Abstract
Background
Chylothorax, defined as the accumulation of chyle in the pleural space, is a rare but significant cause of pleural effusion. Its causes can be broadly classified into traumatic and non-traumatic, for which majority are from surgeries, most often cardiothoracic surgery, due to damage of the thoracic duct and its tributaries. Chylothorax after retroperitoneal surgery, such as nephrectomy, is exceptionally rare, and so far has only been reported in a few case reports.
Research design
Here we present a case of a 67-year-old female with hyperlipidemia who developed chylothorax after an uncomplicated left robot-assisted partial nephrectomy for renal angiomyolipoma. On postoperative day 2 (POD2), she developed pleuritic chest pain and a left-sided pneumothorax, followed by increasing pleural effusion. A chest drain was inserted on POD5, and pleural fluid analysis revealed a chylous effusion with a triglyceride level of 15 mmol/L. The effusion was managed with dietary fat restriction, octreotide therapy, and pleural drainage. The chest drain was removed on POD15 after the drainage volume decreased to <30 mL/day for two consecutive days, and no recurrence was observed. Given the negative clinical, biochemical and imaging features for malignancy and lack of cystic lung disease and low VEGF-D level inconsistent with Lymphangioleiomyomatosis (LAM), nephrectomy was considered the most likely cause for the chylothorax.
Conclusion
This case highlights the importance of early diagnosis and a multidisciplinary approach in managing postoperative chylothorax, even in rare instances following retroperitoneal surgery.
Introduction
Chylothorax, defined as the accumulation of chyle in the pleural space, represents a rare cause of pleural effusion, accounting for up to 3% of cases. 1 It typically results from disruption, obstruction or damage to the thoracic duct and its tributaries. 2 The causes of chylothorax can be categorized into nontraumatic or traumatic. Nontraumatic cases being the minority, are mostly from malignancy, in particular lymphoma.3,4
Traumatic chylothorax constitutes the majority of chylothorax cases and is primarily attributed to surgery, with cardiothoracic and esophageal surgeries being the most common causes.1,2 Chylothorax post retroperitoneal or abdominal surgery is rare, and has only been documented in a few case reports. 5 Here we present a case of chylothorax after nephrectomy for renal angiomyolipoma.
Case
A 67-year-old Chinese lady with a significant past medical history of hyperlipidaemia and depression underwent uncomplicated left robot-assisted partial nephrectomy for a left renal angiomyolipoma (AML). There was no report of diaphragmatic puncture or injury intra-operatively.
On post-operative day 2 (POD2), she developed left sided pleuritic chest pain. She denied other associated symptoms of cough, sputum production, dyspnoea, wheeze and palpitations.
On physical examination, her blood pressure was 130/84, heart rate was 84 beats/min and regular, respiratory rate 18 breaths/min, temperature 37.1°C, and oxygen saturation was 94% on ambient air. Auscultation of the lungs were normal with bilateral vesicular breath sounds, no wheeze was present. A chest radiograph detected a left pneumothorax of 2.1 cm in the apex-cupola distance (Figure 1(a)). Left to Right: (a) CXR on POD2 showing a left pneumothorax. (b) CXR on POD3 showing decrease in size of the left pneumothorax and a new small left pleural effusion. (c) CXR on POD4 showing increase in size or of the left pleural effusion.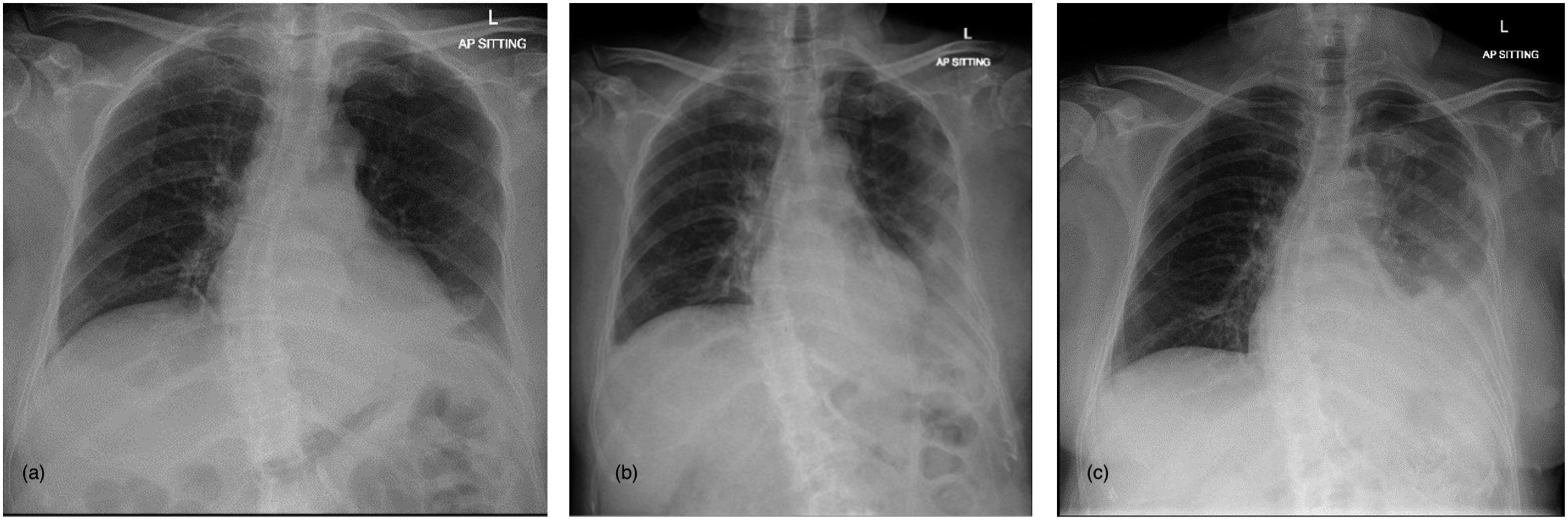
She was initially treated conservatively with observation and supplemental oxygen which showed interval improvement in size of her pneumothorax (Figure 1(b)).However, her chest pain persisted and subsequent chest radiographs detected an increasing left pleural effusion (Figure 1(c)). A CT Pulmonary Angiogram was done on POD4 (Figure 2) which confirmed the presence of a left hydropneumothorax with a large low-density effusion component and a small pneumothorax component, with associated collapse of the left lower lobe. No pulmonary embolus, masses, or lung cysts were detected. A left chest drain was inserted on POD5 for diagnostic and therapeutic purposes with resolution of patient’s symptoms (Figure 3). CT Pulmonary Angiogram on POD4 showing a left hydropneumothorax with a low-density effusion and a small pneumothorax component. Ultrasound image during left pleural drain catether insertion showing the left pleural effusion.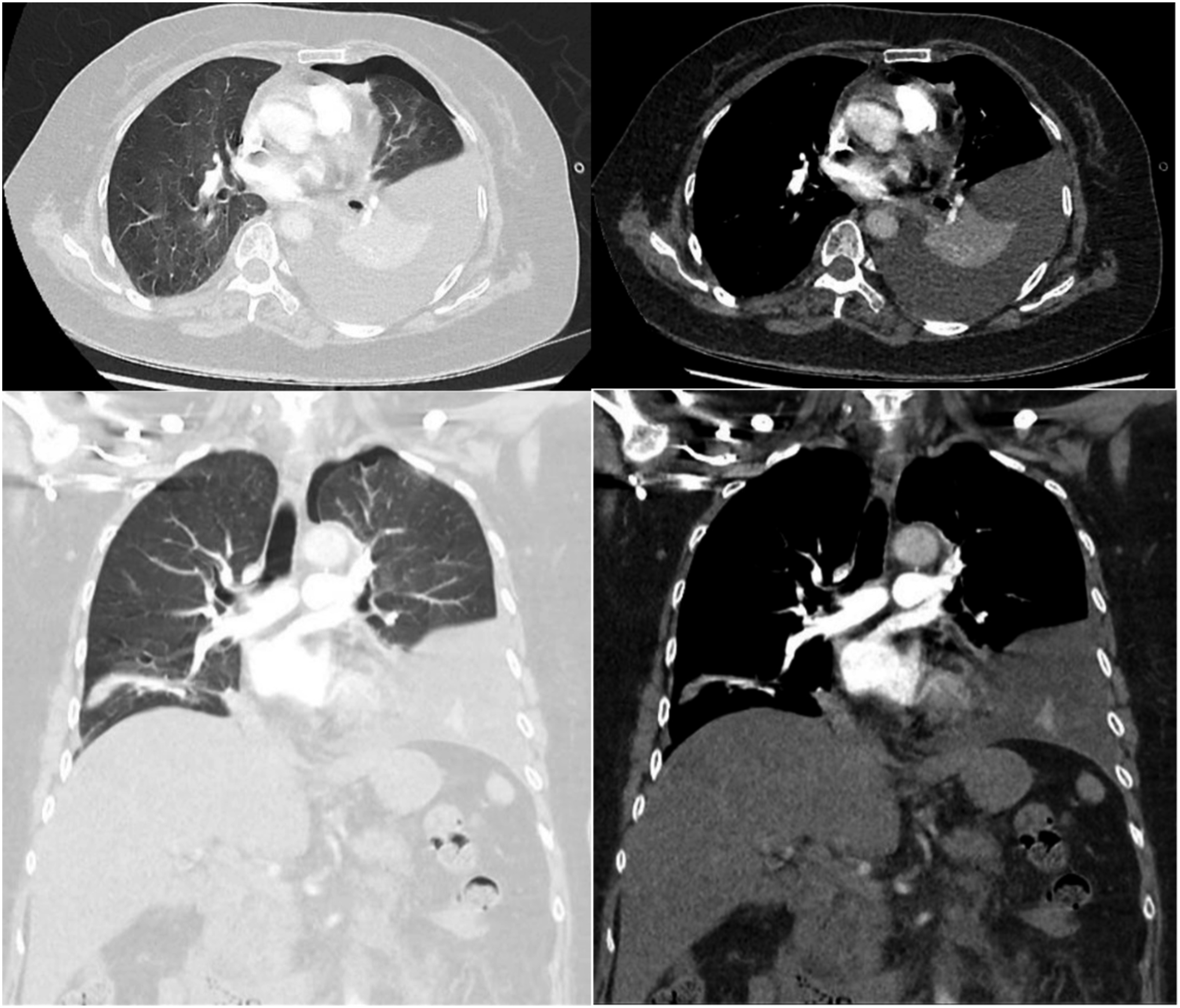

Thick, milky pleural fluid was drained from the left pleural space. Pleural fluid analysis showed a lymphocyte predominant (lymphocytes 75%, neutrophils 25%), exudate (pleural protein 41.6 g/L, serum protein 60 g/L; pleural lactate dehydrogenase (LDH) 491U/L, serum LDH 270U/L). Pleural fluid pH was >7.80, glucose 7.4 mmol/L and adenosine deaminase, ADA 4.0IU/L. Most significantly, pleural fluid triglyceride level was 15 mmol/L and total cholesterol was 1.71 mmol/L, indicative of a chylous effusion. Microbiological studies were negative, including gram stain, bacterial culture, acid-fast bacilli stain, and acid-fast bacilli culture. No malignancy cells were detected on cytological examination of the pleural fluid. Histology of the left renal mass was confirmed to be AML. Lymphoma screen panel from blood was negative for clonal lymphoproliferative disorder.
Subsequent cross sectional imaging of the abdomen and pelvis did not reveal any abdominal masses, ascites or collections. A vascular endothelial growth factor (VEGF) level was sent and returned negative. Of note, the patient did not have any clinical features of tuberous sclerosis. She declined lymphoscintigraphy.
Upon diagnosis of chylothorax, multi-disciplinary discussion was conducted involving urologists, respiratory physicians, and general surgeons on the possible etiology as well as management. After discussion, the patient was kept nothing by mouth and initiated on subcutaneous octreotide. This successfully reduced the volume of chyle production and chest drain output decreased from 800 mL/day to <30 mL/day. A medium chain triglyceride diet was introduced as the chest drain output decreased. The chest drain was removed on POD15 (12 days after insertion)when the output was <30 mL/day for 2 consecutive days. Repeated chest radiographs at 1 week (Figure 4(a)) and 3 months (Figure 4(b)) after chest drain removal did not show evidence of re-accumulation of the chylothorax. Left to Right: (a) CXR 1 week after chest drain removal. (b) CXR 3 months after chest drain removal. No recurrence in pleural effusion was detected.
Discussion
Chylothorax occurs due to chyle leakage from the lymphatic system and its accumulation in the pleural space. Chyle is composed of lipids, electrolytes, proteins, and lymphocytes. 1 The effusion is usually unilateral, with right sided (50%) being more than left sided (33.3%), but less commonly bilateral (16.66%), which depend on the location of the leak. 6 Chyle leak can occur due to disruption, obstruction, or damage in the thoracic duct (TD), cisterna chyli, or its tributaries. The cisterna chyli is located at the level of the renal vessels, and is prone to injury in retroperitoneal surgery.2,5
Chylothorax can be classified as traumatic or non-traumatic. Traumatic causes can be further grouped into iatrogenic versus non-iatrogenic from penetration or blunt injuries. Amongst the iatrogenic chylothorax, esophagectomy has a reported incidence of 0.4–4%, 7 followed by other thoracic surgeries such as pulmonary resection or catheterisation of the subclavian vein.3,7 Chylothorax occurring after retroperitoneal or abdominal surgeries is extremely rare and is only reported in limited case reports.2,5 A delay of 2 to 7 days can be observed before clinical signs of chylothorax become evident in non-major injuries. This delay occurs because lymph fluid initially accumulates in the posterior mediastinum until it causes a rupture of the mediastinal pleura. 3
Non-traumatic causes are relatively uncommon and encompass malignancies, sarcoidosis, amyloidosis, retrosternal goitre, superior vena cava thrombosis, benign tumours, congenital duct abnormalities and diseases of the lymphatics such as yellow nail syndrome, Lymphangioleiomyomatosis (LAM), and haemagiomatosis. 6 Amongst these, the most prevalent cause of non-traumatic chylothorax is the obstruction of the thoracic duct due to malignancies, with lymphoma accounting for approximately 70% of cases. 6 LAM typically affects females of childbearing age. It is characterized by the abnormal growth of smooth muscle cells in the lungs and can be associated with extrapulmonary manifestations. Chylothorax may be observed in up to 40% of LAM patients in their lifetime. Affected patients also may present with nodular interstitial proliferation of the smooth muscle in the lungs, lymph nodes, and thoracic duct, as well as cystic changes in the lungs. Extrapulmonary findings include benign renal angiomyolipomas, and perivascular epithelioid cell tumors with visceral organ involvement.1,3,8 Although no definitive biomarkers are available for the diagnosis of LAM, some studies suggest the use of vascular endothelial growth factor D (VEGF-D) in patients with suspected LAM. They reported that VEGF-D levels greater than or equal to 800 pg/mL have a high sensitivity and specificity for pulmonary lymphangioleiomyomatosis.8,9 The case we presented was diagnostically challenging in terms of determination of the aetiology, given the low incidence of nephrectomy causing chylothorax, and lack of evidence to demonstrate any obvious injury to the TD or its tributaries intra-operatively. Moreover, the presence of kidney AML and nodular lung changes with chylothorax made LAM a reasonable differential. In her case, the lack of cystic lung disease and low VEGF-D level argued against LAM. Malignancy was also considered, but given the lack of other clinical, biochemical and imaging features suggestive of malignancy such as lymphoma and the negative flow cytometry, malignancy induced chylothorax was unlikely in her case. Taking into account the timeline of the chylothorax developing 2 days after the nephrectomy, surgery was considered the most likely cause in her case.
Often described as ‘milky’, the gross appearance of pleural fluid can give a clue of the presence of chylothorax, but it is neither sensitive nor specific, as about 50% of chylothorax may demonstrate effusions that are bloody, turbid, serous, or serosanguineous,3,10 and pseudochylothorax or empyema can also cause a milky appearance of the effusion. 3 Chylothorax is diagnosed by fluid profile with a triglyceride (TG) level >1.24 mmol/Land a cholesterol level <5.18 mmol/L. When the above levels are intermediate, definitive diagnosis of chylothorax can be demonstrated by the presence of chylomicrons in the pleural fluid from lipoprotein electrophoresis.1,3,4,11 In our case, patient’s fluid studies showed a typical lymphocytic picture with a clearly elevated triglyceride level, thus lipoprotein electrophoresis was not performed.
Management generally is divided into conservative and invasive approaches. Conservative measures encompass dietary modification with no or low-fat diet. Pharmacological agents include somatostatin analog octreotide, alpha-adrenergic agonists, and drugs targeting at the underlying cause if any can be identified. 4 Interventions are indicated for symptom relief, or in traumatic cases for definitive closure of the injury, as well as for patients with a persistent high output or prolonged leak. In our case, the patient was initially conservatively managed with observation for symptoms, given then the suspicion for chylothorax was low, and the pleural effusion was considered more likely secondary to resolving pneumothorax. However, her symptom persisted with increasing size of the pleural effusion, thus pleural drainage was performed, resulting in rapid improvement of her chest pain. She also received dietary fat elimination and octreotide after chylothorax was confirmed.
Prognosis of chylothorax is variable and depends on the aetiology. It is reported that chylothorax from benign causes tend to have better outcomes. Bilateral chylothoraces, malignant chylothorax and chronic chylothorax with nutrient loss are reported as poor prognostic factors. 1 Our patient’s pleural effusion resolved after 12 days of drainage. She has none of the poor prognostic factors so far, although continued follow up is warranted.
Conclusion
We present a case of chylothorax after nephrectomy for renal angiomyolipoma. Chylothorax is extremely rare post nephrectomy, but should be considered especially if there is evidence of injuries to the thoracic duct, cisterna chyli, or its tributaries intra-operatively. Our case suggests that pleural drainage and octreotide in combination with a medium chain triglyceride diet may be effective in the management of chylothorax post nephrectomy, avoiding the need for surgery.
Footnotes
Acknowledgements
The authors would like to express our gratitude to our fellow colleagues for their shared care of this patient.
Author contributions
First author wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Ethical approval
Ethical approval was not sought for the present study because the institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymised information to be published in this article.
Trial registration
Not applicable because it is not a trial.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
