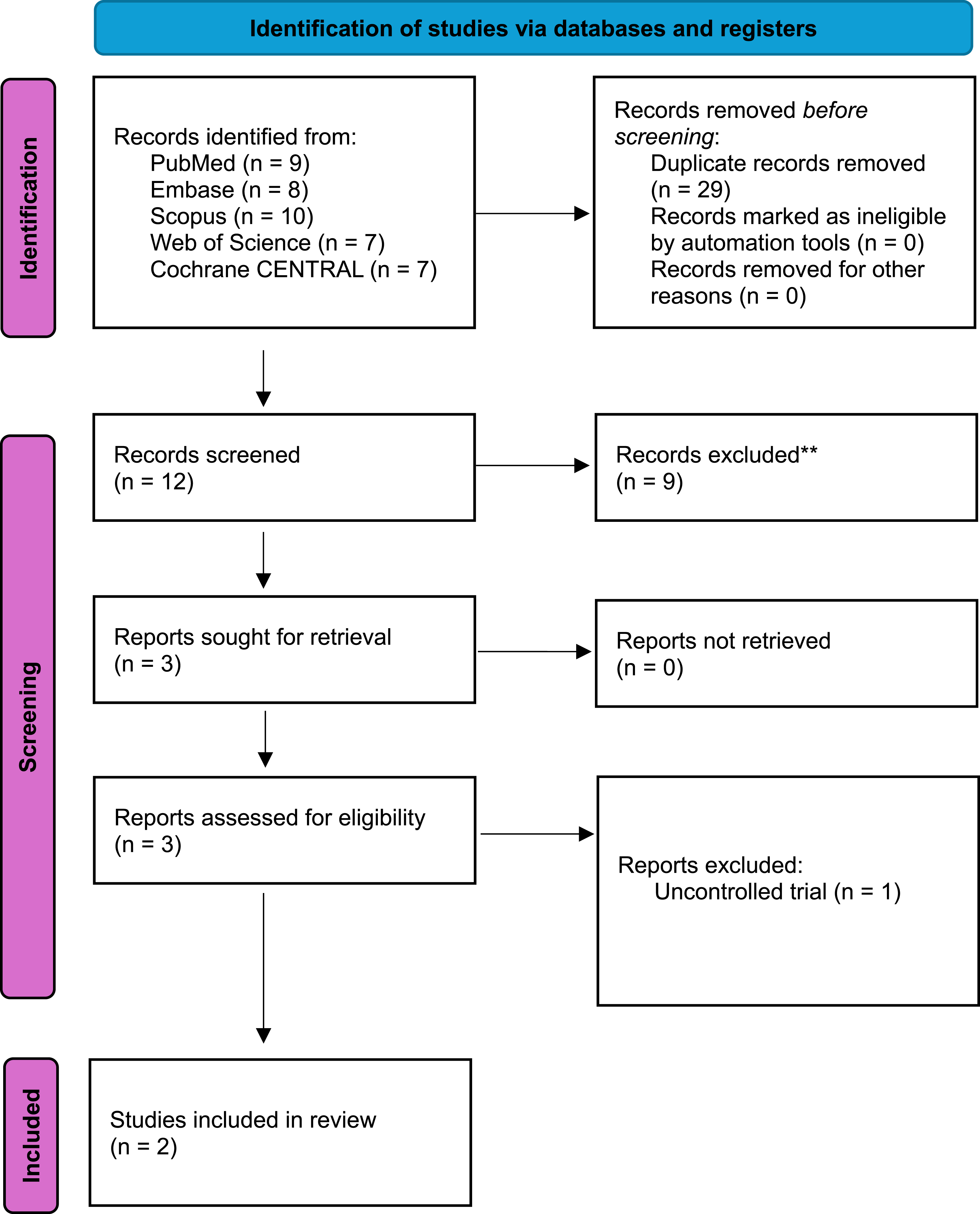
PRISMA 2020 flow diagram of study selection.
Study characteristics
The three included trials were randomized, double-blinded, vehicle-controlled trials conducted between 2020 and 2024 across multiple countries. The RCT by Peppers et al., 5 which involved participants with a mean age of 29.3 years, used tapinarof cream in concentrations of 0.5%–1%, applied once or twice daily, and defined treatment success using the Investigator Global Assessment (IGA), requiring clear or almost clear and a minimum 2-grade improvement. The ADORING 1 and 2 trials 6 included younger participants with mean ages of 15.6 and 16.4 years, respectively, and assessed tapinarof 1% applied once daily. In these two trials, treatment success was measured using the Validated IGA for Atopic Dermatitis (vIGA-AD), requiring a 2-grade or greater improvement from baseline with a final score of clear or almost clear. Although Peppers et al. 5 used IGA and the ADORING trials 6 used vIGA-AD, both instruments share identical five-point ordinal structure and clinical anchors, representing conceptually equivalent endpoints.
Treatment success
The pooled analysis of the three included trials demonstrated a significant improvement in treatment success for patients treated with tapinarof compared to those receiving the vehicle. The pooled odds ratio (OR) for treatment success was 3.58 (95% CI 2.20 to 5.82; I2 = 59%, p = 0.09) (see Figure 2), indicating that patients treated with tapinarof were over three times more likely to achieve the desired treatment outcomes, compared to those using the vehicle. Pooled odds ratios of treatment success and adverse drug events for patients treated with tapinarof compared to those receiving vehicle.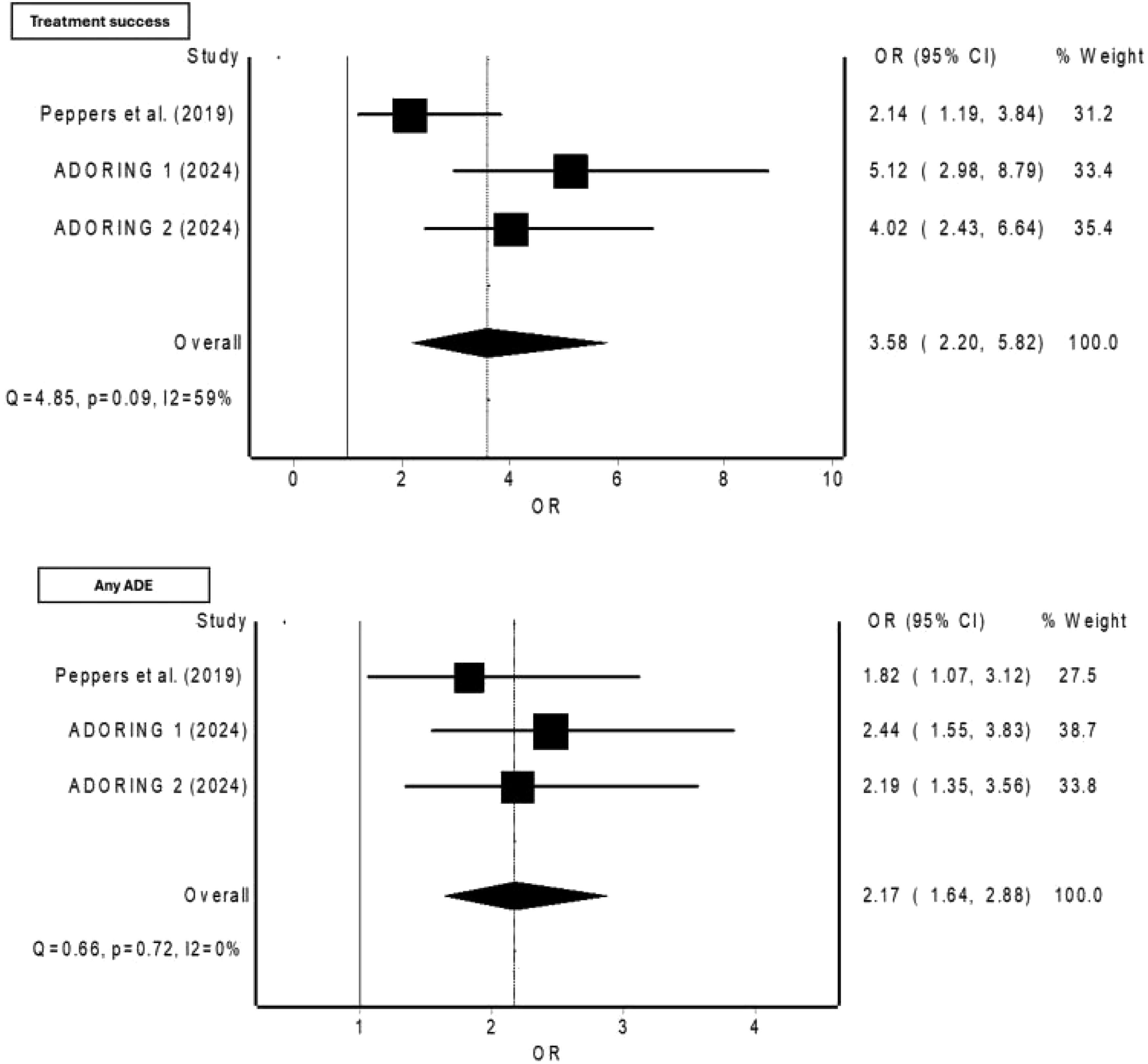
Adverse events
The pooled analysis of adverse events across the included trials revealed that patients treated with tapinarof had a higher likelihood of experiencing adverse events compared to those in the vehicle group. The pooled odds ratio (OR) for adverse events was 2.17 (95% CI 1.64 to 2.88; I2 = 0%, p = 0.72) (see Figure 1), indicating that tapinarof-treated patients were more than twice as likely to report adverse events.
Discussion
The findings of this systematic review and meta-analysis provide significant evidence supporting the efficacy of tapinarof in the treatment of atopic dermatitis (AD). The pooled analysis demonstrated a strong treatment effect, with patients receiving tapinarof being over three times more likely to achieve treatment success compared to those using a vehicle. This is consistent with previous research showing that tapinarof, a nonsteroidal aryl hydrocarbon receptor agonist, effectively modulates immune responses and restores skin barrier function, which are critical in managing the symptoms of AD. 7 The consistency of the results across all three trials, despite differences in the patient populations and tapinarof concentrations, further supports its robust efficacy.
In terms of safety, the pooled analysis showed a that patients treated with tapinarof were more than twice as likely to experience adverse events compared to those receiving the vehicle. However, it is important to note that the adverse events reported in the included studies5,6 were generally mild to moderate, such as skin irritation and nasopharyngitis, which did not significantly impact the continuation of treatment. This suggests that while tapinarof may increase the risk of certain adverse effects, these are typically manageable and do not outweigh the benefits of treatment success.
The results of this review are in line with the current need for effective, nonsteroidal treatment options for AD, especially considering the limitations and side effects associated with long-term use of topical corticosteroids and calcineurin inhibitors. 8 Tapinarof’s favorable efficacy and manageable safety profile make it a promising alternative for patients with AD, particularly those seeking nonsteroidal therapy options. 9 However, clinicians should remain mindful of the potential for adverse events and weigh these risks against the clinical benefits when considering tapinarof for treatment.
One limitation of this analysis is the relatively short follow-up period across the included studies, which limits conclusions on long-term outcomes. Further research, particularly long-term studies, is needed to better understand the sustained efficacy and safety of tapinarof in diverse populations and to solidify its role in the treatment of AD, especially as it seeks broader regulatory approval.
Conclusion
In conclusion, our review supports use of tapinarof as an effective treatment for atopic dermatitis, with a strong likelihood of achieving significant clinical improvement. While the risk of adverse events is elevated, these side effects are generally mild and manageable, suggesting that tapinarof may offer a valuable alternative for patients seeking nonsteroidal treatment options for AD.
Footnotes
Authors contributions
Chia Siang Kow: Conceptualization; data curation; formal analysis; roles/writing – original draft; writing – review & editing; final approval of the version to be published. Syed Shahzad Hasan: Formal analysis; validation; roles/writing – original draft; writing – review & editing; final approval of the version to be published. Kaeshaelya Thiruchelvam: Formal analysis; validation; roles/writing – original draft; writing – review & editing; final approval of the version to be published.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
