Abstract
Eczema herpeticum is an uncommon complication of atopic dermatitis, but often has a typical recognisable appearance. This report serves to highlight this feature in a patient who was misdiagnosed initially with a bacterial skin infection and then with a severe cutaneous drug reaction.
Introduction
Eczema herpeticum (EH) is an uncommon complication of poorly controlled atopic dermatitis where there is superimposed herpes simplex virus type 1 (HSV-1) infection. If disseminated, it may be mistaken for Stevens–Johnson syndrome, especially if mucosal erosions are present.
Clinical presentation
A 38-year-old Chinese engineer presented with an erosive painful vesicular rash over his face, neck, trunk and limbs, with sparing of orogenital mucosa.
He had presented to the emergency department three days previously with a six-day history of an itchy and painful rash over his face. There were no oral or genital ulcers, visual disturbances or evidence of photosensitivity on other exposed parts of the body. There was no preceding upper respiratory tract infection, use of oral or topical drugs or cosmetics or exposure to sick contacts. He was treated for impetigo with a course of oral amoxicillin-clavulanate.
He subsequently returned to the Emergency Department when the rash became painful and had progressed to involve his trunk and then limbs.
He had a background of poorly controlled atopic dermatitis which mainly affected his face, trunk and flexural regions of his limbs. He was previously treated with azathioprine, but he defaulted on the treatment and switched to Traditional Chinese Medicine. He had no history of herpes simplex infection or EH.
Physical examination revealed multiple confluent monomorphic circular erosions with yellowish crusting over his face, trunk and limbs on a background of thickened lichenified skin (Figures 1 and 2). No intact vesicles were present. Conjunctivae, buccal mucosae and anogenital mucosae were not inflamed. Nikolsky’s sign was negative. There was no lymphadenopathy, keratoconjunctivitis, meningism or change in mentation. The impression of the emergency physician was Stevens–Johnson syndrome.
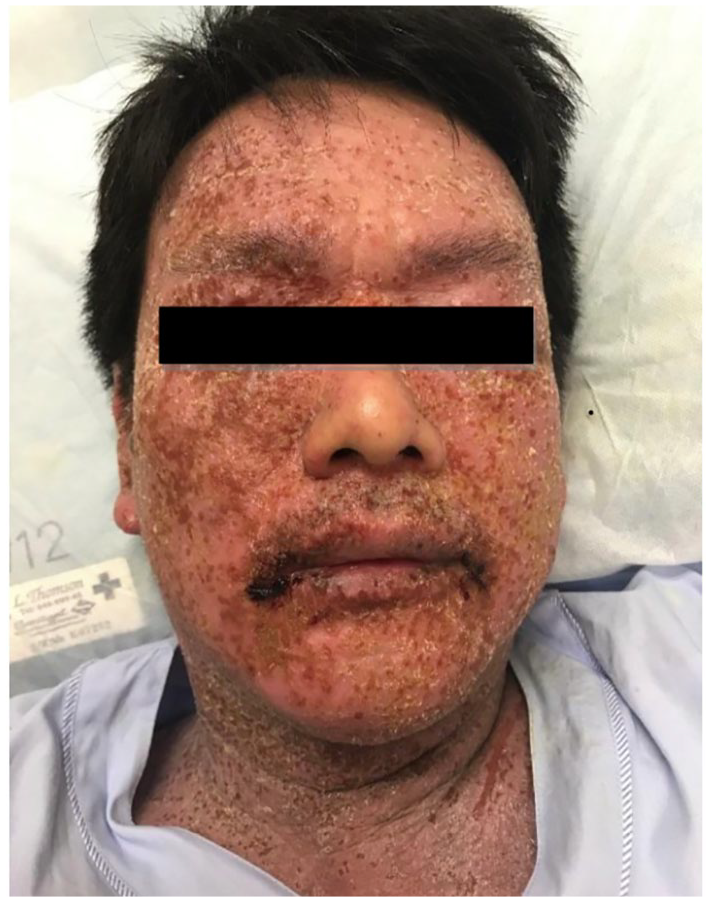
Widespread and confluent eruption of crusted erosions on the face. Hemorrhagic crusting is seen at the corners of the mouth.
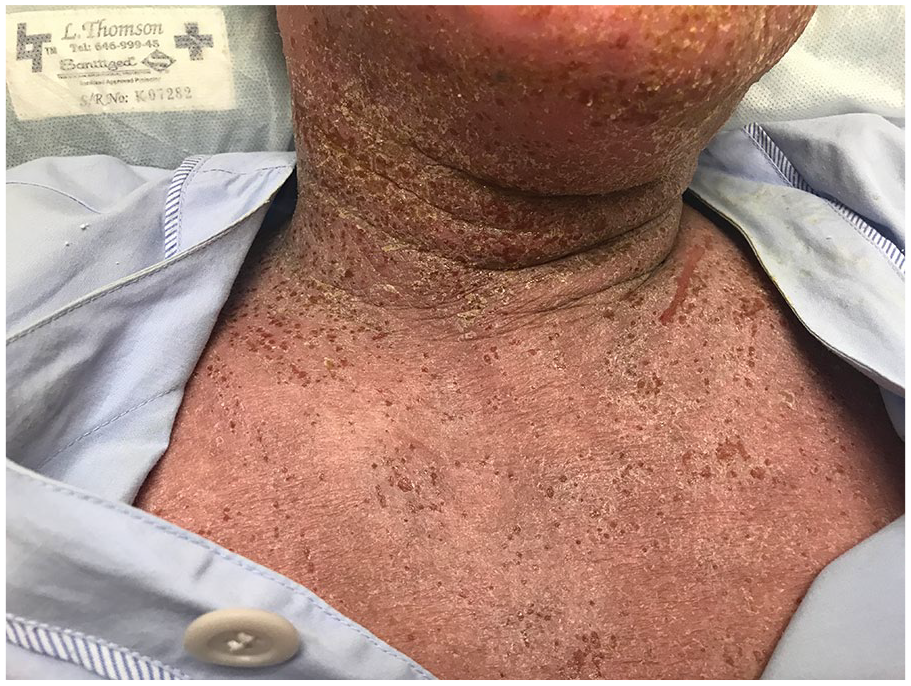
Close-up of monomorphic circular crusted erosions on a background of dry lichenified skin over the trunk.
On admission, his temperature was 39°C. Other vital signs were normal. Full blood count revealed a total white cell count of 5.12×109/L (reference 4.0–10.0×109/L), a neutrophil count of 80.0% (reference 40–75%), a lymphocyte count of 13.0% (reference 15–41%), an eosinophil count of 0% (reference 0–6%) and an atypical mononuclear cell of 2.0%. His procalcitonin level was 0.09 µg/L (reference <0.49µg/L) and his C-reactive protein was 51.9 mg/L (reference 0.2–9.1 mg/L). There was no intact blister to provide fluid culture or polymerase chain reaction (PCR) investigations.
He was reviewed by the dermatology team and diagnosed with EH with secondary impetiginisation. As he was clinically stable, he was treated with oral aciclovir 400 mg five times a day and oral cephalexin 500 mg t.i.d. for one week. He made an uneventful recovery and was thereafter commenced on oral methotrexate 10 mg q.w. This has resulted in good control of his eczema while reducing the risk of bacterial and viral infections.
Discussion
EH, otherwise known as Kaposi’s varicelliform eruption, is a dermatological emergency that must be urgently recognised for immediate systemic treatment. The clues to diagnosis are a history of atopic dermatitis, a rapidly progressing eruption, unique morphology of the eruption, predilection for facial area and fever. Although classically described as multiple clustered monomorphic dome-shaped punched-out vesicles, scratching gives rise to the clinical appearance of monomorphic circular erosions, as in this patient. Lesions often become secondarily impetiginised at the time of clinical presentation.
EH is a superimposed herpes infection on a background of atopic eczema. It is often due to HSV-1. 1 The virus is also known sometimes to superimpose over other dermatoses such as psoriasis, pemphigus vulgaris or burns. 1 One must not confuse the clinical presentation with Stevens–Johnson syndrome, in which true erosions develop on at least two mucosal surfaces in addition to the presence of erythema multiforme-like lesions with epidermal denudation occurring in <10% of the body surface area. It is instructive to recognise that EH lesions only occur over lesions of atopic dermatitis. 2
Risk factors for EH include an early onset of atopic dermatitis, severity of eczema and higher levels of serum immunoglobulin E. 3 Patients are more likely to have associated features of atopy (asthma and food allergy), as well as a history of other cutaneous infections. 4 Asians are also at higher risk of developing EH, and are more likely to suffer prolonged hospitalisation from this condition. 5
The pathophysiology of EH is multifactorial: epithelial dysfunction in the skin barrier (involving filaggrin), low production of antimicrobial peptides cathelicidin LL-37, overexpression of interleukins (IL-4, IL-13 and IL25) and beta-defensins and abnormal cell-mediated immunity. 6 SID1 transmembrane family member 2 loss-of-function mutations preventing keratinocytes from mounting host defense against HSV-1 has also been described. 7
While EH is an emergency, outcomes are good if it is identified and treated early. A nationwide paediatric retrospective study suggested that up to 98.2% of patients have only a minor risk of mortality, with more than two thirds suffering only minor loss of function. 5
The current gold standard investigation is HSV-1 PCR testing of blister fluid. However, in practice, intact vesicles containing fluid are typically seen only in the early stages of the condition. Samples taken from crusted lesions have low recovery rates of HSV-1. 8 A Tzanck smear may be performed, but it is neither sensitive nor specific. 9 Blood may be taken for HSV-1 immunoglobulin M levels, but antibodies can take up to 10 days to develop after a primary infection, and there is a high rate of false positives and negatives. 10
Astute clinical judgement and immediate empirical treatment remain the cornerstone of management. Patients who do not receive timely treatment may develop complications such as encephalitis, hepatitis and pneumonitis. For less sick patients, oral aciclovir 200–400 mg can be offered five times daily for a week. 11 For severely ill or immunocompromised patients, a regimen of intravenous aciclovir 5–15 mg/kg/day t.i.d. should be considered. 12 An alternative is 1 g oral valaciclovir t.i.d. 12 Anti-staphylococcal treatment is indicated if bacterial super-infection or impetiginisation is evident. 12
Up to 13% of patients recur after a first episode of EH. 3 Hence, for patients with atopic dermatitis who have recurrent orolabial or anogenital herpetic infection, oral prophylaxis with aciclovir 400 mg b.i.d. should be considered. 11 Oral valaciclovir 500 mg q.d. is a useful alternative in patients with recurrent EH. 13 While there have been suggested links between corticosteroid use and EH, the data remain inconclusive. In fact, the majority of patients with EH are not on corticosteroids. 3
Conclusion
In summary, one must have a high index of suspicion for EH in a patient with atopic dermatitis who develops an acute painful flare. The key aspects of diagnosis include a background of atopic dermatitis, systemic symptoms, rapid onset and morphological features of monomorphic punched-out lesions. Prompt systemic antiviral treatment is crucial to prevent complications and scarring. Chemoprophylaxis could be considered in patients with recurrent EH.
Footnotes
Acknowledgements
None.
Authors’ contributions
G.H.Z.W. was involved in the research and drafting of the manuscript. D.C.W.A. was involved in the selection of the patient and case, and in editing and fine-tuning the manuscript.
Availability of data and materials
Not applicable.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for his anonymised information to be published in this article.
